Unit 10 SUMMARY Buffers AcidBase Titrations Buffer weak

Unit 10 SUMMARY: Buffers & Acid-Base Titrations Buffer: • weak conjugate acid-base pair. • resist p. H changes (reacts added acid/base) • buffer capacity: more moles of buffer can react more acid/base before significant p. H changes. • p. H range: range of p. H values buffer works. Optimum Capacity and Range: - weak acid with a p. Ka near desired p. H - [HA] = [A–] (so p. Ka = p. H)
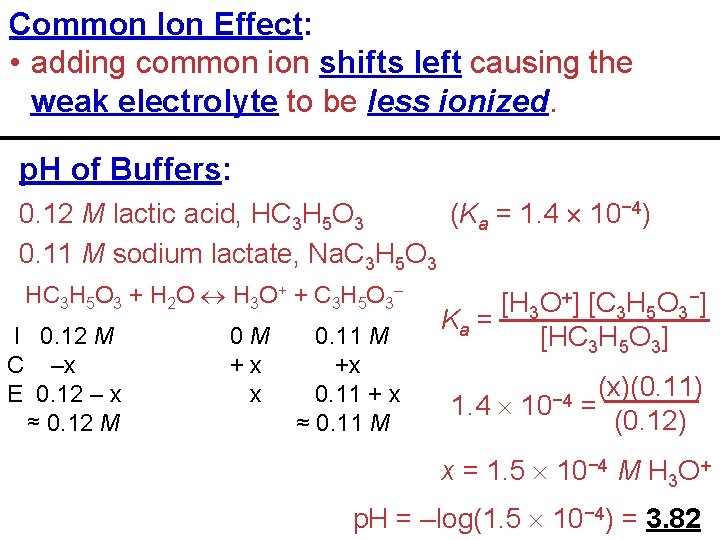
Common Ion Effect: • adding common ion shifts left causing the weak electrolyte to be less ionized. p. H of Buffers: 0. 12 M lactic acid, HC 3 H 5 O 3 (Ka = 1. 4 10− 4) 0. 11 M sodium lactate, Na. C 3 H 5 O 3 HC 3 H 5 O 3 + H 2 O H 3 O+ + C 3 H 5 O 3– I 0. 12 M C –x E 0. 12 – x ≈ 0. 12 M 0 M +x x 0. 11 M +x 0. 11 + x ≈ 0. 11 M [H 3 O+] [C 3 H 5 O 3−] Ka = [HC 3 H 5 O 3] 1. 4 10− 4 (x)(0. 11) = (0. 12) x = 1. 5 10− 4 M H 3 O+ p. H = –log(1. 5 10− 4) = 3. 82

Titration The analytical technique used to calculate the moles in an unknown soln. mass % molarity (g /gtotal) (mol/L) molar mass buret (g/mol) titrant known vol. (V) end point: known conc. (M) indicator color change equivalence point: equal stoich. amounts (mols) react completely analyte known vol. (V) unknown conc. (M)(or moles)

SA with SB At Veq, p. H = 7 At Veq… moles added = moles reacted …the solution contains only water & salt. (irrelevant conjugate)

WA with SB • At Veq, p. H > 7 (only water & conj. base) A– + H 2 O ↔ HA + OH–

WB with SA • At Veq, p. H < 7 (only water & conj. acid) HA + H 2 O ↔ H 3 O+ + A– Indicators: weak acids with diff color conjugates • choose one that changes color (has p. Ka) near p. H of equivalence point (Veq) of titration.

Verbally describe the visual curve: WA with SB Weak acids: • moderately low initial p. H • gradual p. H rise (not flat then jump) • subtle p. H change at equiv. point (less steep) Weak bases: (same but “drop” not “rise”)

Titration Calculations • Calculate unknown moles of the ANALYTE. (then calculate: M OR molar mass OR mass %) ØStoich: (L X mol Y) MX • Calculate unknown VOLUME of TITRANT (m. L) added to reach equivalence (all reacted). ØStoich: (L X mol Y L Y) MX MY • Calculate unknown p. H at any point in the titration (especially at equivalence). ØStoich: What’s in the flask? Find excess H+ , OH–
- Slides: 8