Unit 10 DETERMINING FORMULAS AND NOMENCLATURE CHEMICAL FORMULAS



















- Slides: 20

Unit 10 DETERMINING FORMULAS AND NOMENCLATURE

CHEMICAL FORMULAS n Clearest and simplest method of designating compounds Shows number of atoms using subscripts n Shows the kind of elements in the compound n

EXAMPLE OF CHEMICAL FORMULA VITAMIN A C 20 H 30 O

SALTS n COMPOUNDS THAT BEGIN WITH A METAL or NH 4

SALTS n BINARY SALTS – Contain only two elements: a metal and a nonmetal –To name a binary salt: l. Name the metal l. Drop ending of nonmetal and add "ide"

PRACTICE NAMING BINARY SALTS 1. 2. 3. 4. 5. Na. Cl Ca. F 2 K 2 O lithium nitride aluminum iodide

TERNARY SALTS n Contain three or more elements metal and polyatomic ion n To name a ternary salt: –Name metal –Name polyatomic ion

PRACTICING WRITING TERNARY SALTS 1. 2. 3. 4. 5. K 3 PO 4 Ca. CO 3 Al(NO 3)3 barium hydroxide cesium chromate

Multiple Ox# SALTS n n The metals may have more than one oxidation number To name the salt: –Name the metal –Put Roman numeral after the metal to indicate oxidation state being used –Same ending as other salts

PRACTICING SPECIAL SALTS 1. 2. 3. 4. 5. Fe. Cl 2 6. nickel II iodide Fe. Cl 3 7. chromium II phosphate Cu(NO 2)2 Pb. SO 4 cobalt III hydroxide

COVALENT (nonmetal) COMPOUNDS n BEGINS WITH NONMETAL OTHER THAN H

NAMING COVALENT COMPOUNDS n Name the first elements using prefix to indicate the number of atoms present if more than one l DO NOT USE A PREFIX TO INDICATE ONE ATOM

NAMING COVALENT COMPOUNDS n Name the second element l Use prefixes to indicate the number of atoms present in the formula Add "ide" to the end of the second element l

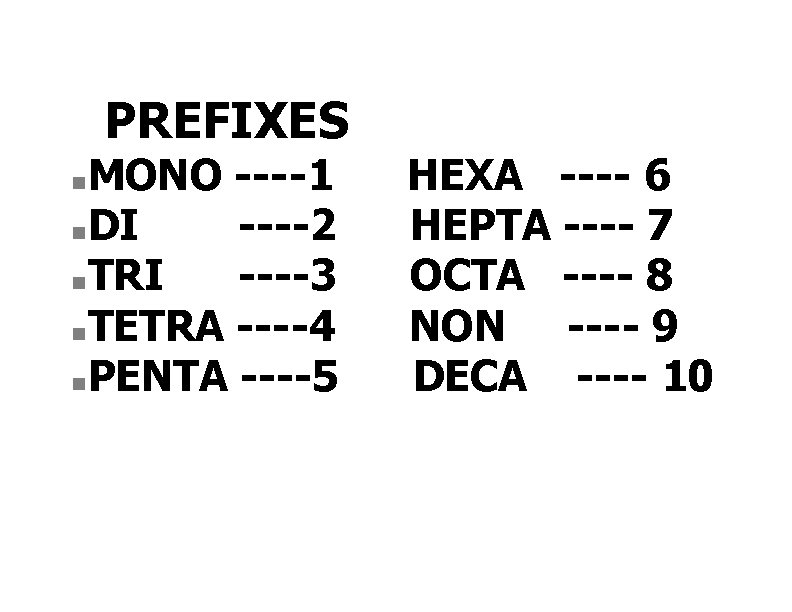
PREFIXES MONO ----1 n DI ----2 n TRI ----3 n TETRA ----4 n PENTA ----5 n HEXA ---- 6 HEPTA ---- 7 OCTA ---- 8 NON ---- 9 DECA ---- 10

PRACTICING COVALENT COMPOUNDS 1. 2. 3. 4. 5. N 2 O 3 CS 2 P 2 O 5 carbon monoxide dinitrogen trioxide

ACIDS n Compounds that begin with an "H"

BINARY ACIDS n n Contain only two elements: H and a nonmetal To name a binary acid l. Begin with the prefix "hydro" l. Drop normal ending of nonmetal name and add "ic" l. Add the word "acid"

PRACTICING BINARY ACIDS 1. HCl 4. hydroiodic acid 2. HF 5. hydrobromic 3. H 2 S acid 6. hydrophosphoric acid

TERNARY ACIDS n Contain three elements: H and polyatomic ions To name a ternary acid –Look at the polyatomic ion name and if the polyatomic ion name ends in: l"ate" - change to "ic" l"ite" - change to "ous" n. Add the word "acid" n

PRACTICING WRITING TERNARY ACIDS 1. 2. 3. 4. H 2 CO 3 H 2 SO 4 HNO 3 HCl. O 3 5. phosphorous acid 6. oxalic acid 7. nitrous acid