Unit 1 Day 7 Solubility Warmup What is

Unit 1_ Day 7_ Solubility Warm-up: What is the meaning of diffusion? Provide an example. Objective : Students will be able to explain dissolving and diffusion by using the particle model of matter.

Demo • Demo: Body Spray

Another Example:

Liquids: Solutions and Solubility

What is a Solution? • Solute + Solvent = Solution Example: food coloring and water • Solvent: Dissolves the solute. • Solute: Dissolved particles.

Solvation – is the process of attraction and organization of molecule of a solvent with molecules or ions of a solute. – As ions dissolve in a solvent they spread out and are surrounded by solvent molecules.

Demo: Solute and Solvent Mix • When the drink mix is added to the water, the water molecules (the solvent) and the sucrose molecules (which comprise the solute) are attracted to each other. • The mutual attraction between the water molecules and the sucrose molecules cause the sugar to dissolve in water.

Solution Formation • Think of a cup of tea… – Have you noticed that sugar granules dissolve faster than sugar cubes? – Why does sugar dissolves faster when you stir? – Explain how it dissolves faster in hot tea than iced tea?

What Affects Solution Formation? • Compositions of Solute and Solvent • Surface Area/ Stirring • Temperature

Composition of Solute and Solvent • Constituents of a solution must be compatible. For example, oil and water do not mix.

Surface Area/ Stirring • Stirring speeds up the process of dissolution because it bring new solvent in contact with the surface of the solute. This increases the relative surface area of the liquid.

Demo: Sugar in water 1. Why does sugar disappear when it dissolves in water? 2. What can we conclude about what sugar is made of based on this lab?

Temperature • At higher temperatures, the kinetic energy of the solvent is higher thus they move faster. As they move faster, there’s an increase in the frequency and force of the collisions between molecules. Thus, there is faster dissolving. • In conclusion: http: //youtu. be/n 5 nubvw. JJQM

Independent Practice Demo: Food coloring diffuses in Hot and Cold water 1. Create a drawing that illustrates the difference between the hot particles and the cold particles. 2. Explain your drawing. How the particles of the hot water are different from the particles of the cold water? 3. What conclusion can you draw about particles? Defend your answer with evidence from today’s lab.


Density Objective: Students will be able to compare the densities of multiple substances both quantitatively and qualitatively.

Density= Mass Volume D= m V

Mass • Mass: Amount of matter in a substance. • Don’t confuse with weight. • Weight: the force with which the earth pulls on the substance.

Question Which weighs more? 50 kilograms of iron Or 50 kilograms of feathers

Question Which has a greater density? iron Or feathers

Common Units of Density= Mass Volume g/m. L = g/cm 3 1 cm 3=1 m. L 1 dm 3=1 L kg/m 3

DENSITY d=m/v DENSITY OF WATER = 1 g/cm 3 • If the object has a higher density, it sinks. • If the object has a lower density, it floats. • if the object has the same density, it flinks (floats in the middle of the water).

AN OBJECT FLOATS CAUSES: • Object is less dense than the fluid • Object decreases its mass and becomes less dense than the fluid. • Object increases its volume and becomes denser than the fluid.

AN OBJECT SINKS CAUSES: • Object is denser than the fluid • Object increases its mass and becomes denser than the fluid. • Object decreases its volume and becomes denser than the fluid.

Making a general statement comparing the densities of solids, liquids and gases • Solids are more dense. • Liquids are less dense than solids • Gases are least dense

What substances have the greatest density and why? • Solids have the greatest densities because the particles in the solids are packed tightly together and there is little space to move.

What substances have the least densities? • Gases have the least densities because the particles in gases are not packed tightly together and move around more freely.
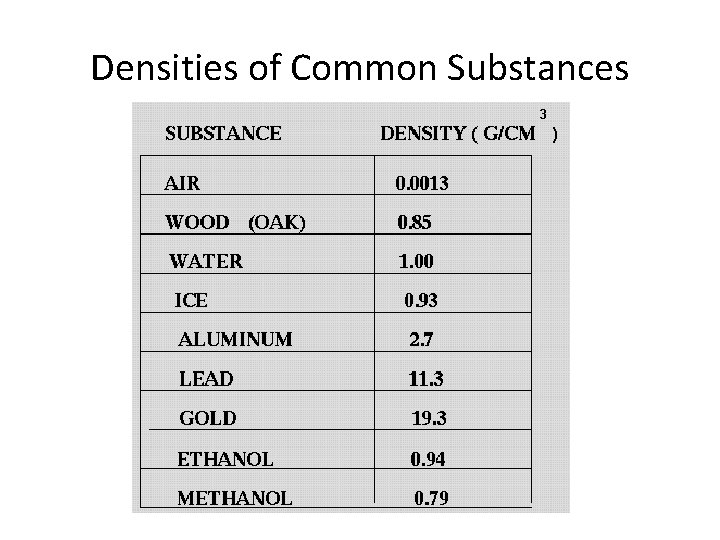
Densities of Common Substances

Density as a Function of Temperature For most substances, as temperature increases the volume increases. As a result, the density decreases.

Density of Water

Density of Water At 4 o. C water has its maximum density of 1 g/cm 3 Convert to kg/m 3: 1000 kg/m 3

Life Application • How does density help engineers? Knowing the density of substances allows engineers to design dams, build ships and reinforce concrete to avoid stresses that may break structures like bridges apart.

Life Application • How does density help manufacturers? Density allows manufacturers to make moulds, create alloys, and design material such as plastics to fit a number of application from toys to automobiles.

Let’s Practice • Density Practice Problems Worksheet. “I” 1. What is the density of CO gas if 0. 196 g occupies a volume of 100 ml? “WE” 2. A block of wood 3 cm on each side has a mass of 27 g. What is the density of the block? (Hint, don’t forget to find the volume of the wood. ) 3. An irregularly shaped stone was lowered into a graduated cylinder holding a volume of water equal to 2 ml. The height of the water rose to 7 ml. If the mass of the stone was 25 g, what was its density? “YOU” Complete problems 4 through 7.

Closure • Exit Slip: Based on what you have learned, explain the diffusion of a lump of sugar in water.
- Slides: 35