Unit 1 Overview Atomic Structure and Properties College



































- Slides: 47

Unit 1 Overview Atomic Structure and Properties

College Board AP Chemistry Resource Videos Topics 1. 1 -1. 4: Moles, Mass Spectrometry, Elemental Composition, and Mixtures https: //www. youtube. com/watch? v=Fpaq 5 BWRMv. Q&feature=youtu. be&fbclid=Iw. AR 2 W 0 dd. I 6 laazwpquzs. FGy. Qo 7 Tmw 8 X-NI 3 Pc 30 Gga_Fy. NZ 4 e. ZAx-s. Za. GQx. U Topics 1. 5 -1. 8 Atomic Structure, Electron Configuration, Spectroscopy, Periodic Trends https: //www. youtube. com/watch? v=7 Szn. PVl. QNM&list=PLo. Ggviqq 4845 Sy 3 Ufn. Nh_Pljz. Apt. MR 7 MQ&index=3

Topic 1. 1 - Molar Mass ● ● Learning Objective: Calculate quantities of a substance or its relative number of particles using dimensional analysis and the mole concept Essential Knowledge ○ One cannot count particles directly while performing laboratory work. Thus, there must be a connection between the masses of substances reacting and the actual number of particles undergoing chemical changes. ○ Avogadro’s number (n = 6. 022 × 1023 mol-1) provides the connection between the number of moles in a pure sample of a substance and the number of constituent particles (or formula units) of that substance. ○ Expressing the mass of an individual atom or molecule in atomic mass units (amu) is useful because the average mass in amu of one particle (atom or molecule) or formula unit of a substance will always be numerically equal to the molar mass of that substance in grams. Thus, there is a quantitative connection between the mass of a substance and the number of particles that the substance contains.

Equation

Video Resources: ● ● ● Converting between grams and particles: https: //www. youtube. com/watch? v=cvi 4 IJMZ 13 Q&disable_polymer=true The Mole: http: //www. bozemanscience. com/ap-chem-003 -the-mole Mole Conversions: http: //www. bozemanscience. com/mole-conversions Practice Problems ● How many carbon atoms are contained in 2. 8 g of C 2 H 4 ? ○ 1. 2 x 1023 atoms of C ● 0. 450 gram of Fe contains how many atoms? ○ 4. 85 x 1021 atoms of Fe

Topic 1. 2 - Mass Spectroscopy of Elements ● ● Learning Objective: Explain the quantitative relationship between the mass spectrum of an element and the masses of the element’s isotopes. Essential Knowledge: ○ The mass spectrum of a sample containing a single element can be used to determine the identity of the isotopes of that element and the relative abundance of each isotope in nature. ○ The average atomic mass of an element can be estimated from the weighted average of the isotopic masses using the mass of each isotope and its relative abundance.

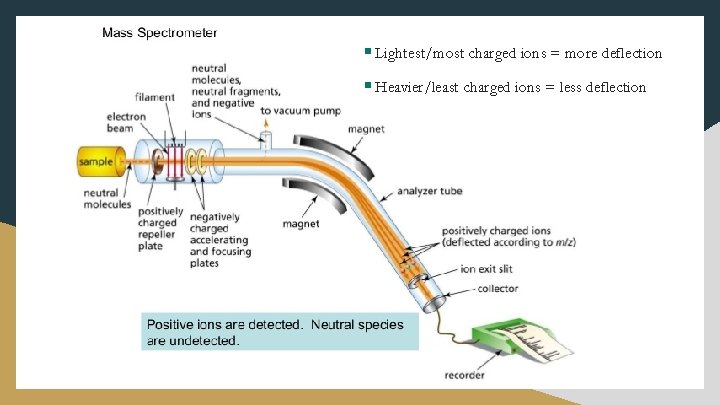
Atomic Detection § Mass Spectrometer – most accurate method currently available for comparing masses of atoms § This instrument works as follows: Ø Step 1: Ionization Sample is passed into a beam of high-speed electrons, which knocks electrons off the atoms or molecules being analyzed; creates (+) ions Ø Step 2: Acceleration An electric field is used to accelerate these ions into a singular beam

