Unit 13 Acids and Bases Properties electrolyte sour

Unit 13 Acids and Bases

Properties • • electrolyte sour taste sticky feel turn litmus red • • electrolyte bitter taste slippery feel turn litmus blue • react with bases to • react with acids to form water and a salt (ionic compound)

Examples ACIDS: Most citrus fruits, tea, battery acid, vinegar, milk, soda, apples. BASES: Common household bases include baking soda, lye, ammonia, soap, and antacids. .

Indicators • Indicators are substances that change color in the presence of an acid or a base – Indicators are made up of weak acids or weak bases – Examples of indicators include p. H paper, red and blue litmus paper, and phenolphthalein

Acids Affect Indicators: Blue litmus paper turns red in contact with an acid. It remains blue when in contact with a base or neutral solution.

Bases affect indicators: Red litmus paper turns blue in contact with a base. It remains red when in contact with an acid or neutral solution. Phenolphthalein turns pink in a base. It is colorless in an acid or neutral solution.

Definitions • There are 3 definitions used to describe acids and bases: • Arrhenius • BrØnsted-Lowry • Lewis • The most traditional is Arrhenius acids and bases.
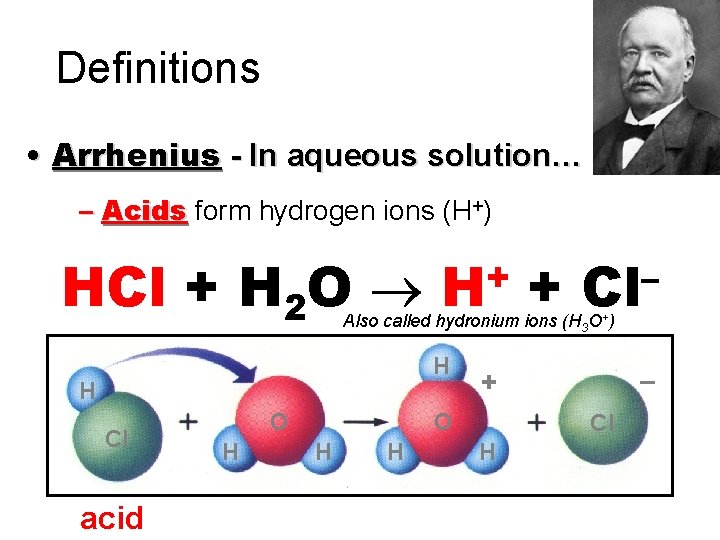
Definitions • Arrhenius - In aqueous solution… – Acids form hydrogen ions (H+) HCl + H 2 O + H + – Cl Also called hydronium ions (H 3 O+) H H Cl acid O H H – + Cl H
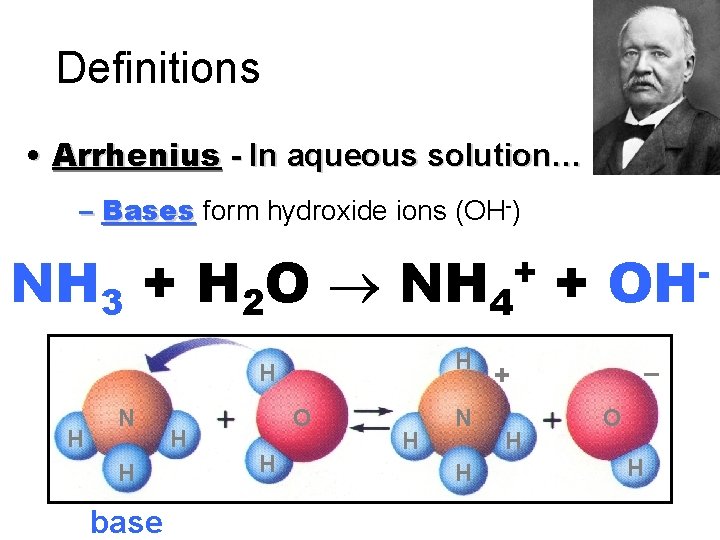
Definitions • Arrhenius - In aqueous solution… – Bases form hydroxide ions (OH-) NH 3 + H 2 O NH 4 + + H H H N H base H O H H N H OH – + H O H
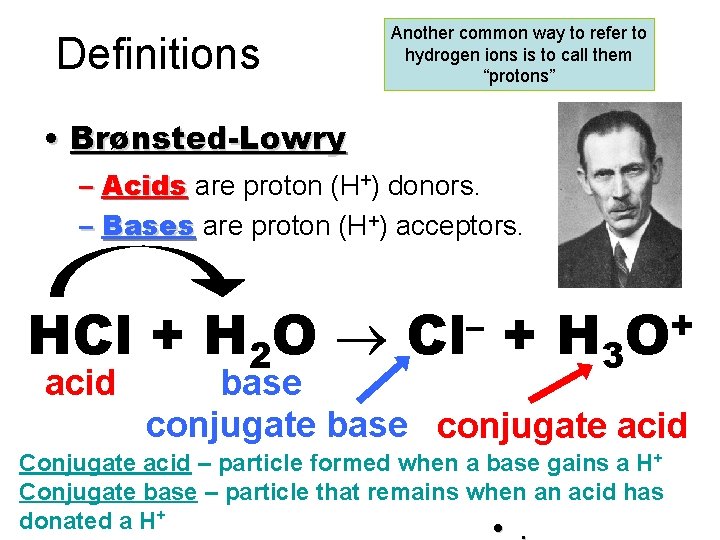
Definitions Another common way to refer to hydrogen ions is to call them “protons” • Brønsted-Lowry – Acids are proton (H+) donors. – Bases are proton (H+) acceptors. HCl + H 2 O acid – Cl + H 3 + O base conjugate acid Conjugate acid – particle formed when a base gains a H+ Conjugate base – particle that remains when an acid has donated a H+ • .
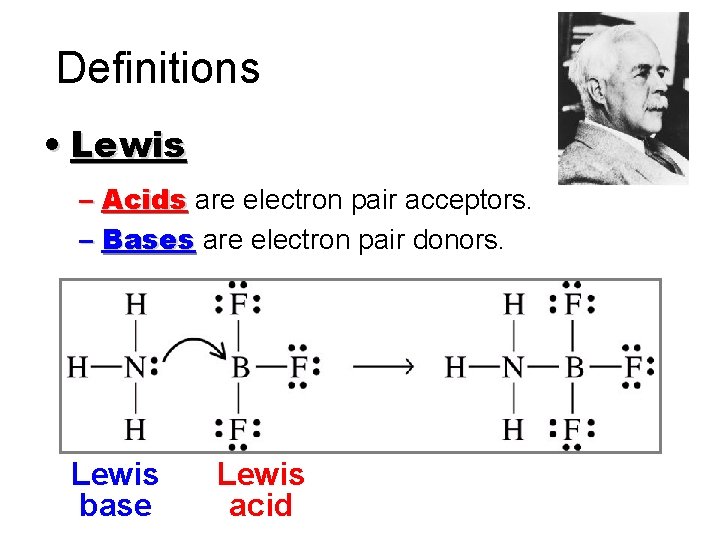
Definitions • Lewis – Acids are electron pair acceptors. – Bases are electron pair donors. Lewis base Lewis acid

