Unit 11 Equilibrium Acids and Bases reversible reaction

Unit 11: Equilibrium / Acids and Bases reversible reaction: R P and R Acid dissociation is a reversible reaction. H 2 SO 4 2 H+ + SO 42– P
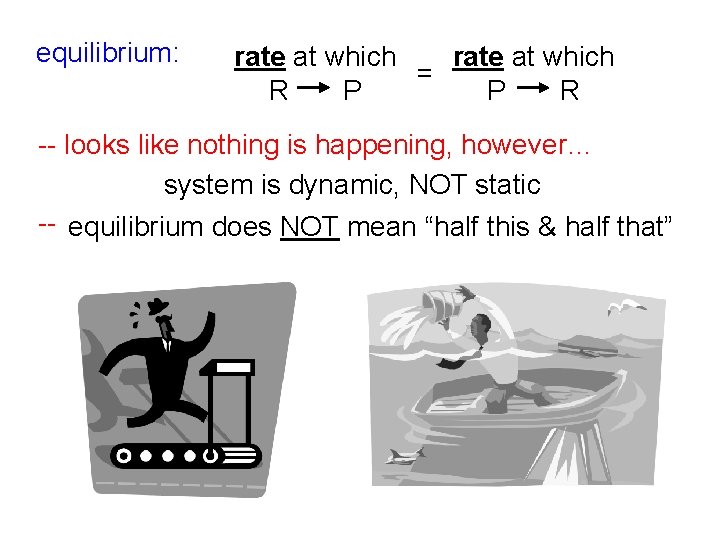
equilibrium: rate at which = R P P R -- looks like nothing is happening, however… system is dynamic, NOT static -- equilibrium does NOT mean “half this & half that”

Le Chatelier’s principle: When a system at equilibrium is disturbed, it shifts to a new equilibrium that counteracts the disturbance. N 2(g) + 3 H 2(g) Disturbance 2 NH 3(g) Equilibrium Shift Add more N 2……………… Add more H 2……………… Add more NH 3……………. Remove NH 3……………… Add a catalyst……………. . no shift Increase pressure…………
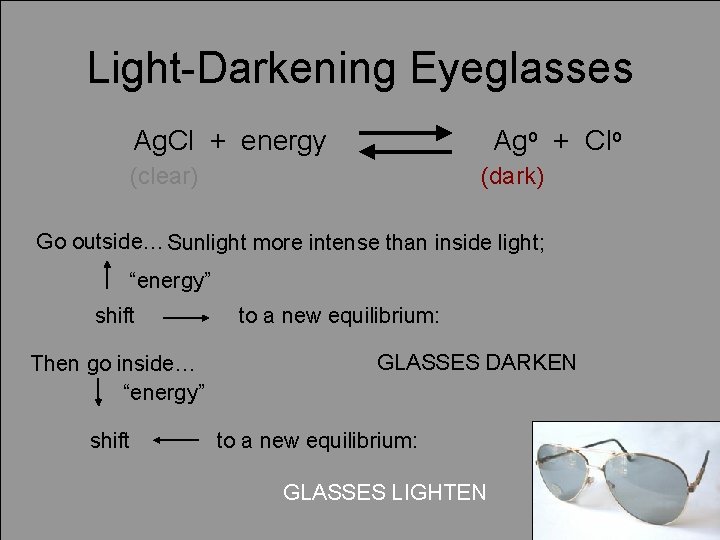
Light-Darkening Eyeglasses Ag. Cl + energy Ago + Clo (clear) (dark) Go outside… Sunlight more intense than inside light; “energy” shift Then go inside… “energy” shift to a new equilibrium: GLASSES DARKEN to a new equilibrium: GLASSES LIGHTEN
![In a chicken… Ca. O + CO 2 In summer, [ CO 2 ] In a chicken… Ca. O + CO 2 In summer, [ CO 2 ]](http://slidetodoc.com/presentation_image_h2/75d8a48dfefc17380a969352422b9b10/image-5.jpg)
In a chicken… Ca. O + CO 2 In summer, [ CO 2 ] in a chicken’s blood due to panting. Ca. CO 3 (eggshells) I wish I had sweat glands. -- shift ; eggshells are thinner How could we increase eggshell thickness in summer? -- give chickens carbonated water [ CO 2 ] , shift -- put Ca. O additives in chicken feed [ Ca. O ] , shift
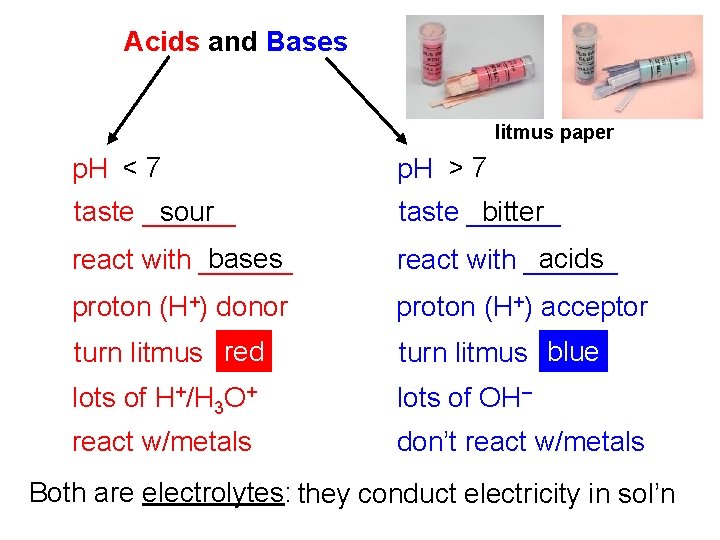
Acids and Bases litmus paper p. H < 7 p. H > 7 sour taste ______ bitter taste ______ bases react with ______ acids react with ______ proton (H+) donor proton (H+) acceptor turn litmus red turn litmus blue lots of H+/H 3 O+ lots of OH– react w/metals don’t react w/metals Both are electrolytes: they conduct electricity in sol’n
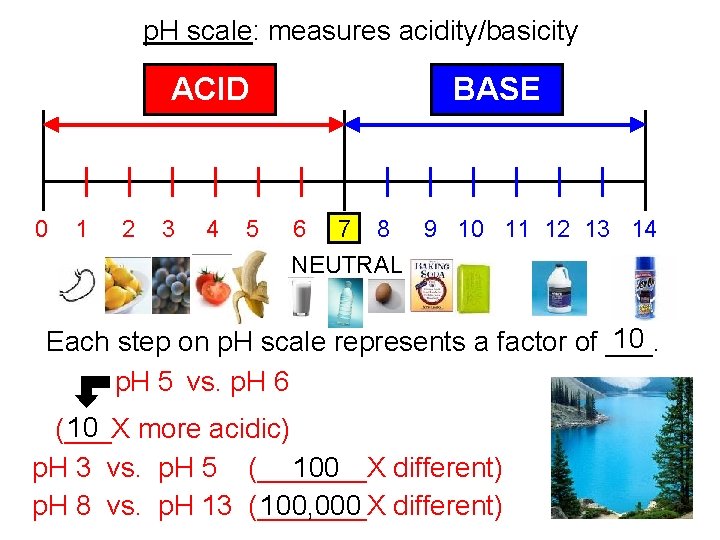
p. H scale: measures acidity/basicity ACID 0 1 2 3 4 5 BASE 6 7 8 9 10 11 12 13 14 NEUTRAL 10 Each step on p. H scale represents a factor of ___. p. H 5 vs. p. H 6 10 more acidic) (___X p. H 3 vs. p. H 5 (_______X different) 100, 000 different) p. H 8 vs. p. H 13 (_______X

Acid Nomenclature binary acids: acids w/H and one other element Binary Acid Nomenclature 1. Write “hydro. ” 2. Write prefix of the other element, followed by “ -ic acid. ” hydrofluoric acid HF hydrochloric acid HCl hydrobromic acid HBr HI hydroiodic acid H 2 S hydrosulfuric acid