Atomic Detection Ø Step 3: Deflection The magnetic field of the ion interacts with an applied magnetic field, which changes the path of the ion according to its mass & charge § Lightest/most charged ions = more deflection § Heavier/least charged ions = less deflection Ø Step 4: Detection A comparison of the positions where the ions hit the detector plate gives accurate values of their relative masses; also, can be used to determine isotopic composition of a natural element

§ Lightest/most charged ions = more deflection § Heavier/least charged ions = less deflection

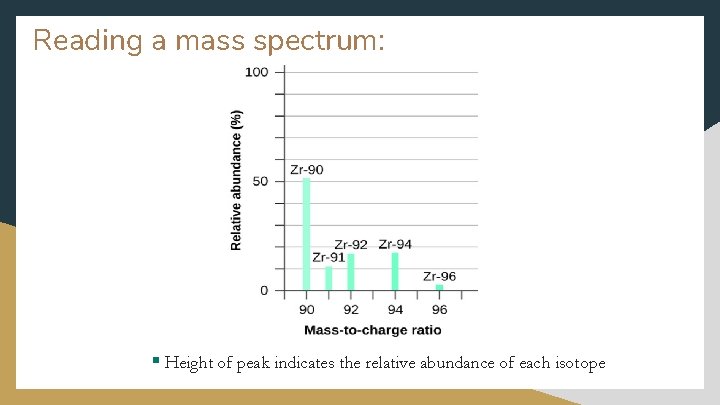
Reading a mass spectrum: § Height of peak indicates the relative abundance of each isotope

Atomic Mass § Most elements occur as a mixture of two or more isotopes § The percent abundance of each isotope can be used to find a weighted average of their masses (multiply the atomic mass and % abundance in decimal form for each isotope and add the results) § Example: copper isotopes Isotopes Copper-63 Copper-65 At. Mass 62. 94 amu 64. 93 amu % Abundance 69. 17% 30. 83% Cu-63 62. 94 x 0. 6917 = 43. 54 amu Cu-65 64. 93 x 0. 3083 = 20. 02 amu Weighted at. mass = 43. 54 amu + 20. 02 amu = 63. 56 amu

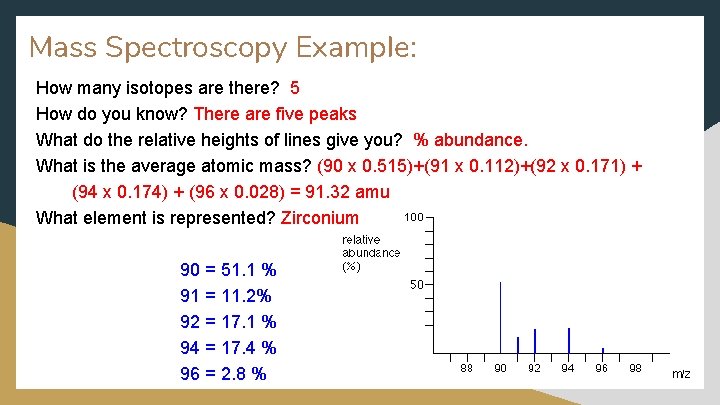
Mass Spectroscopy Example: How many isotopes are there? 5 How do you know? There are five peaks What do the relative heights of lines give you? % abundance. What is the average atomic mass? (90 x 0. 515)+(91 x 0. 112)+(92 x 0. 171) + (94 x 0. 174) + (96 x 0. 028) = 91. 32 amu What element is represented? Zirconium 90 = 51. 1 % 91 = 11. 2% 92 = 17. 1 % 94 = 17. 4 % 96 = 2. 8 %

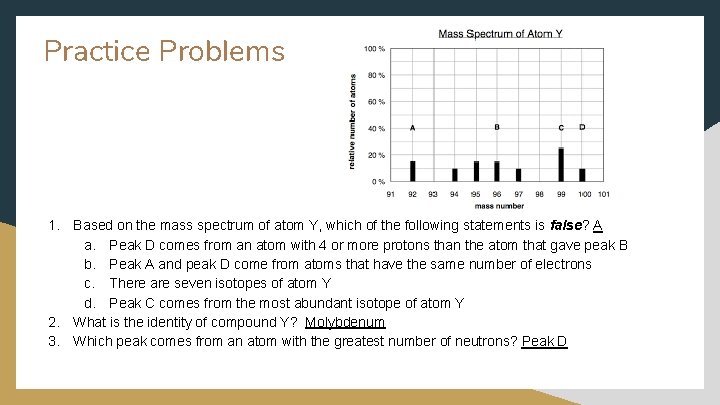
Practice Problems 1. Based on the mass spectrum of atom Y, which of the following statements is false? A a. Peak D comes from an atom with 4 or more protons than the atom that gave peak B b. Peak A and peak D come from atoms that have the same number of electrons c. There are seven isotopes of atom Y d. Peak C comes from the most abundant isotope of atom Y 2. What is the identity of compound Y? Molybdenum 3. Which peak comes from an atom with the greatest number of neutrons? Peak D

