Unit 11 Acids and Bases Hydronium and Hydroxide

Unit 11 Acids and Bases

Hydronium and Hydroxide • Two important ions in this unit are hydronium and hydroxide • Hydronium is H 3 O+ – – Often abbreviated as H+ The higher the hydronium concentration, the more acidic The higher the hydronium concentration, the lower the p. H The higher the hydronium concentration, the lower the hydroxide concentration • Hydroxide is OH- – The higher the hydroxide concentration, the more basic – The higher the hydroxide concentration, the higher the p. H – The higher the hydroxide concentration, the lower the hydronium concentration

Properties of Acids and Bases Acids • • • p. H of 0 -6 Taste sour Turns litmus paper red Neutralizes a base to form water and a salt Proton donor Electrolyte (conducts electricity in water) Reacts with some metals to produce hydrogen gas Higher concentration of hydronium ions than hydroxide ions [H 3 O+] > 1. 0 x 10 -7 M Bases • p. H of 8 -14 • Taste bitter • Turns litmus paper blue or blue yellow • Neutralizes an Acid to form water and a salt • Proton acceptor • Electrolyte • Feels slippery • Higher concentration of hydroxide ions than hydronium ions • [OH-] > 1. 0 x 10 -7 M

Examples of Acids and Bases Acids Monoprotic- having one acidic hydrogen • HNO 3 • HCl • HC 2 H 3 O 2 Polyprotic- having more than one acidic hydrogen (diprotic or triprotic) • H 2 SO 4 • H 3 PO 4 Bases Monobasic- having one OH- group • Li. OH • Na. OH • KOH • NH 4 OH Polybasic- having more than one OH- group • Mg(OH)2 • Ca(OH)2 • Al(OH)3

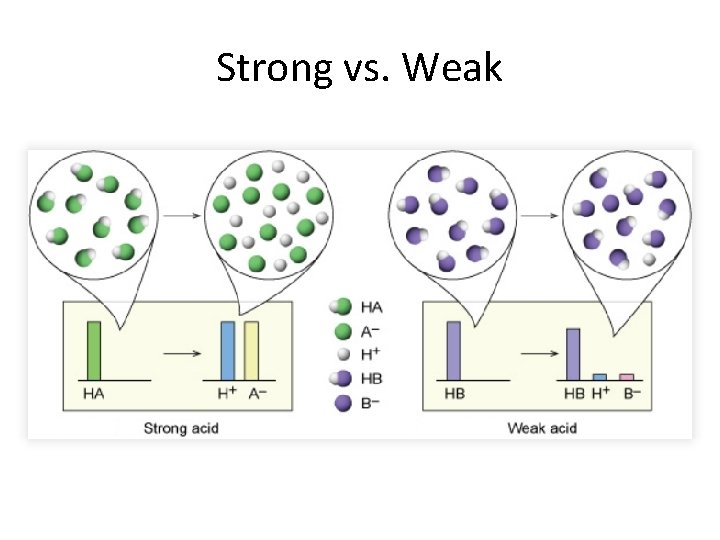
Strong v. Weak Facts • To dissociate means to separate into ions when dissolved in water • Strong= completely dissociates in water (100%) • Weak= does not completely dissociate in water (< 5%) • A strong acid/base does not become a weak acid just because it is diluted. – In other words, concentrated HCl and diluted HCl are both strong acids because both completely dissociate in water. • Strong acids/bases are good conductors of electricity; whereas weak acids/bases are poor conductors of electricity.
Strong vs. Weak

Strong v. Weak Acid Strong Acid • 100% dissociated • All of the acid separates into ions in water • Strong electrolyte • Example: HCl, HI, HBr, HNO 3, HCl. O 3 , HCl. O 4, H 2 SO 4 *You have to know ALL 7 strong acids Weak Acid • Partially dissociated (usually less than 5%) • Some molecules become ions in water but most of the acid molecules stay as molecules • Weak electrolyte • Examples (if it is not one of the 7 strong acids, then it is weak): Acetic acid, hydrofluoric acid, nitrous acid