Oxyacids oxyacids: acids containing H, O, and one other element Common oxyanions (polyatomic ions that contain oxygen) that combine with H to make oxyacids: Br. O 3 CO 32 Cl. O 3 IO 3 - bromate carbonate chlorate iodate NO 3 PO 43 SO 42 - nitrate phosphate sulfate

Oxyacid Nomenclature Write the name of the polyatmic ion and change the ending to “-ic acid. ” NO HYDRO! HBr. O 3 HCl. O 3 H 2 CO 3 H 2 SO 4 H 3 PO 4 bromic acid chloric acid carbonic acid sulfuric acid phosphoric acid These are the “most common” forms of the oxyacids. Notice that each polyatomic ion ended in “-ate”

Oxyacid Nomenclature Con’t If an oxyacid differs from the “-ate” form by the # of O atoms, the name changes are as follows: one more Ox = per_____ic acid “most common” # of Ox = _____ic acid one less Ox = _____ous acid two fewer Ox = hypo_____ous acid HCl. O 4 HCl. O 3 HCl. O 2 HCl. O phosphorus acid hypobromous acid Perchloric acid Chlorous acid Hypochlorous acid H 3 PO 3 HBr. O persulfuric acid H 2 SO 5

Oxyacid Naming Rules Becomes an acid ending with An ion with a name ending with -ous acid -ite -ic acid -ate Hill, Petrucci, General Chemistry An Integrated Approach 1999, page 60

Naming Acids Practice Formula Name 1 HCl hydrochloric acid __________ 2 HCl. O hypochlorous acid __________ H 2 SO 4 3 ________ sulfuric acid HF 4 ________ hydrofluoric acid 5 H 3 N HIO 4 6 ________ hydronitric acid __________ periodic acid

Common Oxyacid Names The following table lists the most common families of oxy acids. one more HCl. O 4 oxygen atom perchloric acid most “common” HCl. O 3 chloric acid H 2 SO 4 sulfuric acid one less oxygen HCl. O 2 chlorous acid H 2 SO 3 H 3 PO 3 HNO 2 sulfurous acid phosphorous acid nitrous acid two less oxygen’s HCl. O hypochlorous acid H 3 PO 4 phosphoric acid HNO 3 nitric acid H 3 PO 2 hypophosphorous acid

Common Acids Strong Acids (dissociate ~100%) hydrochloric acid: HCl H+ + Cl– -- stomach acid; pickling: cleaning metals w/conc. HCl sulfuric acid: H 2 SO 4 2 H+ + SO 42– -- #1 chemical; (auto) battery acid nitric acid: HNO 3 H+ + NO 3– -- explosives; fertilizer

Common Acids (cont. ) Weak Acids (dissociate very little) acetic acid: CH 3 COOH H+ + CH 3 COO– -- vinegar; naturally made by apples hydrofluoric acid: HF H+ + F– -- used to etch glass citric acid, H 3 C 6 H 5 O 7 -- lemons or limes; sour candy ascorbic acid, H 2 C 6 H 6 O 6 -- vitamin C lactic acid, CH 3 CHOHCOOH -- waste product of muscular exertion
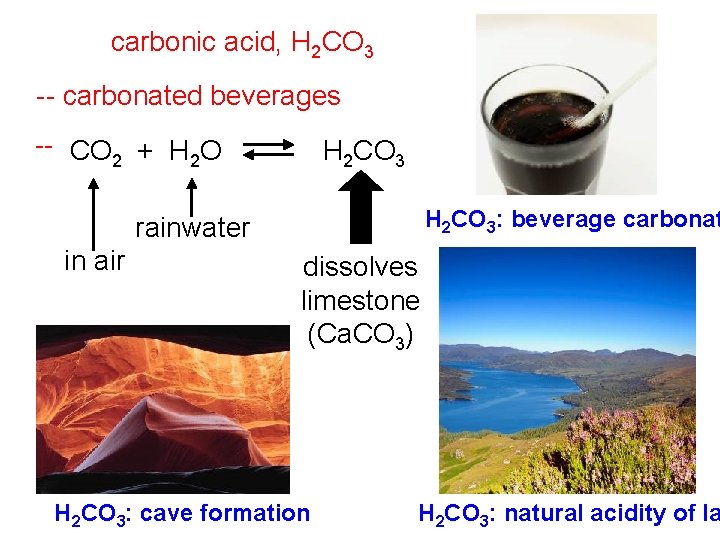
carbonic acid, H 2 CO 3 -- carbonated beverages -- CO 2 + H 2 O H 2 CO 3: beverage carbonat rainwater in air dissolves limestone (Ca. CO 3) H 2 CO 3: cave formation H 2 CO 3: natural acidity of la
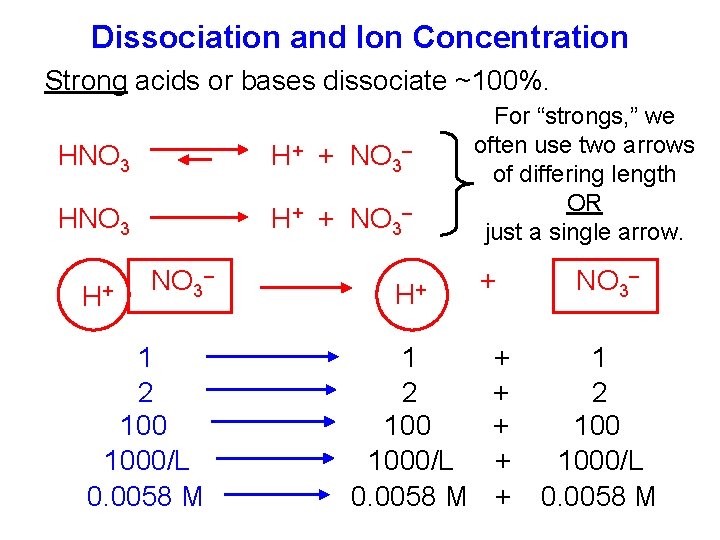
Dissociation and Ion Concentration Strong acids or bases dissociate ~100%. HNO 3 H+ + NO 3– H+ NO 3– 1 2 1000/L 0. 0058 M H+ 1 2 1000/L 0. 0058 M For “strongs, ” we often use two arrows of differing length OR just a single arrow. + + + NO 3– 1 2 1000/L 0. 0058 M
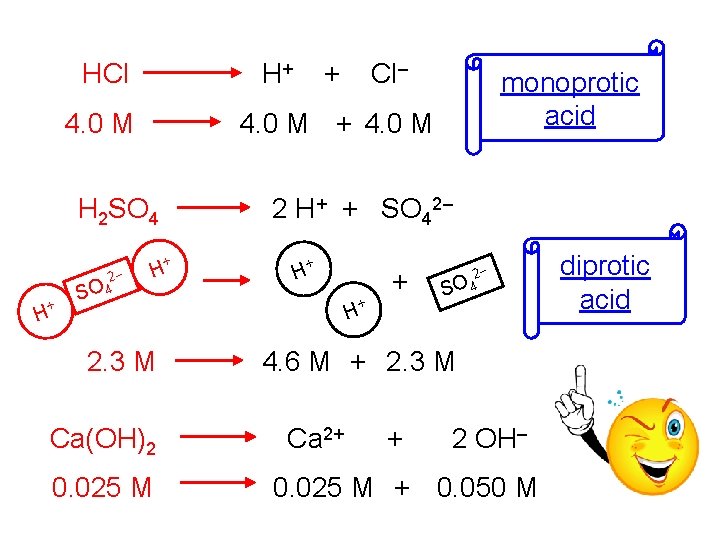
HCl H+ 4. 0 M + H Cl– monoprotic acid 4. 0 M + 4. 0 M H 2 SO 4 2– SO 4 + 2 H+ + SO 42– + H 2. 3 M Ca(OH)2 0. 025 M + H + + 2– SO 4 H 4. 6 M + 2. 3 M Ca 2+ + 2 OH– 0. 025 M + 0. 050 M diprotic acid