White Board Questions 1. When you wafted a substance your nose burned. Would this substance be an acid or a base? ACID 2. A hydrogen ion (H+) can also be called a Proton H 3 O + _____ or ______. 3. Arrhenius acids are compounds that break H+ up in water to give off _______. 4. What color litmus paper would you use to Blue turns red test an acid? What color will it turn? 5. If your food tastes bitter, which do you think it could possibly be an acid or a base? BASE

White Board Questions accepts 6. A BrØnsted-Lowry base _____ hydrogen ions. 7. Phenolphthalein turns pink when it comes in base contact with a(n) _____. 8. Which of the scientists defined the typical acid? Arrhenius 9. If you are eating and it has a sour taste, would that be an acid or a base? acid 10. If a piece of red litmus paper turns blue than it base is a(n) ______.

Naming Acids • Binary acids – Contains 2 different elements: H and another – Always has “hydro-” prefix – Root of other element’s name – Ending “-ic”

Examples of Binary Acids • • HI is hydroiodic acid H 2 S is hydrosulfuric acid HBr is hydrobromic acid HCl is hydrochloric acid

Naming Acids • Ternary Acids - Oxyacids – Contains 3 different elements: H, O, and another – No prefix – Name of polyatomic ion (p. 147) – Ending “–ic” for polyatomic ion ending in “-ate” and “–ous” for ion ending in “-ite”

Examples of Ternary Acids • Cl. O 3 is chlorate so HCl. O 3 is chloric acid • PO 4 is phosphate so H 3 PO 4 is phosphoric acid • PO 3 is phosphite so H 3 PO 3 is phosphorous acid • NO 2 is nitrite HNO 2 is nitrous acid • NO 3 is nitrate HNO 3 is nitric acid

Naming Acids cont. • HC 2 H 3 O 2 or CH 3 COOH Name is acetic acid Common name = vinegar

Practice Naming Acids • H 2 SO 3 – Sulfurous acid • HF – Hydrofluoric acid • H 2 Se – Hydroselenic acid • Perchloric acid – HCl. O 4 • Carbonic acid – H 2 CO 3 • Hydrobromic acid – HBr

Ion Product of Water Self- ionization of water – the simple dissociation of water H 2 O H+ + OH- Concentration of ea. ion in pure water: [H+] = 1. 0 x 10 -7 M + [OH-] = 1. 0 x 10 -7 M Ion-product constant for water (Kw), Where Kw = 1. 0 x 10 -14 Kw = [H+] [OH-] Acid [H+] > [OH-] Base [H+] < [OH-] Neutral [H+] = [OH-]
![Calculating [H+] and [OH-] • reverse the p. H equation [H+] = 1 x Calculating [H+] and [OH-] • reverse the p. H equation [H+] = 1 x](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-21.jpg)
Calculating [H+] and [OH-] • reverse the p. H equation [H+] = 1 x 10 -p. H and [OH-] = 1 x 10 -p. OH • The p. H of a solution is 8. Find the [H+] and [OH-] and determine whether it is acidic, basic, or neutral. [H+] = 1 x 10 -8 M [OH-] = 1 x 10 -(14 -8) M = 1 x 10 -6 M – basic
![Examples 1. If the [H+] in a solution is 1. 0 x 10 -5 Examples 1. If the [H+] in a solution is 1. 0 x 10 -5](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-22.jpg)
Examples 1. If the [H+] in a solution is 1. 0 x 10 -5 M, is the solution acidic, basic or neutral? 1. 0 x 10 -5 M p. H 5 = acidic What is the concentration of the [OH-]? Use the ion-product constant for water (Kw): Kw = [H+] [OH-] 1. 0 x 10 -14 = [1. 0 x 10 -5] [OH-] 1. 0 x 10 -14 = [OH-] 1. 0 x 10 -5 1. 0 x 10 -(14 -5) 1. 0 x 10 -9 M

Examples 2. If the p. H is 9, what is the concentration of the hydroxide ion? 14 = p. H + p. OH Kw = [H+] [OH-] 1. 0 x 10 -14 M = [1. 0 x 10 -9 M] [OH-] 14 = 9 + p. OH 1. 0 x 10 -5 M = [OH-] 5 = p. OH 3. If the p. OH is 4, what is the concentration of the hydrogen ion? 14 = p. H + p. OH Kw = [H+] [OH-] 1. 0 x 10 -14 M = [H+] [1. 0 x 10 -4 M] 14 = p. H + 4 1. 0 x 10 -10 M = [H+] 10 = p. H