Strong v. Weak Base Strong Base Weak Base • 100% dissociated • All of the base separates into ions in water • Strong electrolyte • Example: Li. OH, Na. OH, KOH, Rb. OH, Cs. OH, Ca(OH)2 , Ba(OH)2 , and Sr(OH)2 • Partially dissociated (usually less than 5%) • Some molecules become ions in water but most of the base molecules stay as molecules • Weak electrolytes • Examples (if it’s not strong it is weak): • Ammonia, Ammonium hydroxide, Silver hydroxide *you have to know ALL 8 of the strong bases

Electrolytes • Salts are ionic compounds that are neither acids nor bases • Electrolytes are ions that conduct electricity when they are dissolved in water • Strong acids and bases are strong electrolytes – This is because 100% of the substance dissociates • Soluble salts are also strong electrolytes – This is because 100% of the substance dissociates • Weak acids and bases are weak electrolytes – This is because a very small amount of the substance dissociates • Insoluble salts are weak electrolytes – This is because a very small amount, if any at all, of the substance dissociates • Solubility rules can be used to determine whether a salt is soluble or insoluble

Types of Equations • In a molecular equation (sometimes called a balanced equation), all substances are represented as neutral compounds using the molecular formula. – Example: HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l) • In a complete ionic equation (sometimes called an ionic equation), all substances that dissociate in water are represented by the formula of their ions and all substances that do not dissociate in water are represented using the molecular formula. – Example: H+ (aq) + Cl- (aq) + Na+ (aq) + OH- (aq) Na+ (aq) + Cl- (aq) + H 2 O (l) • In a net ionic equation only the species involved in the chemical reaction are shown (spectator ions are not shown) – Example: H+ + OH- (aq) H 2 O (l) – Spectator ions: Cl- (aq) and Na+ (aq)

Writing Ionic and Net Ionic Equations • STEP 1: write the correct BALANCED molecular equation INCLUDING states of matter – Hint: use solubility rules to determine states of matter • STEP 2: write the complete ionic equation – Any substance that is a soluble salt, a strong acid, or a strong base gets fully dissociated – Any substance that is a solid, liquid, or a gas, is a weak acid, or a weak base does NOT get dissociated – Polyatomic ions stay together as a group they do not further dissociate – Do not forget to include the charge of each ion – Do not forget to balance • STEP 3: Cross off any spectator ions in order to write the net ionic equation – Spectator ions are ions that do NOT take place in the chemical reaction – Spectator ions appear as ions in both the reactants and products of a complete ionic equation • You only need to include states of matter in the molecular equation

Net Ionic Equation • Example #1: Write the net ionic equation for the reaction between nitric acid and lithium hydroxide and identify the spectator ion(s). • Molecular Equation: HNO 3 (aq) + Li. OH (aq) H 2 O (l) + Li. NO 3 (aq) • Complete Ionic Equation: H+ + NO 3 - + Li+ + OH- H 2 O + Li+ + NO 3 - • Net Ionic Equation: H+ + OH- H 2 O • Spectator ion(s): Li+ and NO 3 -

Net Ionic Equation • Example #2: Write the net ionic equation for the reaction between hydrofluoric acid and calcium hydroxide and identify the spectator ion(s). • Molecular Equation: 2 HF (aq) + Ca(OH)2 (aq) Ca. F 2 (aq) + H 2 O (l) • Complete Ionic Equation: 2 HF (aq) + Ca 2+ (aq) + 2 OH- (aq) Ca 2+ (aq) + 2 F- (aq) + H 2 O (l) • Net Ionic Equation: 2 HF (aq) + 2 OH- (aq) 2 F- (aq) + H 2 O (l) • Spectator ion(s): Ca 2+ (aq)

Net Ionic Equations • The net ionic equation between any strong acid and strong base will always be: H+ + OH- H 2 O

Acid/Base Theories Arrhenius Theory • Acids produce H+ ions in H 2 O whereas a base produces OH- ions in H 2 O. • Salts produce neither H+ ions nor OH- ions in H 2 O • Good for describing strong acids and strong bases Bronsted-Lowry Theory • H+ is a proton • Acid is a proton donor (H+) where as a base is a proton acceptor • When an acid or a base reacts with water, water can act as an acid or base.