p. H Calculations Recall that the hydronium ion (H 3 O+) is the species formed when hydrogen ion (H+) attaches to water (H 2 O). OH– is the hydroxide ion. H+ The number of front wheels is the same as the number of trikes… H 3 O + …so whether we’re counting front wheels (i. e. , H+) or trikes (i. e. , H 3 O+) doesn’t much matter. For this class, in any aqueous sol’n, ( or [ H 3 O+ ] [ OH– ] = 1 x 10– 14 [ H+ ] [ OH– ] = 1 x 10– 14 )
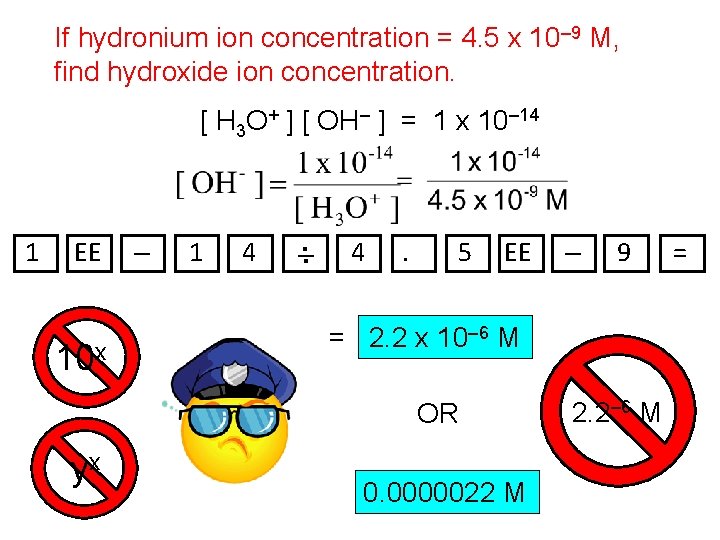
If hydronium ion concentration = 4. 5 x 10– 9 M, find hydroxide ion concentration. [ H 3 O+ ] [ OH– ] = 1 x 10– 14 1 EE 10 x – 1 4 –. . 4 . 5 EE 9 = 2. 2 x 10– 6 M OR yx – 0. 0000022 M 2. 2– 6 M =
![Given: [ H 3 O+ ] [ OH– ] = 1 x 10– 14 Given: [ H 3 O+ ] [ OH– ] = 1 x 10– 14](http://slidetodoc.com/presentation_image_h2/75d8a48dfefc17380a969352422b9b10/image-22.jpg)
Given: [ H 3 O+ ] [ OH– ] = 1 x 10– 14 A. [ OH– ] = 5. 25 x 10– 6 M B. [ OH– ] = 3. 8 x 10– 11 M C. [ H 3 O+ ] = 1. 8 x 10– 3 M D. [ H+ ] = 7. 3 x 10– 12 M Find: [ H+ ] = 1. 90 x 10– 9 M [ H 3 O+ ] = 2. 6 x 10– 4 M [ OH– ] = 5. 6 x 10– 12 M [ H 3 O+ ] = 7. 3 x 10– 12 M Find the p. H of each sol’n above. p. H = –log [ H 3 O+ ] ( or p. H = –log [ H+ ] A. p. H = –log [ H 3 O+ ] = –log [1. 90 x 10– 9 M ] – B. log 3. 59 1 . 9 C. EE 2. 74 – 9 = 8. 72 D. 11. 13 )
![A few last equations… p. OH = –log [ OH– ] p. H + A few last equations… p. OH = –log [ OH– ] p. H +](http://slidetodoc.com/presentation_image_h2/75d8a48dfefc17380a969352422b9b10/image-23.jpg)
A few last equations… p. OH = –log [ OH– ] p. H + p. OH = 14 p. OH [ H 3 O+ ] = 10–p. H ( or [ H+ ] = 10–p. H ) [ OH– ] = 10–p. OH [ H 3 O+ ] = 10–p. H = –log [ H 3 O+ ] [ H 3 O+ ] [ OH– ] = 1 x 10– 14 [ OH– ] = 10–p. OH = –log [ OH– ]
![+ ] ]==10 –p. H [ H 3 O 1+ 10–p. H If p. + ] ]==10 –p. H [ H 3 O 1+ 10–p. H If p.](http://slidetodoc.com/presentation_image_h2/75d8a48dfefc17380a969352422b9b10/image-24.jpg)
+ ] ]==10 –p. H [ H 3 O 1+ 10–p. H If p. H = 4. 87, find [ H 3 O+ ]. p. H = –log [ + ]] H 3 O 1+ p. H + p. OH = 14 [ H 3 O+ ] = 10–p. H = 10– 4. 87 + ]] [ H 3 O 1+ + ] ][ [OH – 14 [ H 3 O 1+ OH– 1–] =] =1 1 x x 1010 – ]] = –p. OH [ OH 1– = 10 10–p. OH = –log [ – ]] OH 1– – ]] [ OH 1– On a graphing calculator… 2 nd log 10 x – 4 . 8 7 = [ H 3 O 1+ ] = 1. 35 x 10– 5 M
![If [ OH– ] = 5. 6 x 10– 11 M, find p. H If [ OH– ] = 5. 6 x 10– 11 M, find p. H](http://slidetodoc.com/presentation_image_h2/75d8a48dfefc17380a969352422b9b10/image-25.jpg)
If [ OH– ] = 5. 6 x 10– 11 M, find p. H [ H 3 O 1+ ] = 10–p. H = –log [ H 3 O 1+ p. H + p. OH = 14 p. OH Find [ H 3 O+ ] = 1. 79 x 10– 4 M [ H 3 O 1+ ] ] [ H 3 O 1+ ] [ OH 1– ] = 1 x 10– 14 [ OH 1– ] = 10–p. OH = –log [ OH 1– ] Find p. OH = 10. 25 Then find p. H… p. H = 3. 75