Examples 4. A solution has a p. H of 4. Calculate the p. OH, [H+] and [OH-]. Is it acidic, basic, or neutral? 14= p. H + p. OH 14= 4 + p. OH 10= p. OH –Acidic since p. H is 4
![Practice Problems: Classify each solution as acidic, basic or neutral. 1. [H+] = 1. Practice Problems: Classify each solution as acidic, basic or neutral. 1. [H+] = 1.](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-25.jpg)
Practice Problems: Classify each solution as acidic, basic or neutral. 1. [H+] = 1. 0 x 10 -10 M Basic p. H 10 2. [H+] = 0. 001 M 1. 0 x 10 -3 acid p. H 3 3. [OH-] = 1. 0 x 10 -7 M Neutral 4. [OH-] = 1. 0 x 10 -4 M 14=p. H+4 base p. H 10
![[OH-] p. OH p. H [H+] 1 x 10 -14 14 0 1 x [OH-] p. OH p. H [H+] 1 x 10 -14 14 0 1 x](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-26.jpg)
[OH-] p. OH p. H [H+] 1 x 10 -14 14 0 1 x 10 -13 13 1 1 x 10 -12 12 2 1 x 10 -11 11 3 1 x 10 -10 10 4 1 x 10 -9 9 5 1 x 10 -8 8 6 1 x 10 -7 7 7 1 x 10 -6 6 8 1 x 10 -5 5 9 1 x 10 -4 4 10 1 x 10 -3 3 11 1 x 10 -2 2 12 1 x 10 -1 1 13 1 x 10 -13 1 x 100 0 14 1 x 10 -14 Increasing acidity Neutral Increasing basicity
![White Board Practice Fill in the chart. [OH-] p. OH p. H [H+] 1. White Board Practice Fill in the chart. [OH-] p. OH p. H [H+] 1.](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-27.jpg)
White Board Practice Fill in the chart. [OH-] p. OH p. H [H+] 1. 0 X 10 -8 8 6 1. 0 X 10 -2 2 12 1 x 10 -12 1. 0 X 10 -4 4 10 1. 0 X 10 -10 3 11 1. 0 X 10 -11 1 x 10 -3