Conjugate Pairs • Using Bronsted-Lowry’s definition of acids and bases • Conjugate Acid/Base pairs: A pair of species that are related to each other by 1 H+ • The acid becomes a CB and the base becomes a CA • The acid and base are on the reactants side of the equation • The conjugates are on the products of the side of the equation

Conjugate Pairs • When an acid reacts with water it produces H 3 O+ (hydronium ion, considered the conjugate acid) and a conjugate base (everything left over once the H+ ion is removed) • HCl + H 2 O H 3 O+ Cl • Acid + Base Conjugate acid(CA) + conjugate base(CB) • The CA and the CB can reform to make the parent Acid and Base therefore the reaction is reversible which is indicated by double arrows. • HA(aq) + H 2 O(l) H 3 O+(aq) + A- (aq) • Acid + Base CA + CB

Conjugate Acid/Base Pairs HA + H 2 O H 3 O+ + A- • The conjugate base of a strong acid is a weaker base than water • The conjugate base of a weak acid is a stronger base than water • The conjugate acid of a weak base is a stronger acid than water • The conjugate acid of a strong base is a weaker acid than water

Conjugate Acid/Base Pairs H 2 SO 4 + H 2 O → HSO 4 - + H 3 O+ A B CA H 2 SO 4 is an acid b/c it gives up a proton HSO 4 - is the CB b/c it can accept another proton • H 2 O is the base b/c it accepted the proton • H 3 O+ is the CA b/c it can donate another proton. • •

More Practice Write the formula for the CB of these original acids • • • HNO 3 H 2 O H 3 O + H 2 SO 4 HCO 3 - • • • NO 3 OHH 2 O HSO 4 CO 32 -

More Practice Write the formula for the CA of these original bases: • • • OHH 2 O HCO 3 SO 42 Cl. O 4 - • • • H 2 O H 3 O + H 2 CO 3 HSO 4 HCl. O 4

Conjugate Acids and Bases • Example problems: • Determine the Acid, Base, CA, and CB for the following reactions. HBr + H 2 O H 3 O+ + Br. Acid + Base CA + CB NH 3 + H 2 O NH 4+ + OH NH + H O NH + + OH 3 2 4 Base + Acid CA + CB

Water • Water is the most common amphoteric substance (a substance that can act as both an acid or a base) • Auto- Ionization of water (aka water reacts with itself): H 2 O + H 2 O H 3 O+ + OHWater is both accepting a proton and donating a proton • Water will act as a base when reacted with an acid • Water will act as an acid when reacted with a base • Water has a p. H of 7 and is considered neutral • Hydronium ion concentration determines p. H of a substance