For the following problems, assume 100% dissociation. Find p. H of a 0. 00057 M nitric acid (HNO 3) sol’n. H+ HNO 3 0. 00057 M (GIVEN) p. H NO 3– 0. 00057 M (affects p. H) (“Who cares? ”) [ H 3 O 1+ ] = 10–p. H [ H 3 O 1+ ] p. H = –log [ H 3 O 1+ ] p. H + p. OH = 14 + p. H = –log [ H 3 O+ ] [ H 3 O 1+ ] [ OH 1– ] = 1 x 10– 14 = –log (0. 00057) = 3. 24 p. OH [ OH 1– ] = 10–p. OH = –log [ OH 1– ]
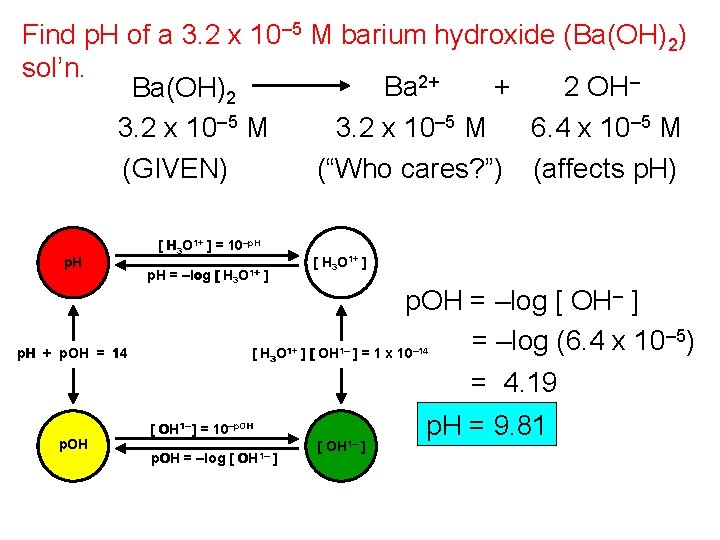
Find p. H of a 3. 2 x 10– 5 M barium hydroxide (Ba(OH)2) sol’n. Ba 2+ + 2 OH– Ba(OH)2 3. 2 x 10– 5 M 6. 4 x 10– 5 M (GIVEN) (“Who cares? ”) (affects p. H) p. H + p. OH = 14 p. OH [ H 3 O 1+ ] = 10–p. H = –log [ H 3 O 1+ ] [ OH 1– p. OH = –log [ OH– ] – 5) = –log (6. 4 x 10 ] = 1 x 10 = 4. 19 – 14 [ OH 1– ] = 10–p. OH = –log [ OH 1– ] p. H = 9. 81
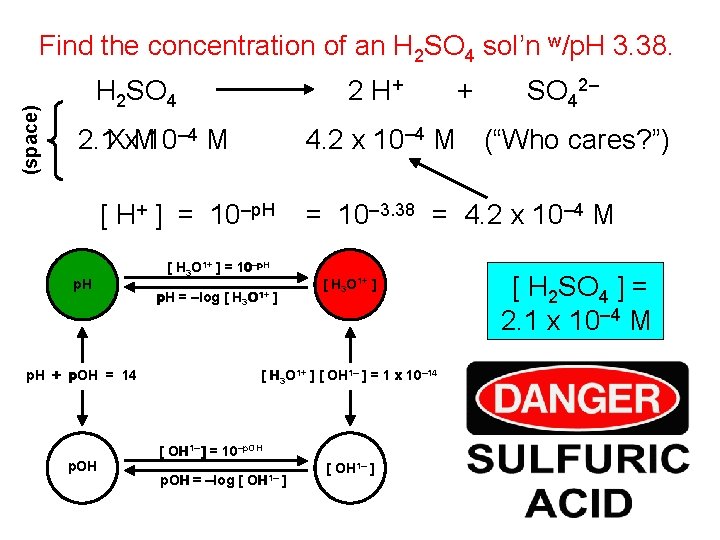
(space) Find the concentration of an H 2 SO 4 sol’n w/p. H 3. 38. 2 H+ H 2 SO 4 [ H+ ] = 10–p. H H 3 O 1+ p. H + p. OH = 14 p. OH = 10– 3. 38 = 4. 2 x 10– 4 M [ H 3 O 1+ ] = 10–p. H = –log [ SO 42– 4. 2 x 10– 4 M (“Who cares? ”) 2. 1 Xx. M 10– 4 M p. H + [ H 3 O 1+ ] ] [ H 3 O 1+ ] [ OH 1– ] = 1 x 10– 14 [ OH 1– ] = 10–p. OH = –log [ OH 1– ] [ H 2 SO 4 ] = 2. 1 x 10– 4 M
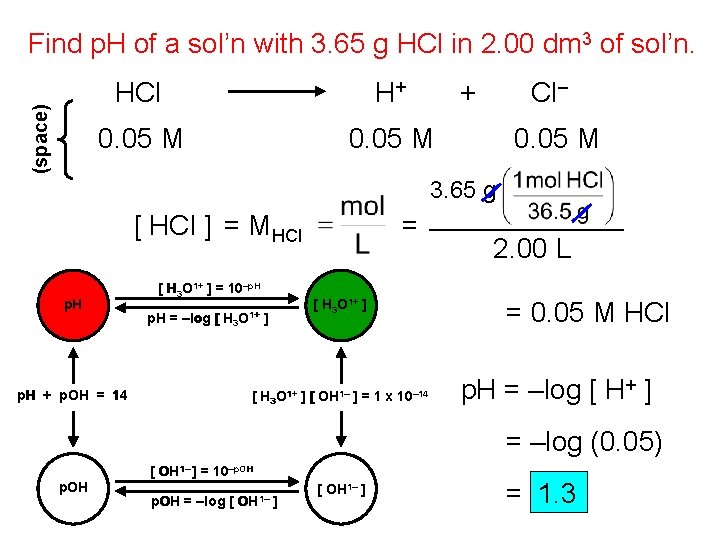
(space) Find p. H of a sol’n with 3. 65 g HCl in 2. 00 dm 3 of sol’n. HCl H+ + 0. 05 M Cl– 0. 05 M 3. 65 g [ HCl ] = MHCl p. H [ H 3 O 1+ ] = 10–p. H = –log [ H 3 O 1+ p. H + p. OH = 14 = [ H 3 O 1+ ] ] [ H 3 O 1+ ] [ OH 1– ] = 1 x 10– 14 2. 00 L = 0. 05 M HCl p. H = –log [ H+ ] = –log (0. 05) p. OH [ OH 1– ] = 10–p. OH = –log [ OH 1– ] = 1. 3
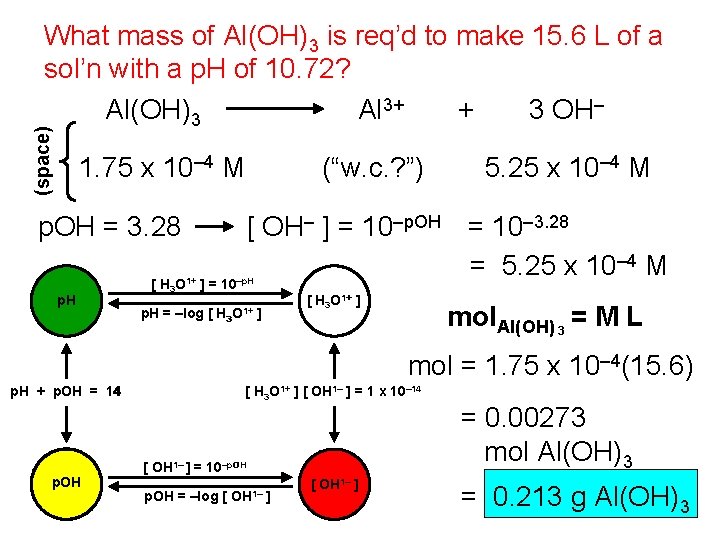
(space) What mass of Al(OH)3 is req’d to make 15. 6 L of a sol’n with a p. H of 10. 72? Al 3+ + 3 OH– Al(OH)3 1. 75 x 10– 4 M p. OH = 3. 28 p. H (“w. c. ? ”) [ OH– ] = 10–p. OH = 10– 3. 28 = 5. 25 x 10– 4 M [ H 3 O 1+ ] = 10–p. H = –log [ 5. 25 x 10– 4 M H 3 O 1+ [ H 3 O 1+ ] ] mol. Al(OH) 3 = M L mol = 1. 75 x 10– 4(15. 6) p. H + p. OH = 14 p. OH [ H 3 O 1+ ] [ OH 1– ] = 1 x 10– 14 [ OH 1– ] = 10–p. OH = –log [ OH 1– ] = 0. 00273 mol Al(OH)3 = 0. 213 g Al(OH)3