Video Resources: ● ● Mass Spectroscopy: http: //www. bozemanscience. com/ap-chem-009 -mass-spectrometry KHAN Academy: https: //www. youtube. com/watch? v=myol. F-h 1 k. KI

Topic 1. 3 - Elemental Composition of Pure Substances ● ● Learning Objective: Explain the quantitative relationship between the elemental composition by mass and the empirical formula of a pure substance. Essential Knowledge: ○ Some pure substances are composed of individual molecules, while others consist of atoms or ions held together in fixed proportions as described by a formula unit. ○ According to the law of definite proportions, the ratio of the masses of the constituent elements in any pure sample of that compound is always the same. ○ The chemical formula that lists the lowest whole number ratio of atoms of the elements in a compound is the empirical formula.

• • The Law of Definite Proportions: a given chemical compound always contains the same elements in the exact same proportions by mass. • Example: any sample of pure water contains 11. 19% hydrogen and 88. 81% oxygen by mass. Empirical Formula: a chemical formula showing the simplest ratio of elements in a compound Video Resources ● ● Empirical Formulas and Chemical Analysis: http: //www. bozemanscience. com/apchem-002 -chemical-analysis Calculating Empirical Formulas: https: //www. youtube. com/watch? v=Tdy. Nr. GYznv. E

Practice Problems ● ● Barium carbonate, a white powder used in paints, enamels, and ceramics, has the following composition: Ba, 69. 58%; C, 6. 090%; O, 24. 32%. What is its empirical formula? ○ Ba. CO 3 A sample of an unknown compound with a mass of 2. 571 grams was found to contain 1. 102 grams of C and 1. 469 grams of oxygen. What is its empirical formula? ○ CO

Topic 1. 4 - Composition of Mixtures ● ● Learning Objective: Explain the quantitative relationship between the elemental composition by mass and the composition of substances in a mixture. Essential Knowledge: ○ While pure substances contain molecules or formula units of a single type, mixtures contain molecules or formula units of two or more types, whose relative proportions can vary. ○ Elemental analysis can be used to determine the relative numbers of atoms in a substance and to determine its purity.

Video Resources ● ● Analyzing Chemical Purity - a worked example: https: //www. youtube. com/watch? v=K 5 HPs. Kdgxs. I AP Chemistry Free Response - another worked example: https: //www. khanacademy. org/science/chemistry/studying-for-ap-chemistryexam/2015 -ap-chemistry-free-response-questions/v/2015 -ap-chemistry-freeresponse-4

Topic 1. 5 - Atomic Structure and Electron Configurations ● ● Learning Objective: Represent the electron configuration of an element or ions of an element using the Aufbau principle. Essential Knowledge: ○ The atom is composed of negatively charged electrons and a positively charged nucleus that is made of protons and neutrons. ○ Coulomb’s law is used to calculate the force between two charged particles. ○ In atoms and ions, the electrons can be thought of as being in “shells (energy levels)” and “subshells (sublevels), ” as described by the electron configuration. Inner electrons are called core electrons, and outer electrons are called valence electrons. The electron configuration is explained by quantum mechanics, as delineated in the Aufbau principle and exemplified in the periodic table of the elements ○ The relative energy required to remove an electron from different subshells of an atom or ion or from the same subshell in different atoms or ions (ionization energy) can be estimated through a qualitative application of Coulomb’s law. This energy is related to the distance from the nucleus and the effective (shield) charge of the nucleus.