Water • Water is neutral because it has equal concentrations of hydronium and hydroxide • [H 3 O+] = [OH-]= 1. 0 x 10 -7 M • Because of this concentration of hydronium ions the p. H of water is 7 • If the concentration of hydronium ions increase the concentration of hydroxide ions decreases • Substances who have higher concentration of hydronium than hydroxide have a p. H < 7 • [H 3 O+] > [OH-] OR [H 3 O+] > 1. 0 x 10 -7 M then p. H < 7 • These solutions are considered acidic • If the concentration of hydroxide ions increases the concentration of hydronium ions decreases • Substances who have higher concentration of hydroxide than hydronium have a p. H > 7 • [OH-] > [H 3 O+] OR [OH-] > 1. 0 x 10 -7 M then p. H > 7 • The solutions are considered basic
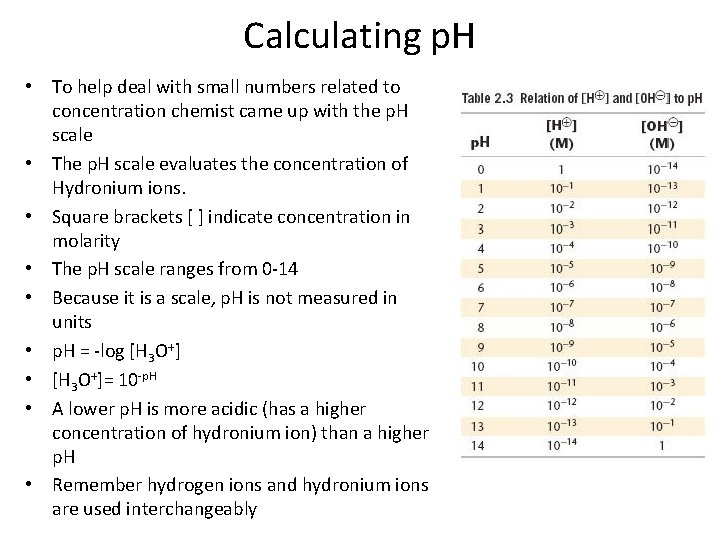
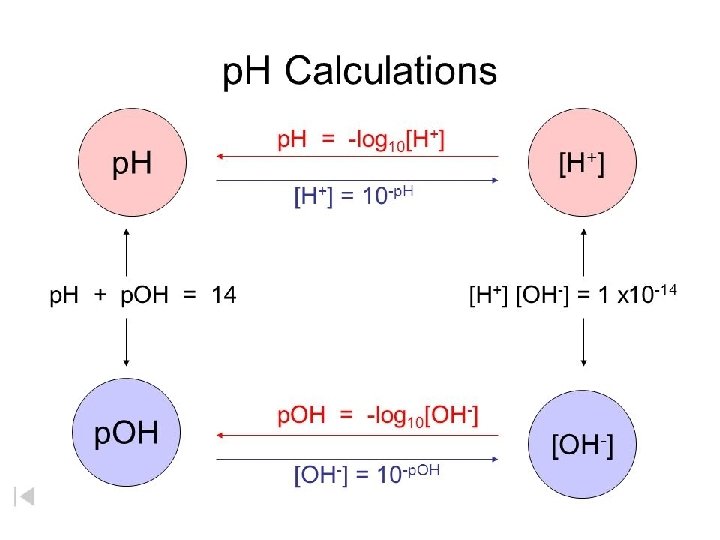
Calculating p. H • To help deal with small numbers related to concentration chemist came up with the p. H scale • The p. H scale evaluates the concentration of Hydronium ions. • Square brackets [ ] indicate concentration in molarity • The p. H scale ranges from 0 -14 • Because it is a scale, p. H is not measured in units • p. H = -log [H 3 O+] • [H 3 O+]= 10 -p. H • A lower p. H is more acidic (has a higher concentration of hydronium ion) than a higher p. H • Remember hydrogen ions and hydronium ions are used interchangeably

Calculating p. H • Find the p. H of a 0. 0025 M HCl solution. -The HCl is a strong acid and is 100% dissociated in water. -The hydronium ion concentration is 0. 0025 Thus: p. H = - log (0. 0025) = - ( - 2. 6) = 2. 6 • What is the concentration of hydrogen ions in a solution that has a p. H of 4. 3? • [H+]= 10 -p. H • [H+]= 10 -4. 3 • [H+]= 5. 0 x 10 -5 M

Calculating p. H • Find the p. H of a 0. 0025 M H 2 SO 4 solution. p. H = - log (2 x 0. 0025) = 2. 30
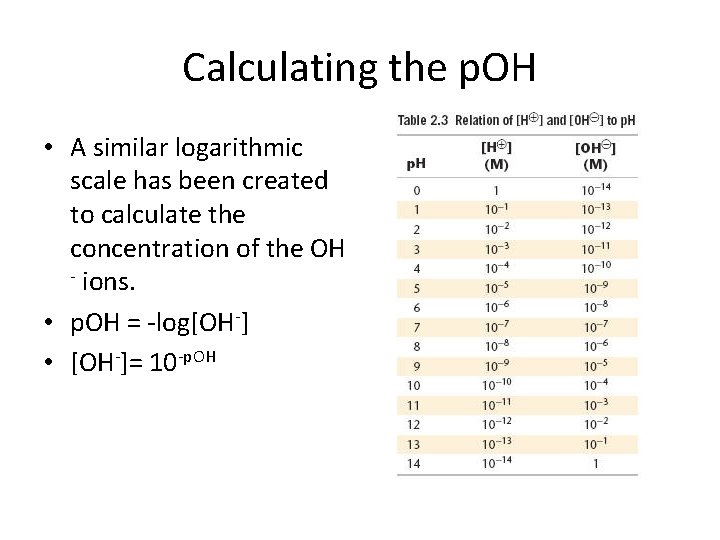
Calculating the p. OH • A similar logarithmic scale has been created to calculate the concentration of the OH - ions. • p. OH = -log[OH-] • [OH-]= 10 -p. OH