Acid-Dissociation Constant, Ka For the generic reaction in sol’n: A + B For strong acids, e. g. , HCl… HCl ~0 H+ + Cl– lots = “BIG. ” Assume 100% dissociation; Ka not applicable for strong acids. C + D
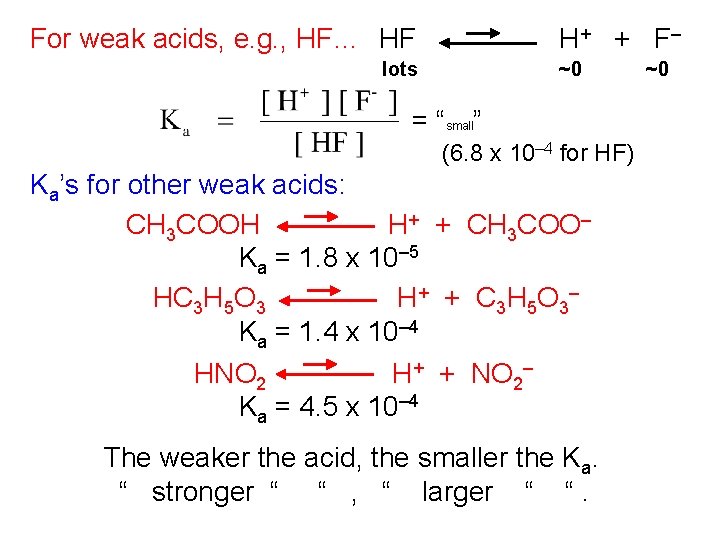
For weak acids, e. g. , HF… HF H + + F– lots ~0 = “small” (6. 8 x 10– 4 for HF) Ka’s for other weak acids: CH 3 COOH H+ + CH 3 COO– Ka = 1. 8 x 10– 5 HC 3 H 5 O 3 H + + C 3 H 5 O 3– Ka = 1. 4 x 10– 4 HNO 2 H+ + NO 2– Ka = 4. 5 x 10– 4 The weaker the acid, the smaller the Ka. “ stronger “ “ , “ larger “ “. ~0

chemicals that change color, depending on the p. H Two examples, out of many: Indicators litmus………………… red in acid, blue in base acid base o pink b phenolphthalein……. . clear in acid, pink in base l e a r

Measuring p. H litmus paper phenolphthalein p. H paper Basically, p. H < 7 or p. H > 7. -- contains a mixture of various indicators -- each type of paper measures a range of p. H -- p. H anywhere from 0 to 14 universal indicator -- is a mixture of several indicators -- p. H 4 to 10 4 5 6 7 8 9 10 R O Y G B I V

Measuring p. H (cont. ) p. H meter -- measures small voltages in solutions -- calibrated to convert voltages into p. H -- precise measurement of p. H
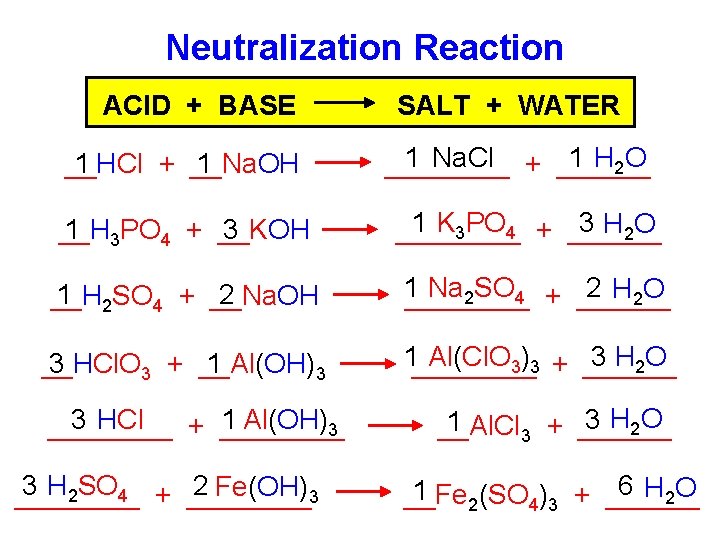
Neutralization Reaction ACID + BASE __HCl + __Na. OH 1 1 __H 1 3 PO 4 + __KOH 3 SALT + WATER 1 Na. Cl + ______ 1 H 2 O ____ 1 K 3 PO 4 + ______ 3 H 2 O ____ 1 2 SO 4 + __Na. OH 2 __H 1 2 H 2 O Na 2 SO 4 + ________ 3 1 __HCl. O 3 + __Al(OH) 3 1____ 3 H 2 O Al(Cl. O 3)3 + ______ 3 HCl 1 Al(OH)3 ____ + ____ 3 H 2 SO 4 + ____ 2 Fe(OH)3 ____ 3 H 2 O 1 __Al. Cl 3 + ______ 6 H 2 O 1 2(SO 4)3 + ______ __Fe
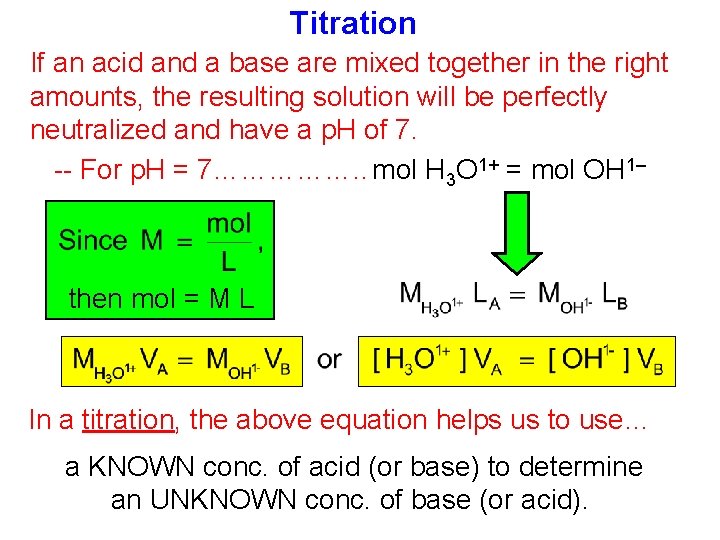
Titration If an acid and a base are mixed together in the right amounts, the resulting solution will be perfectly neutralized and have a p. H of 7. -- For p. H = 7……………. . mol H 3 O 1+ = mol OH 1– then mol = M L In a titration, the above equation helps us to use… a KNOWN conc. of acid (or base) to determine an UNKNOWN conc. of base (or acid).
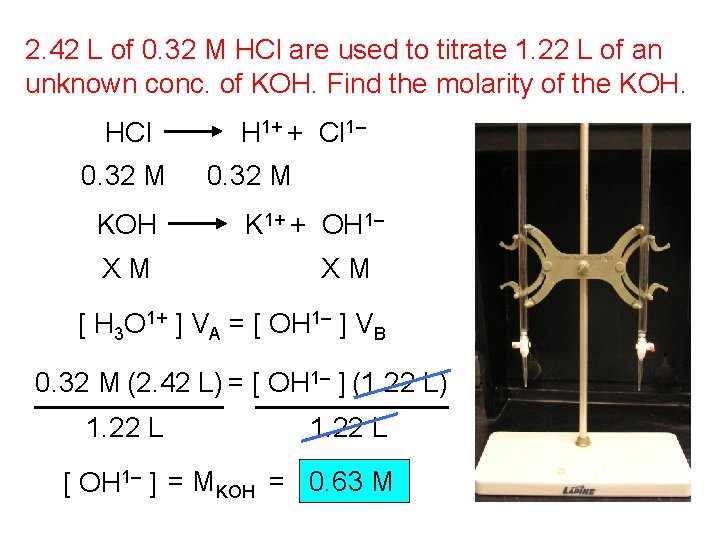
2. 42 L of 0. 32 M HCl are used to titrate 1. 22 L of an unknown conc. of KOH. Find the molarity of the KOH. HCl 0. 32 M KOH XM H 1+ + Cl 1– 0. 32 M K 1+ + OH 1– XM [ H 3 O 1+ ] VA = [ OH 1– ] VB 0. 32 M (2. 42 L) = [ OH 1– ] (1. 22 L) 1. 22 L [ OH 1– ] = MKOH = 0. 63 M

