Unit 11 Organic Chemistry Bonding In Carbon Atoms

Unit 11 Organic Chemistry

Bonding In Carbon Atoms � Covalent � Carbon always forms 4 bonds � Can form both polar and non-polar compounds � Being organic means you have carbon � Non-electrolytes � Molecular Solids � Low Melting points and Low boiling points


Hydrocarbons � Compounds that are made of just hydrogen and carbon � Form the backbone of many types of organic compounds � The hydrogen atoms of a hydrocarbon can be replaced with other atoms or bonds to form new organic compounds
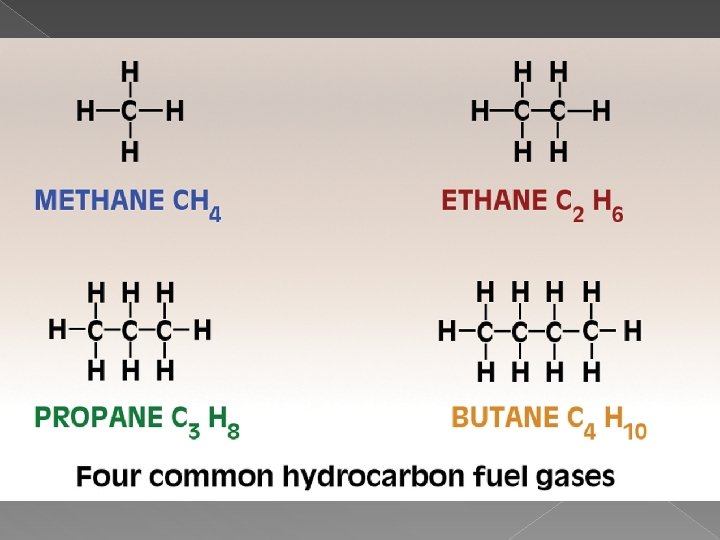

Properties of Hydrocarbons � Covalent � Polar Bonds but Non-Polar molecules � Soft Solids(Think Butter) or Liquids, or gases at room temperature � Bonds are weak � Contain a lot of energy in the bonds and so they are used as fuels


Alkanes � Hydrocarbons that contain all single Hydrogen and Carbon Bonds � These are SATURATED because every carbon is using the maximum number of bonds (all single bonds) � Used primarily for energy � More carbon = more energy � More carbon = higher MP and BP � More carbon = stronger structure


Saturated Vs. Unsaturated � Saturated hydrocarbons contain the maximum number of bonds possible � All single bonds, no unused spaces � In nutrition, these are the bad stored fats that we are told to avoid � Alkanes are saturated hydrocarbons

Unsaturated � These are hydrocarbons that do not have the maximum number of bonds � In these molecules we have spaces created by double or triple bonds within the chain � Alkenes and Alkynes are unsaturated � Nutritionally these are the “good” fats that we should be getting in our diets


Alkenes � Hydrocarbon with at least ONE carbon double bond � Double bond makes these stronger than alkanes � Have higher melting and boiling points � Position of the double bond needs to be indicated in the name � Start with Ethene because of we need at least two carbons for the 2 x bond


How do I name an Alkene? � Carbon with the double bond MUST have the lowest number possible � Once this is established, name and number all groups making sure the double bond keeps its lowest number � All alkenes end with “ene” � Can be branched

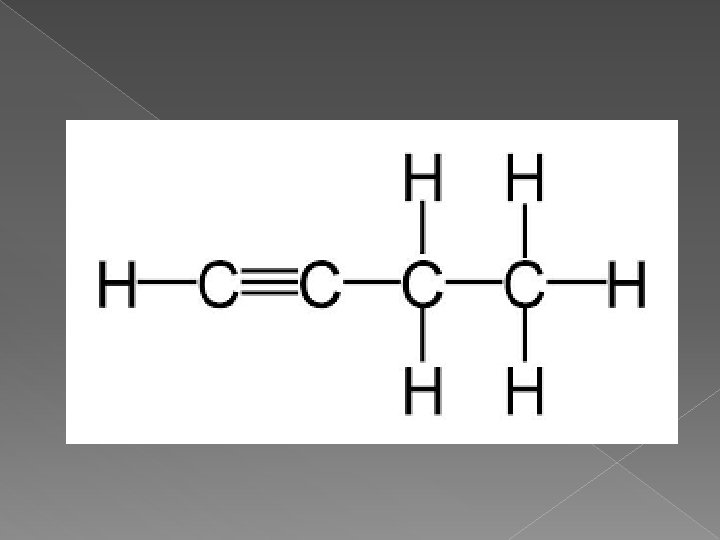
Alkynes � Hydrocarbons that contain at least one carbon-carbon triple bond � End with “yne” � These are the strongest hydrocarbons due to the triple bone � Have high melting points and high boiling points � Start with ethyne because we need at least 2 carbons for the 3 x bond
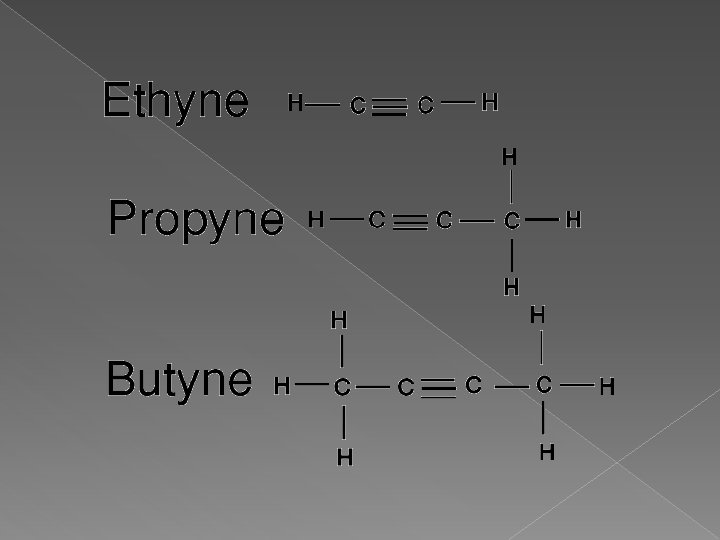

How do I name an Alkyne � Carbon with the triple bond must have the LOWEST number possible in the chain � Once this is established, name and number all attached groups as before � Check to make sure the triple bond has the lowest number � Name with location numbers and end with “yne”
- Slides: 19