Coulomb’s Law ● ● States that like charges repel and opposite charges attract, with a force proportional to the product of the charges and inversely proportional to the square of the distance between them. Effective Nuclear Charge: the net positive charge experienced by valence electrons. It can be approximated by the equation: ○ Zeff = Z – S, where Z is the atomic number and S is the number of shielding (core) electrons. Examples: Carbon Zeff = 6 protons – 2 core electrons = + 4 Nitrogen Zeff = 7 protons – 2 core electrons = + 5

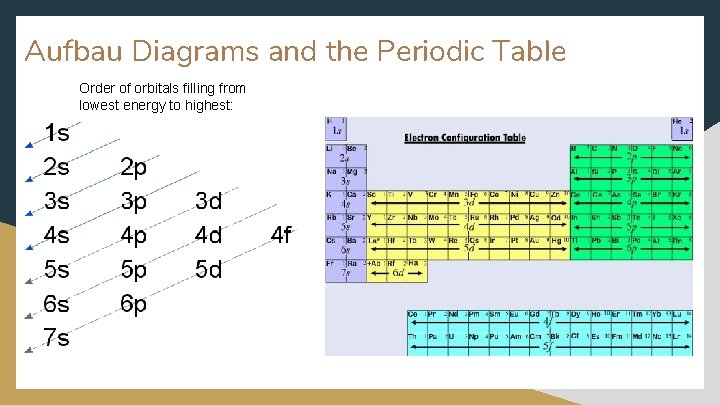
Aufbau Diagrams and the Periodic Table Order of orbitals filling from lowest energy to highest:

Practice Problems ● ● ● Determine the number of protons, neutrons, and electrons in an isotope of germanium-74 ○ Protons = 32, electrons = 32, neutrons = 42 Write the complete electron configuration for K+ ○ 1 s 22 p 63 s 23 p 6 (18 electrons total) What can be said about the ionization energy of the element above? ○ The ionization energy would be very large because the potassium ion has a complete octet and an electron configuration that matches that of argon

Video Resources ● ● ● Coulomb’s Law: http: //www. bozemanscience. com/ap-chem-004 -coulombs-law Coulomb’s Law Concept Map: https: //static 1. squarespace. com/static/50 d 5 cc 57 e 4 b 0 e 383 f 5 b 1 eb 34/t/52 a 32 aa 5 e 4 b 0 a 695 ee 3 df 4 bf/1386424997045/AP+Chem 004+Coulomb%27 s+Law+CM+PDF. pdf Electron Configuration: http: //www. bozemanscience. com/ap-chem-005 -electronconfiguration

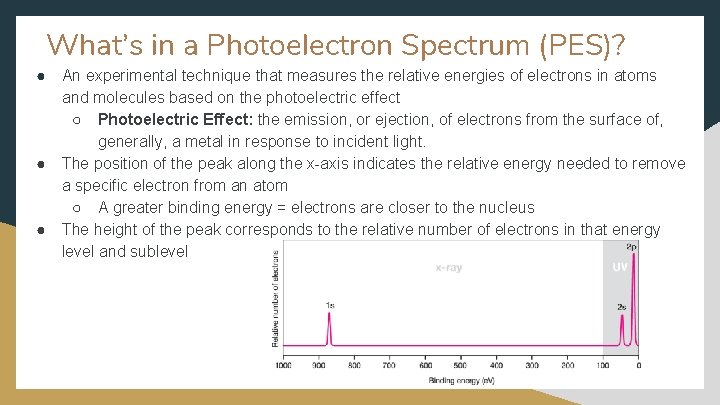
Topic 1. 6 - Photoelectron Spectroscopy ● ● Learning Objective: Explain the relationship between the photoelectron spectrum of an atom or ion and: ○ The electron configuration of the species. ○ The interactions between the electrons and the nucleus. Essential Knowledge: ○ The energies of the electrons in a given shell can be measured experimentally with photoelectron spectroscopy (PES). The position of each peak in the PES spectrum is related to the energy required to remove an electron from the corresponding subshell, and the height of each peak is (ideally) proportional to the number of electrons in that subshell.