458 m. L of HNO 3 (w/p. H = 2. 87) are neutralized w/661 m. L of Ba(OH). What is the p. H of the base? 2 [ H 3 O 1+ ] VA = [ OH 1– ] VB OK OK If we find this, [ H 3 O 1+ ] = 10–p. H we can find the = 10– 2. 87 base’s p. H. – 3 = 1. 35 x 10 M (1. 35 x 10– 3)(458 m. L) = [ OH 1– ] (661 m. L) [ OH 1– ] = 9. 35 x 10– 4 M p. OH = –log (9. 35 x 10– 4) = 3. 03 p. H = 10. 97
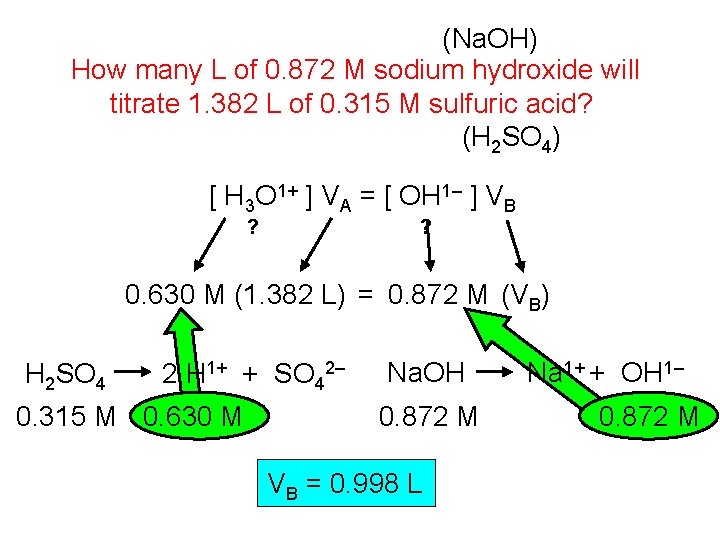
(Na. OH) How many L of 0. 872 M sodium hydroxide will titrate 1. 382 L of 0. 315 M sulfuric acid? (H 2 SO 4) [ H 3 O 1+ ] VA = [ OH 1– ] VB ? ? 0. 630 M (1. 382 L) = 0. 872 M (VB) H 2 SO 4 2 H 1+ + SO 42– 0. 315 M 0. 630 M Na. OH 0. 872 M VB = 0. 998 L Na 1+ + OH 1– 0. 872 M
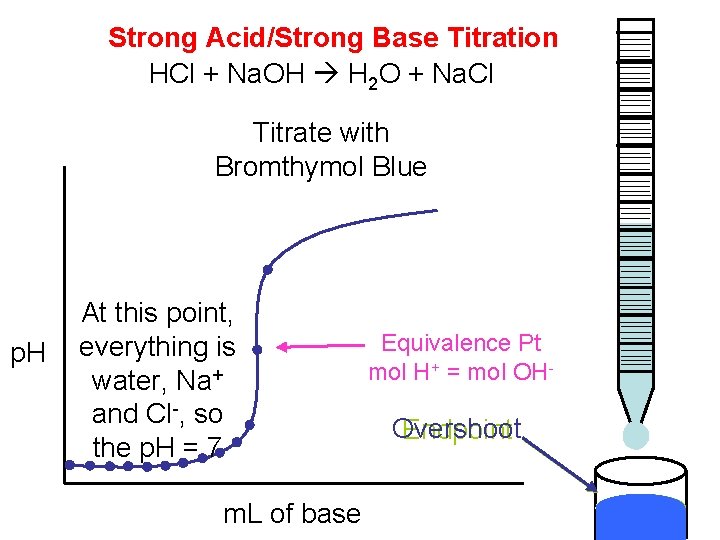
Strong Acid/Strong Base Titration HCl + Na. OH H 2 O + Na. Cl Titrate with Bromthymol Blue p. H At this point, everything is water, Na+ and Cl-, so the p. H = 7 m. L of base Equivalence Pt mol H+ = mol OH- Overshoot Endpoint
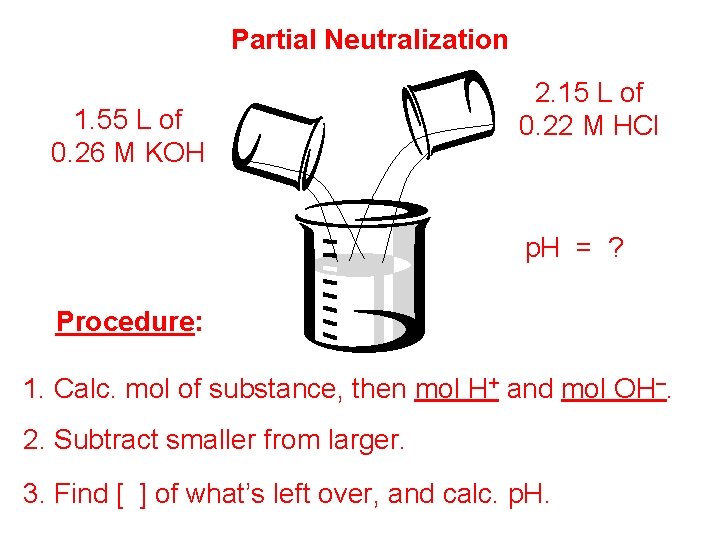
Partial Neutralization 1. 55 L of 0. 26 M KOH 2. 15 L of 0. 22 M HCl p. H = ? Procedure: 1. Calc. mol of substance, then mol H+ and mol OH–. 2. Subtract smaller from larger. 3. Find [ ] of what’s left over, and calc. p. H.
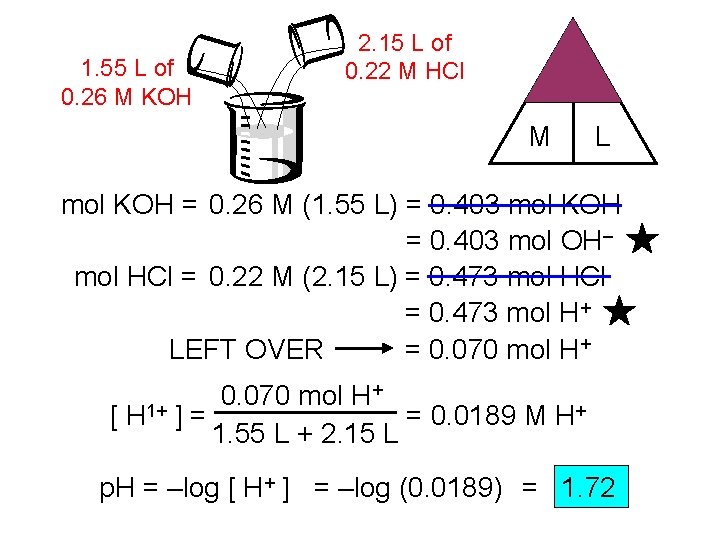
1. 55 L of 0. 26 M KOH 2. 15 L of 0. 22 M HCl mol M L mol KOH = 0. 26 M (1. 55 L) = 0. 403 mol KOH = 0. 403 mol OH– mol HCl = 0. 22 M (2. 15 L) = 0. 473 mol HCl = 0. 473 mol H+ LEFT OVER = 0. 070 mol H+ + 0. 070 mol H [ H 1+ ] = = 0. 0189 M H+ 1. 55 L + 2. 15 L p. H = –log [ H+ ] = –log (0. 0189) = 1. 72
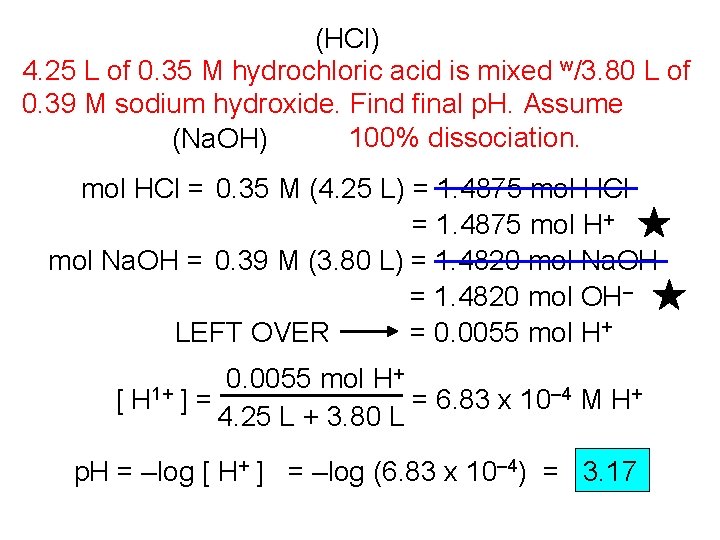
(HCl) 4. 25 L of 0. 35 M hydrochloric acid is mixed w/3. 80 L of 0. 39 M sodium hydroxide. Find final p. H. Assume 100% dissociation. (Na. OH) mol HCl = 0. 35 M (4. 25 L) = 1. 4875 mol HCl = 1. 4875 mol H+ mol Na. OH = 0. 39 M (3. 80 L) = 1. 4820 mol Na. OH = 1. 4820 mol OH– LEFT OVER = 0. 0055 mol H+ + 0. 0055 mol H [ H 1+ ] = = 6. 83 x 10– 4 M H+ 4. 25 L + 3. 80 L p. H = –log [ H+ ] = –log (6. 83 x 10– 4) = 3. 17