![Fill in the chart. [OH-] p. OH p. H [H+] 1. 0 X 10 Fill in the chart. [OH-] p. OH p. H [H+] 1. 0 X 10](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-28.jpg)
Fill in the chart. [OH-] p. OH p. H [H+] 1. 0 X 10 -8 8 6 1. 0 X 10 -2 2 12 1 x 10 -12 1. 0 X 10 -4 4 10 1. 0 X 10 -10 3 11 1. 0 X 10 -11 9 1. 0 X 10 -9 1 1 × 10 -1 1 x 10 -3 1. 0 X 10 -5 1. 0 X 10 -13 5 13
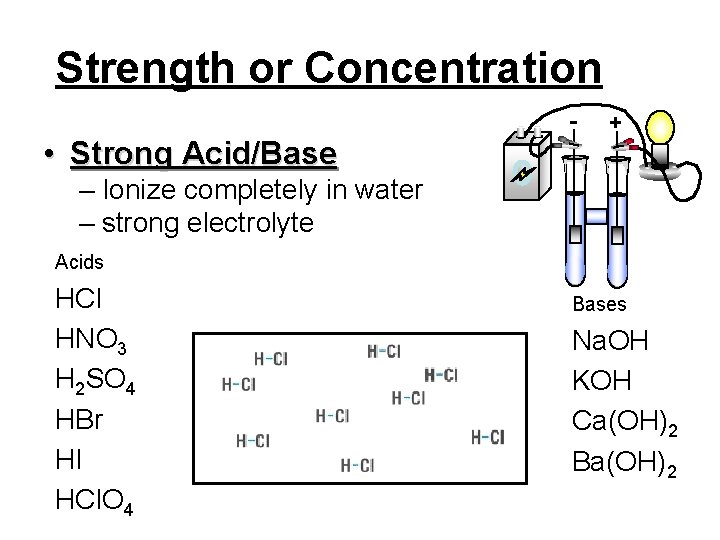
Strength or Concentration - + • Strong Acid/Base – Ionize completely in water – strong electrolyte Acids HCl HNO 3 H 2 SO 4 HBr HI HCl. O 4 Bases Na. OH KOH Ca(OH)2 Ba(OH)2
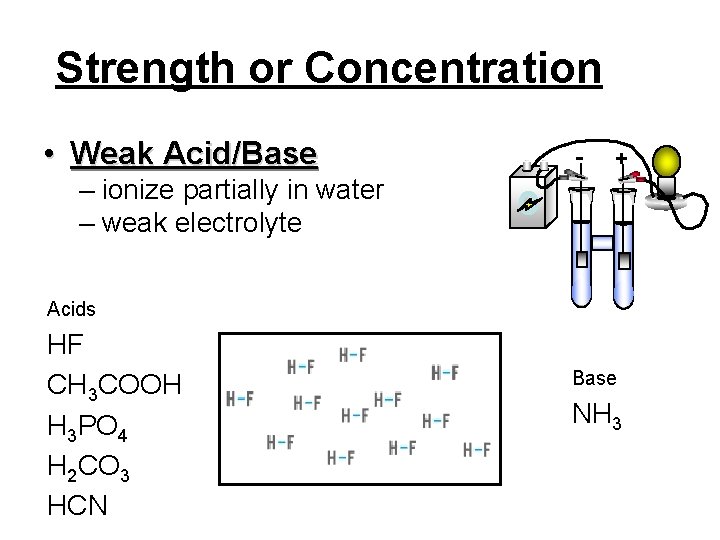
Strength or Concentration • Weak Acid/Base - + – ionize partially in water – weak electrolyte Acids HF CH 3 COOH H 3 PO 4 H 2 CO 3 HCN Base NH 3
Strength or Concentration • How strong or weak an acid or base is, depends on its degree of ionization. - +
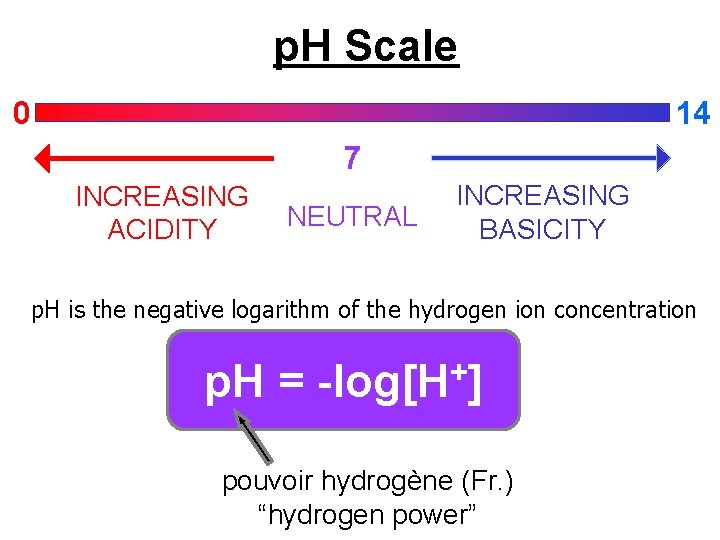
p. H Scale 14 0 7 INCREASING ACIDITY NEUTRAL INCREASING BASICITY p. H is the negative logarithm of the hydrogen ion concentration p. H = + -log[H ] pouvoir hydrogène (Fr. ) “hydrogen power”
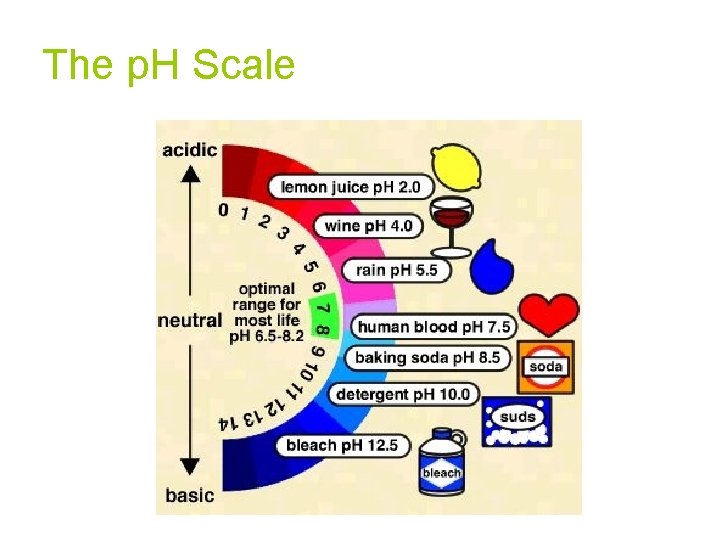
The p. H Scale
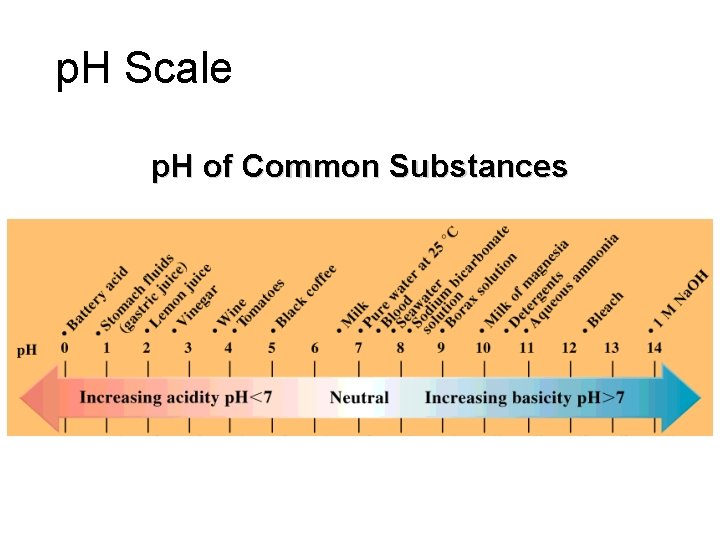
p. H Scale p. H of Common Substances
![p. H formulas p. H = -log[H+] p. OH = -log[OH-] p. H + p. H formulas p. H = -log[H+] p. OH = -log[OH-] p. H +](http://slidetodoc.com/presentation_image/5cff5d2f99aa4fa96d14b27f09ca3c96/image-35.jpg)
p. H formulas p. H = -log[H+] p. OH = -log[OH-] p. H + p. OH = 14