Calculating p. OH • What is the p. OH of a solution that has a hydroxide ion concentration of 4. 82 x 10 -5 M? • p. OH = -log [OH-] • p. OH = - log [4. 82 x 10 -5] = - ( - 4. 32) = 4. 32 • What is the concentration of hydroxide ions in a solution that has a p. OH of 12. 2? • [OH-]= 10 -p. OH • [OH-]= 10 -12. 2 • [OH-]= 6. 3 x 10 -13 M

Another way to calculate p. H and p. OH • p. H + p. OH = 14. 00 • A solution has a p. OH of 11. 76. What is the p. H of this solution? p. H + 11. 76 = 14. 00 p. H = 2. 24 • What is the [H+] when the p. OH is 5? • p. H + 5 = 14. 00 • p. H = 9 • p. H = -log [H+] • 9 = -log [H+] • [H+] = 1. 0 x 10 -9 M
© 2009, Prentice-Hall, Inc.
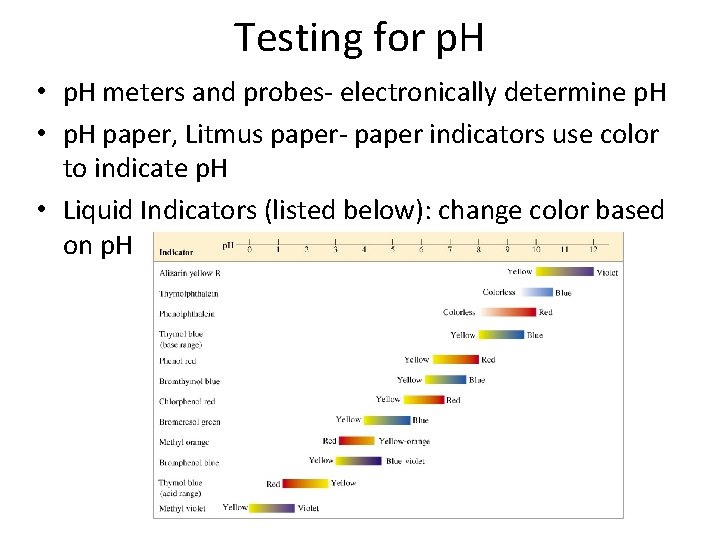
Testing for p. H • p. H meters and probes- electronically determine p. H • p. H paper, Litmus paper- paper indicators use color to indicate p. H • Liquid Indicators (listed below): change color based on p. H

Neutralization Reactions • When an acid reacts with a base a neutralization occurs • In a neutralization reaction water and a salt are always produced • A neutralization reaction does NOT always result in a p. H of 7 – It results in a more neutral substance but not always completely neutral

Neutralization Reaction • When a strong acid reacts with a strong base it will produce a neutral salt and water. – Example: Na. OH + HCl HOH + Na. Cl – The net ionic equation of a reaction between a strong acid and a strong base is always: H+ + OH- H 2 O • When a strong acid reacts with a weak base it produces an acidic salt and water • When a strong base reacts with a weak acid it produces a basic salt and water • When a weak acid reacts with a weak base, it can produce a neutral, acidic, or basic salt

Practice • Write the neutralization reactions that occur between the following acid and base and balance the equation. Determine if the salt will be acidic, basic, neutral, or undetermined. Include states of matter. • Hydroiodic acid and silver hydroxide HI (aq) + Ag. OH (s) H 2 O (l) + Ag. I (s) Ag. I is an acidic salt • Phosphoric acid and potassium hydroxide H 3 PO 4 (aq) + 3 KOH (aq) 3 H 2 O (l) + K 3 PO 4 (aq) K 3 PO 4 is a basic salt

Parent Acid/Base • If you know the salt involved you should be able to determine which acid and base it was formed from – Hint: write the reaction backwards and write water as HOH • Example: Name the parent acid and base that would produce these salts: – Potassium chloride • Potassium hydroxide and hydrochloric acid – Magnesium carbonate • Magnesium hydroxide and carbonic acid