(H 2 SO 4) 5. 74 L of 0. 29 M sulfuric acid is mixed w/3. 21 L of 0. 35 M aluminum hydroxide. Find final p. H. (Al(OH)3) Assume 100% dissociation. mol H 2 SO 4 = 0. 29 M (5. 74 L) = 1. 6646 mol H 2 SO 4 = 3. 3292 mol H+ mol Al(OH)3 = 0. 35 M (3. 21 L) = 1. 1235 mol Al(OH)3 = 3. 3705 mol OH– LEFT OVER = 0. 0413 mol OH– – 0. 0413 mol OH [ OH 1– ] = = 0. 00461 M OH– 5. 74 L + 3. 21 L p. OH = –log (0. 00461) = 2. 34 p. H = 11. 66
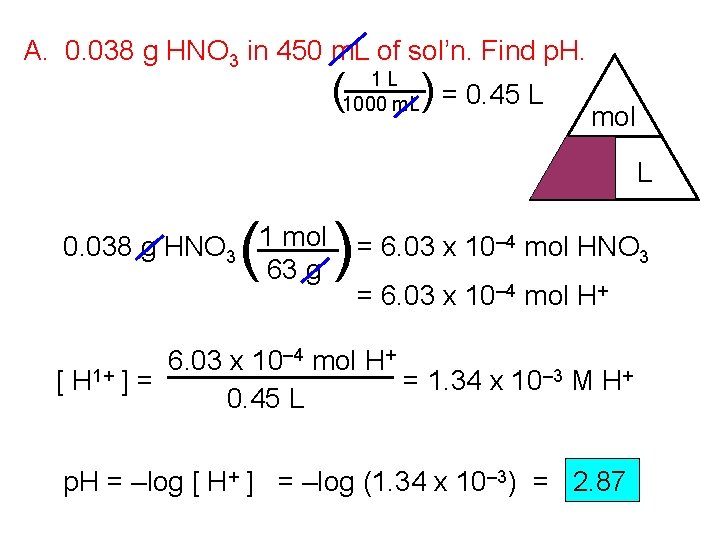
A. 0. 038 g HNO 3 in 450 m. L of sol’n. Find p. H. ( 1 L 1000 m. L ) = 0. 45 L mol M ( 0. 038 g HNO 3 1 mol 63 g ) L = 6. 03 x 10– 4 mol HNO 3 = 6. 03 x 10– 4 mol H+ 6. 03 x 10 [ H 1+ ] = = 1. 34 x 10– 3 M H+ 0. 45 L p. H = –log [ H+ ] = –log (1. 34 x 10– 3) = 2. 87
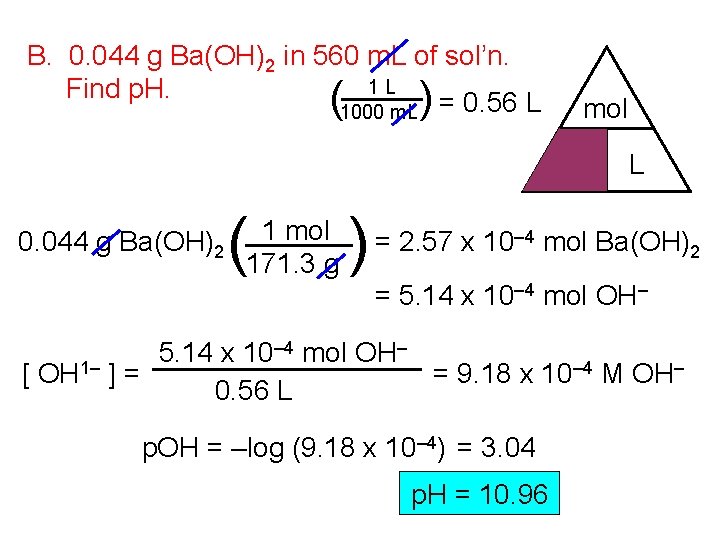
B. 0. 044 g Ba(OH)2 in 560 m. L of sol’n. 1 L Find p. H. (1000 m. L ) = 0. 56 L mol M 0. 044 g Ba(OH)2 ( 1 mol 171. 3 g ) L = 2. 57 x 10– 4 mol Ba(OH)2 = 5. 14 x 10– 4 mol OH– 5. 14 x 10 [ OH 1– ] = = 9. 18 x 10– 4 M OH– 0. 56 L p. OH = –log (9. 18 x 10– 4) = 3. 04 p. H = 10. 96
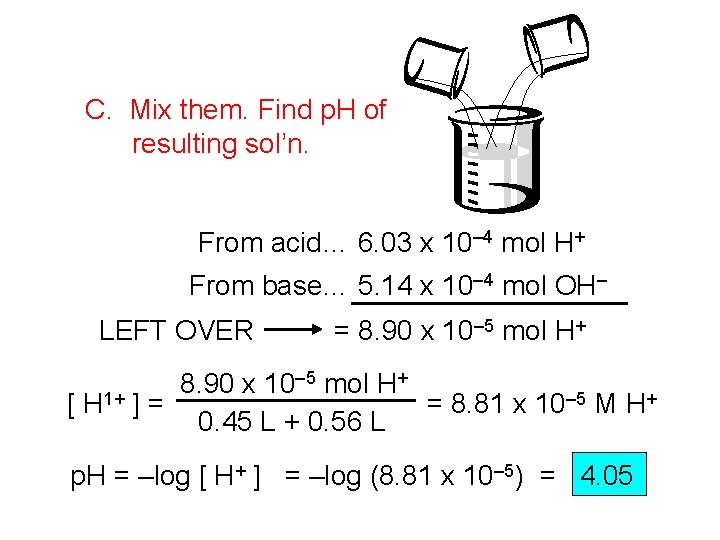
C. Mix them. Find p. H of resulting sol’n. From acid… 6. 03 x 10– 4 mol H+ From base… 5. 14 x 10– 4 mol OH– LEFT OVER = 8. 90 x 10– 5 mol H+ 8. 90 x 10 [ H 1+ ] = = 8. 81 x 10– 5 M H+ 0. 45 L + 0. 56 L p. H = –log [ H+ ] = –log (8. 81 x 10– 5) = 4. 05
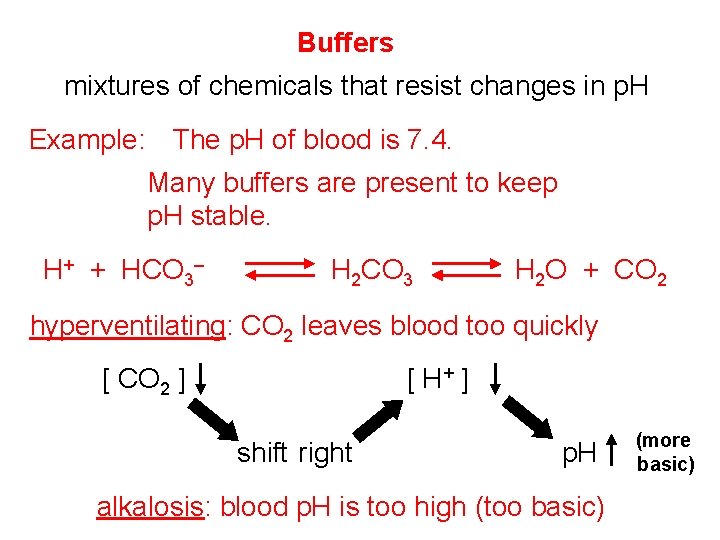
Buffers mixtures of chemicals that resist changes in p. H Example: The p. H of blood is 7. 4. Many buffers are present to keep p. H stable. H+ + HCO 3– H 2 CO 3 H 2 O + CO 2 hyperventilating: CO 2 leaves blood too quickly [ H+ ] [ CO 2 ] shift right p. H alkalosis: blood p. H is too high (too basic) (more basic)