What’s in a Photoelectron Spectrum (PES)? ● ● ● An experimental technique that measures the relative energies of electrons in atoms and molecules based on the photoelectric effect ○ Photoelectric Effect: the emission, or ejection, of electrons from the surface of, generally, a metal in response to incident light. The position of the peak along the x-axis indicates the relative energy needed to remove a specific electron from an atom ○ A greater binding energy = electrons are closer to the nucleus The height of the peak corresponds to the relative number of electrons in that energy level and sublevel

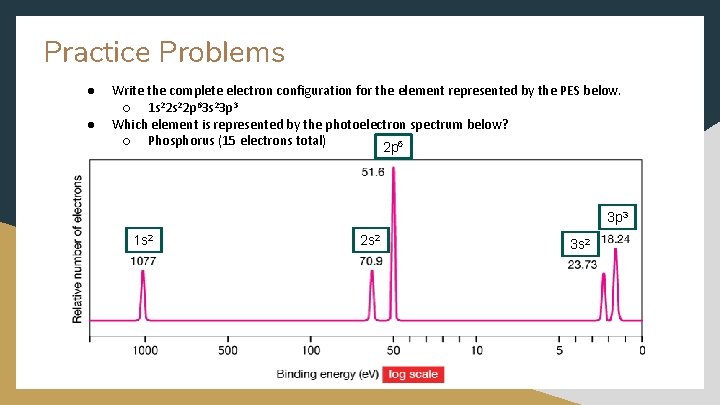
Practice Problems ● ● Write the complete electron configuration for the element represented by the PES below. ○ 1 s 22 p 63 s 23 p 3 Which element is represented by the photoelectron spectrum below? ○ Phosphorus (15 electrons total) 2 p 6 3 p 3 1 s 2 2 s 2 3 s 2

Video Resources ● ● Interpreting PES data: https: //www. youtube. com/watch? v=gy. KD 6 QULa 0 A Photoelectron Spectroscopy: https: //www. youtube. com/watch? v=ZI 6 NQ 9 Lg. HJQ

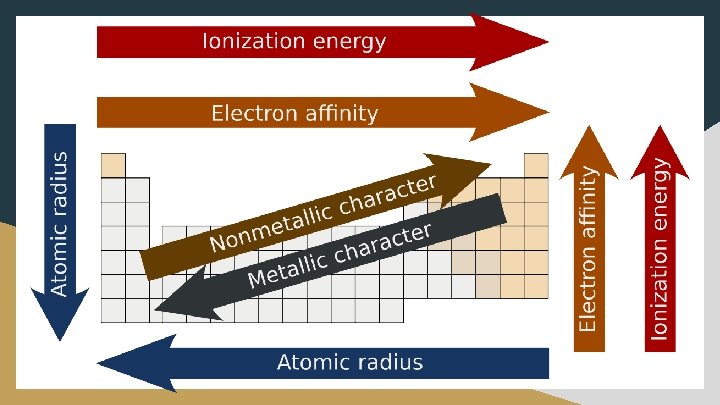
Topic 1. 7 - Periodic Trends ● ● Learning Objective: Explain the relationship between trends in atomic properties of elements and electronic structure and periodicity. Essential Knowledge: ○ The organization of the periodic table is based on the recurring properties of the elements and explained by the pattern of electron configurations and the presence of completely or partially filled shells (and subshells) of electrons in atoms. ○ Trends in atomic properties within the periodic table (periodicity) can be qualitatively understood through the position of the element in the periodic table, Coulomb’s law, the shell model, and the concept of shielding/effective nuclear charge. These properties include: ■ Ionization energy ■ Atomic and ionic radii ■ Electron affinity ■ Electronegativity ○ The periodicity is useful to predict /estimate values of properties in the absence of data.

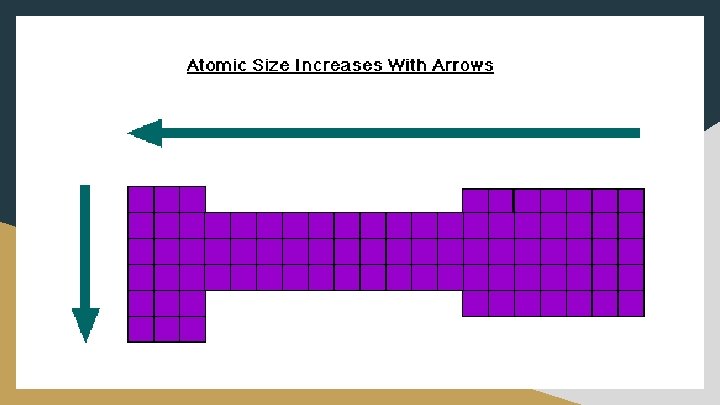
Atomic Radius ● Atomic radius decreases as you move right across the periodic table ○ The effective nuclear charge increases (more protons in nucleus), drawing electrons closer to the nucleus (more attracted); the number of energy levels remains constant as you go across a row, so no extra shielding occurs ● Atomic radius increases as you move down a group ○ New energy levels are added as you move down the group (electrons further from nucleus, so less attracted), increasing the atomic radius. Also, shielding increases because adding more core electrons, which increases the electron repulsion