Neutralization • Chemical reaction between an acid and a base. • Products are a salt (ionic compound) and water.

Neutralization ACID + BASE SALT + WATER HCl + Na. OH Na. Cl + H 2 O strong neutral HC 2 H 3 O 2 + Na. OH Na. C 2 H 3 O 2 + H 2 O weak strong basic – Salts can be neutral, acidic, or basic. – Neutralization does not mean p. H = 7.

Titration standard solution • Titration – Analytical method in which a standard solution is used to determine the concentration of an unknown solution
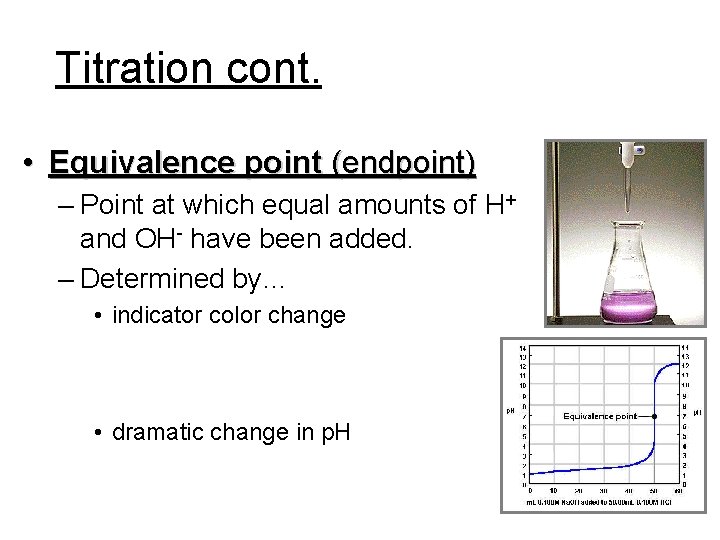
Titration cont. • Equivalence point (endpoint) – Point at which equal amounts of H+ and OH- have been added. – Determined by… • indicator color change • dramatic change in p. H

Titration formula + H moles = moles M V n = M V n OH M: Molarity V: volume n: # of H+ ions in the acid or OH- ions in the base

Titration example • 42. 5 m. L of 1. 3 M KOH are required to neutralize 50. 0 m. L of H 2 SO 4. Find the molarity of H 2 SO 4 MVn = MVn M=? M = 1. 3 M M(50. 0 m. L)(2)=(1. 3 M)(42. 5 m. L)(1) V = 50. 0 m. LV = 42. 5 m. L n=2 KOH n=1 M= 55. 25 100 M = 0. 55 M H 2 SO 4

Review of Acid and Base Definitions • Arrhenius Most specific/exclusive definition Created by Svante Arrhenius, Swedish Acid: compound that creates H+ in an aqueous solution HNO 3 H+ + NO 3 Base: compound that creates OH- in an aqueous solution Na. OH Na+ + OH-

Review of Acid and Base Definitions • Bronsted-Lowry More general definition than Arrhenius definition Most commonly used definition Created by 2 scientists around the same time (1923) Acid: Molecule or ion that is a proton (H+) donor HCl + H 2 O H 3 O+ + Cl. Base: Molecule or ion that is a proton (H+) acceptor NH 3 + H 2 O NH 4+ + OH-

Review of Acid and Base Definitions • Lewis Most general definition Defined by electrons and bonding rather than H+ Created by the same scientist who electron-dot diagrams are named after Acid: atom, ion, or molecule that accepts an electron pair to form a covalent bond NH 3 + Ag+ [Ag(NH 3)2]+ Base: atom, ion, or molecule that donates an electron pair to form a covalent bond BF 3 + F- BF 4 -
- Slides: 44