H+ + HCO 3– H 2 CO 3 H 2 O + CO 2 Remedy: Breathe into bag. [ CO 2 ] [ H+ ] shift left acidosis: blood p. H is too low (too acidic) Maintain blood p. H with a healthy diet and regular exercise. (more acidic; closer to normal) p. H

More on buffers: -- a combination of a weak acid and a salt -- together, these substances resist changes in p. H
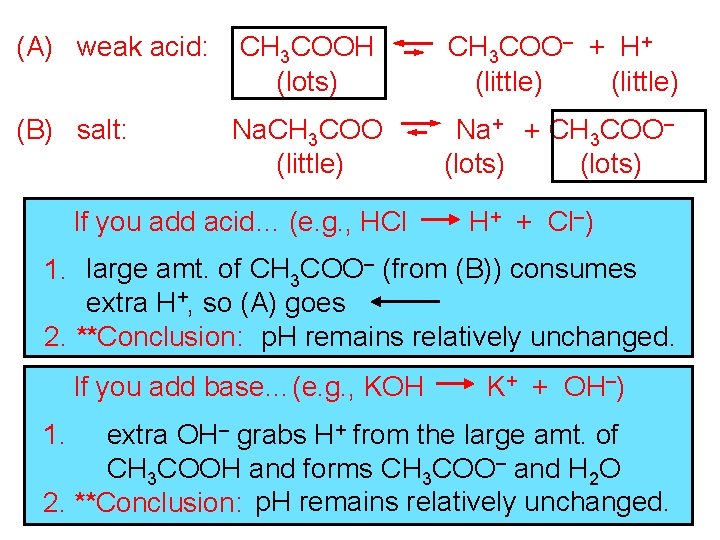
(A) weak acid: CH 3 COOH (lots) CH 3 COO– + H+ (little) (B) salt: Na. CH 3 COO (little) Na+ + CH 3 COO– (lots) If you add acid… (e. g. , HCl H+ + Cl–) 1. large amt. of CH 3 COO– (from (B)) consumes extra H+, so (A) goes 2. **Conclusion: p. H remains relatively unchanged. If you add base…(e. g. , KOH K+ + OH–) extra OH– grabs H+ from the large amt. of CH 3 COOH and forms CH 3 COO– and H 2 O 2. **Conclusion: p. H remains relatively unchanged. 1.
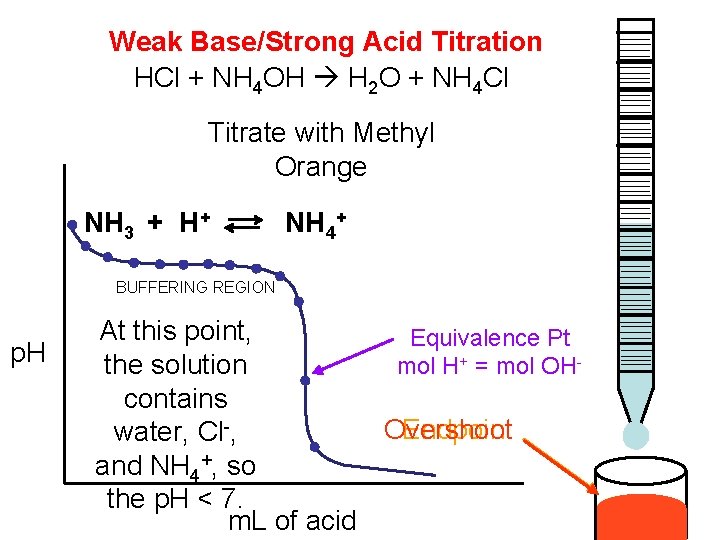
Weak Base/Strong Acid Titration HCl + NH 4 OH H 2 O + NH 4 Cl Titrate with Methyl Orange NH 3 + H+ NH 4+ BUFFERING REGION p. H At this point, Equivalence Pt the solution mol H+ = mol OHcontains Endpoint Overshoot water, Cl-, and NH 4+, so the p. H < 7. m. L of acid
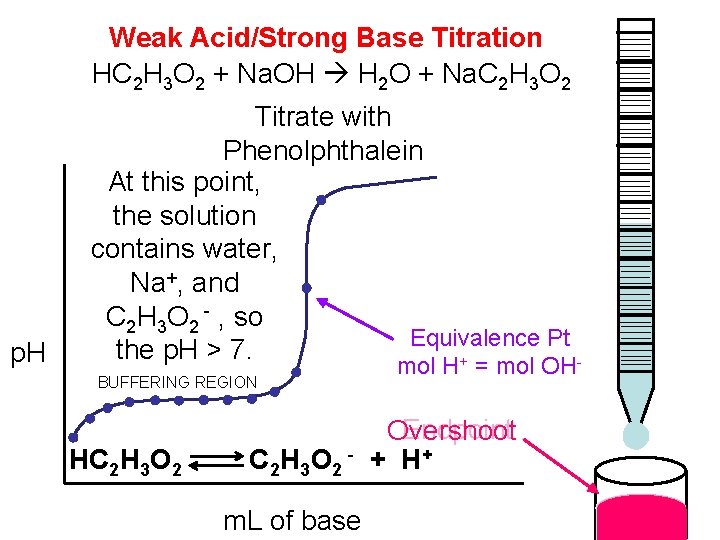
Weak Acid/Strong Base Titration HC 2 H 3 O 2 + Na. OH H 2 O + Na. C 2 H 3 O 2 p. H Titrate with Phenolphthalein At this point, the solution contains water, Na+, and C 2 H 3 O 2 - , so Equivalence Pt the p. H > 7. + BUFFERING REGION HC 2 H 3 O 2 m. L of base mol H = mol OH Endpoint Overshoot + H+
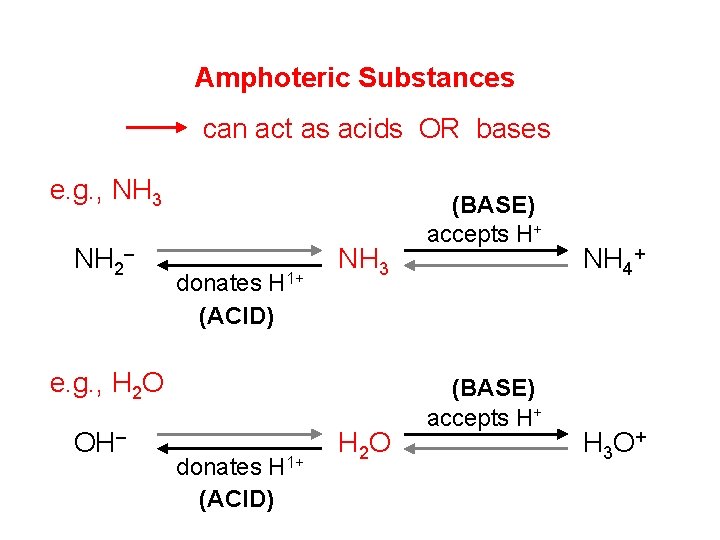
Amphoteric Substances can act as acids OR bases e. g. , NH 3 NH 2– donates H 1+ (ACID) NH 3 e. g. , H 2 O OH– donates H 1+ (ACID) H 2 O (BASE) accepts H+ NH 4+ H 3 O +
- Slides: 55