Titration Setup • Titration- an experimental way to determine the concentration of an acid or a base • Buret- a graduated glass tube with a tap at one end, for delivering known volumes of a liquid, especially in titrations. • Titrant- standard solution of known concentration (usally strong) • Stopcock- determines flow of liquid • Analyte- unknown solution

Titrations • Titration- an experimental way to determine the concentration of an acid or a base • A neutralization reaction that reacts an unknown concentration of acid or base with a known concentration of base or acid – One of the substances involved in the titration must be a strong acid or base – Titrations are never done with a weak acid AND a weak base • Equivalence point- point at which there are equal moles of H+ and OH- – Shown on a titration curve by a drastic change in p. H – Can also be shown by a change in color of an indicator – When performing a titration, it is best to use an indicator that changes colors at the p. H of the equivalence point • The p. H of the analyte is measured throughout the titration • Data is then used to create a graph called a titration curve • The titration curve along with the data is then used to calculate the concentration of the unknown acid/base

Titrations
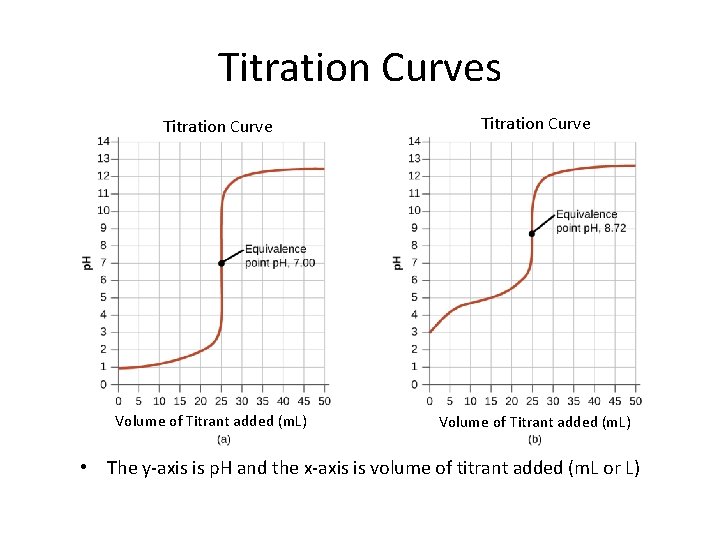
Titration Curves Titration Curve Volume of Titrant added (m. L) • The y-axis is p. H and the x-axis is volume of titrant added (m. L or L)

Equivalence Point • The equivalence point of a titration involving a strong acid and a strong base occurs at p. H = 7 – This all of the acid and base have reacted and a neutral salt is produced • The equivalence point of a titration involving a strong acid and a weak base occurs at p. H < 7 – This all of the acid and base have reacted an acidic salt is produced • The equivalence point of a titration involving a weak acid and a strong base occurs at p. H > 7 – This all of the acid and base have reacted and a basic salt is produced
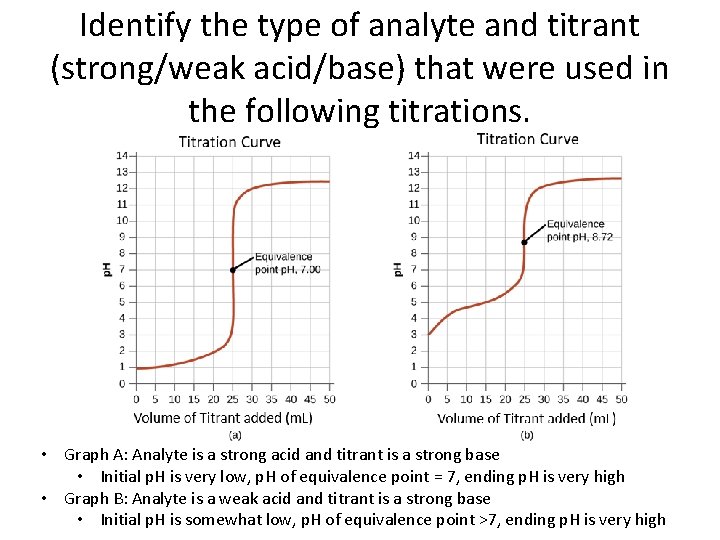
Identify the type of analyte and titrant (strong/weak acid/base) that were used in the following titrations. • Graph A: Analyte is a strong acid and titrant is a strong base • Initial p. H is very low, p. H of equivalence point = 7, ending p. H is very high • Graph B: Analyte is a weak acid and titrant is a strong base • Initial p. H is somewhat low, p. H of equivalence point >7, ending p. H is very high