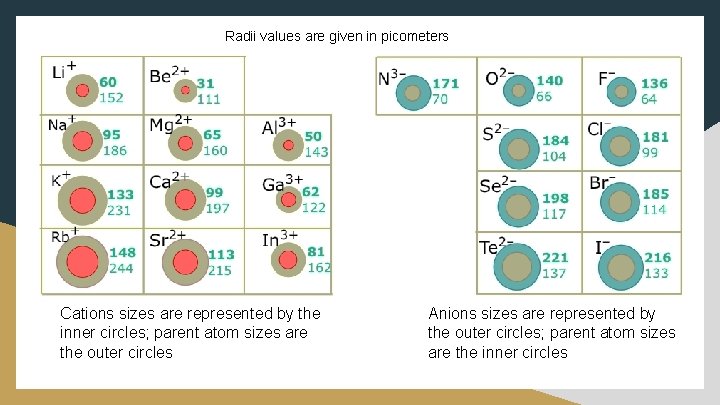
Ionic Radius ● Cations are smaller than their parent atoms because the protons outnumber the electrons ○ Effective nuclear charge increases while electron repulsion decreases ● Anions are larger than their parent atoms because the electrons outnumber the protons ○ Effective nuclear charge decreases while electron repulsion increases

Radii values are given in picometers Cations sizes are represented by the inner circles; parent atom sizes are the outer circles Anions sizes are represented by the outer circles; parent atom sizes are the inner circles

Ionic Radius Isoelectronic – ions containing the same number of electrons o How do you determine their size? Consider the number of protons. o Which ion should be slightly smaller—oxide or fluoride? Solution: Both have 10 electrons, but F-1 < O-2 because fluorine has an extra proton (greater attraction for electrons)

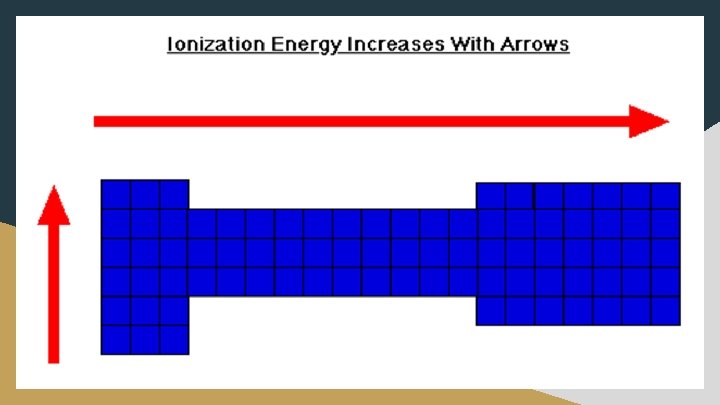
Ionization Energy (I. E. ) ● The energy required to remove an electron from an atom; creates a cation ○ X + I. E. X+ + e○ Ionization energy increases as you move right across a row on the periodic table ■ Effective nuclear charge increases as you move toward the right ● The number of protons in the nucleus increases without the addition of shielding electrons. ● As the attraction for electrons increases, it is more difficult to remove electrons and the ionization energy increases ○ Ionization energy decreases as you move down a group ■ A new energy level is added as you move down the group ■ Electrons in higher energy level are located further from the nucleus so they are less attracted and are therefore easier to remove ○ Smaller atomic radius = greater ionization energy