Titrations • @ equivalence point: Moles of H+ = moles of OHMA VA n. A = MB VB n. B • M: Molarity • V: Volume • N: # of H+ ions in the acid or OH- ions in the base • M and V are used to determine sig figs

Titration Problem #1 • If 50. 0 m. L of an unknown concentration of HCl is titrated with of 100. 0 m. L of 1. 0 M Ca(OH)2, what is the concentration of the acid? MVn = MVn M (50. 0 m. L) (1) = (1. 0 M) (100. 0 m. L) (2) M= 4. 0 M © 2009, Prentice-Hall, Inc.

Titration Problem #2 • What volume of 0. 65 M sodium hydroxide is needed to fully neutralizes 25. 0 m. L of 0. 70 M phosphoric acid during a titration? MVn = MVn (0. 70 M)(25. 0 m. L) (3) = (0. 65 M) V (1) V = 81 m. L

Titration #3 • What is the p. H of a sulfuric acid solution if it requires 89. 0 m. L of a 0. 455 M KOH solution to neutralize 120. 0 m. L of the acid?

Buffers Solutions • Buffer Solution- A solution that resists change in p. H when an acid or a base is added to the solution • A buffer consists of: – a weak acid and a salt of its conjugate base in equal amounts – OR a weak base and a salt of its conjugate acid in equal amounts • The buffer solution is continuously converting between acid and its conjugate base to keep the p. H of the solution within a specific range • Used to maintain a stable p. H • Example: Buffer system in blood, aspirin buffer coating

Buffer Examples • Which of the following pairs would make a good buffer and why? A. B. C. D. HCl and Na. Cl HCl and Na. OH HF and Na. F HF and Na. OH C – because it is a weak acid and a salt of its conjugate base

Unit 11 Formulas •
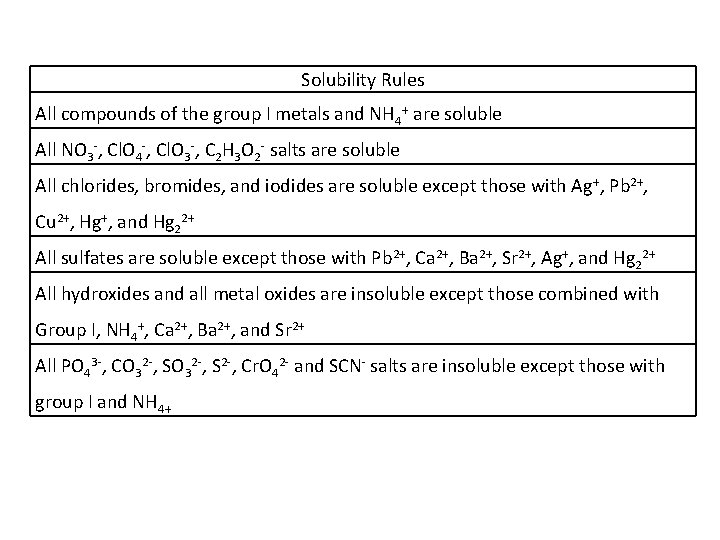
Solubility Rules All compounds of the group I metals and NH 4+ are soluble All NO 3 -, Cl. O 4 -, Cl. O 3 -, C 2 H 3 O 2 - salts are soluble All chlorides, bromides, and iodides are soluble except those with Ag+, Pb 2+, Cu 2+, Hg+, and Hg 22+ All sulfates are soluble except those with Pb 2+, Ca 2+, Ba 2+, Sr 2+, Ag+, and Hg 22+ All hydroxides and all metal oxides are insoluble except those combined with Group I, NH 4+, Ca 2+, Ba 2+, and Sr 2+ All PO 43 -, CO 32 -, S 2 -, Cr. O 42 - and SCN- salts are insoluble except those with group I and NH 4+
- Slides: 50