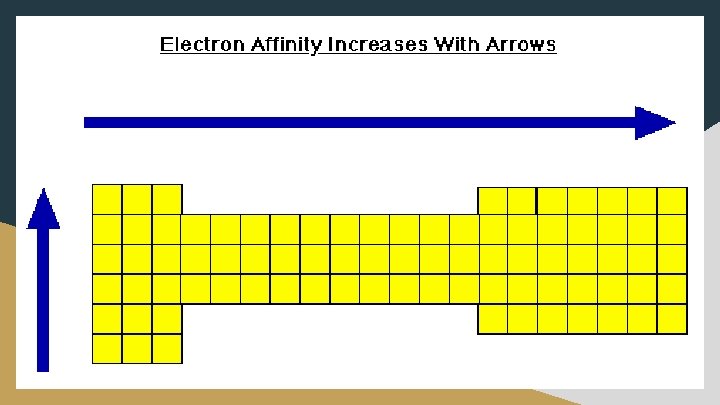
Electron Affinity (E. A. ) ● The amount of energy released when an electron is added to a neutral atom; creates an anion ○ X + e- X- + E. A. ○ Values are negative ○ A more negative electron affinity corresponds to a greater attraction for an electron ● E. A. increases as move right across periods ○ The electrons added to energy levels become closer to the nucleus, thus a stronger attraction between the nucleus and its electrons exists ● E. A. decreases as go down a group ○ The electrons are added to energy levels that are further from the nucleus, thus a weaker attraction between the nucleus and its electron exists


Electronegativity o Electronegativity = the ability of an atom to attract shared electrons to itself when bonded in a molecule; think of it as “tug of war” • Scale of values (0 -4. 0) developed by Linus Pauling • Fluorine has the highest eneg. ; highest Zeff and smallest size • Francium has the lowest eneg. ; lowest Zeff and largest size


Practice Problems ● Use the following set of elements to answer the questions that follow: Rb, Cd, Pd, Cl, Si ○ Which element has the largest atomic radius? Rb ○ ● Which element has the highest electronegativity? Cl According to the data in the table below, element Y represents which second period element? Answer = Be


Video Resources ● ● ● Professor Dave: https: //www. youtube. com/watch? v=he. Pb 00 Cqv. P 0 KHAN Academy - Atomic Radius: https: //www. khanacademy. org/science/chemistry/periodic-table/periodic -table-trends-bonding/v/atomic-radius-trend KHAN Academy - Ionization Energy: https: //www. khanacademy. org/science/chemistry/periodictable/periodic-table-trends-bonding/v/ionization-energy-trends KHAN Academy - Electron Affinity: https: //www. khanacademy. org/science/chemistry/periodictable/periodic-table-trends-bonding/v/electron-affinity KHAN Academy - Electronegativity: https: //www. khanacademy. org/science/chemistry/periodictable/periodic-table-trends-bonding/v/electronegativity-trends

Topic 1. 8 - Valence Electrons and Ionic Compounds ● ● Learning Objective: Explain the relationship between trends in the reactivity of elements and periodicity Essential Knowledge: ○ The likelihood that two elements will form a chemical bond is determined by the interactions between the valence electrons and nuclei of elements. ○ Elements in the same column of the periodic table tend to form analogous compounds. ○ Typical charges of atoms in ionic compounds are governed by their location on the periodic table and the number of valence electrons.

Practice Problems ● ● ● Element X has an electron configuration of 1 s 22 p 5. What is the most likely compound that will be formed between lithium and element X? ○ Li. X (element X needs to gain 1 electron to achieve an octet, making it a -1 ion) Explain why ionic compounds have high boiling points. ○ Large difference in electronegativities give elements in ionic compounds a strong electrostatic force of attraction. A large amount of energy is required to break the strong coulombic forces between oppositely charged ions Which of the following ions is isoelectronic (has same number of electrons) with S 2 -? ○ O 2○ K 1+ ○ Na 1+ ○ Mg 2+

Practice Problems ● What is the most likely empirical formula of a compound formed by element X and oxygen? ○ XO ○ X 2 O 3 ○ X 2 O (X has 1 valence e- and O has 2 valence e-) ○ XO 2 ● Which elements could element X possibly represent? ○ Sodium, Potassium, Rubidium, Cesium

Video Resources ● ● Bozeman Science Periodicity: http: //www. bozemanscience. com/ap-chem-006 -periodicity KHAN Academy - Valence Electrons and Ionic Compounds: https: //www. youtube. com/watch? v=XPnl. IEk 7 Xr. U