Unit 1 Chemistry Bonding A Chemical Bond is

Unit 1: Chemistry Bonding!!!

A Chemical Bond… • …is a union between the electron structures of atoms. Three types of bonds – ionic, covalent, and hydrogen

Ionic Bonds • Formed when electrons are transferred from one atom to another forming ions. • Oppositely charged ions attract forming an ionic bond. Example: Na. Cl (table salt)


Covalent Bonds • Formed when electrons are shared between atoms. Single bond = share one pair of e. Double bond = share two pairs of e. Triple bond = share three pairs of e-
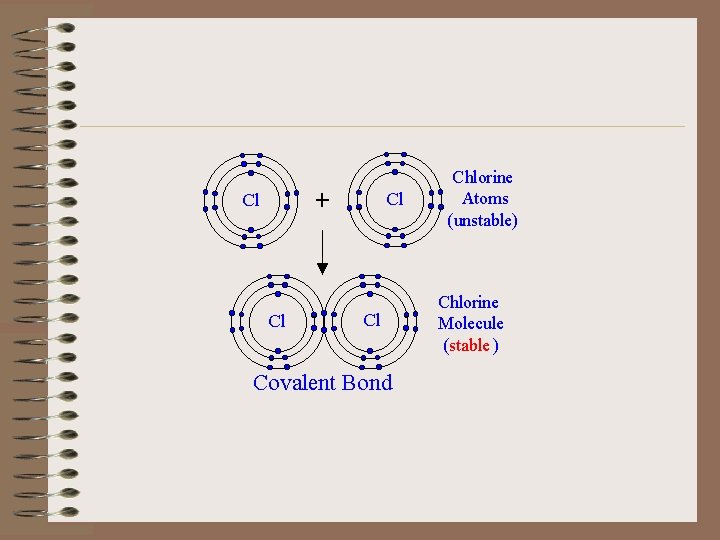

Covalent bonds cont. • When the atoms are shared equally it is said to be a nonpolar covalent bond. • When the atoms are shared unequally it is said to be a polar covalent bond.
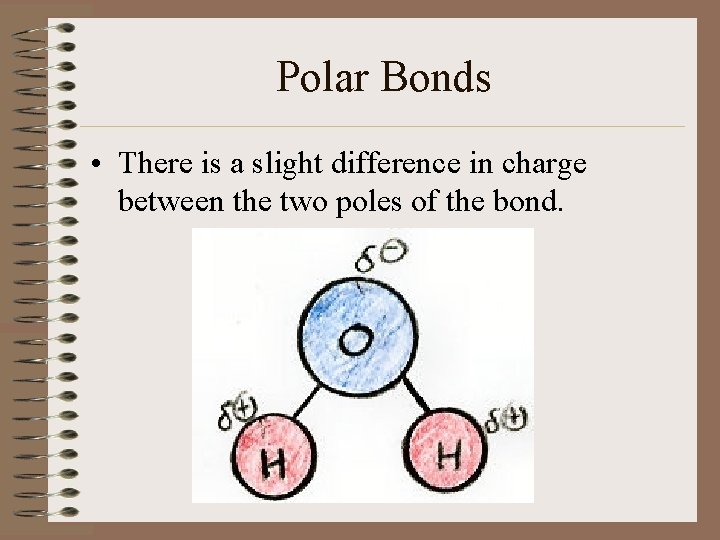
Polar Bonds • There is a slight difference in charge between the two poles of the bond.

Water Molecules • Water is an example of a polar molecule b/c there is an uneven distribution of ebetween the hydrogen atoms and the oxygen atom.

Hydrogen Bonds • Two water molecules are held together by hydrogen bonds • The hydrogen atom (+) of one water molecule is attracted to the oxygen (-) of another water molecule.
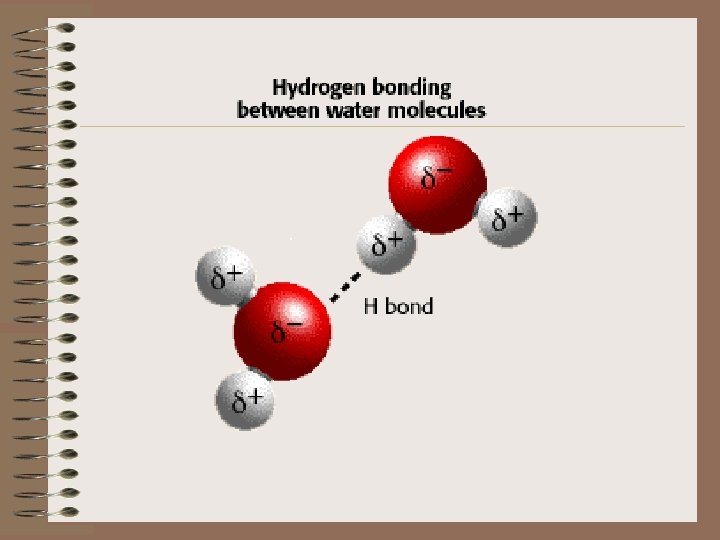

• Ability of water to form multiple hydrogen bonds allows it to be cohesive. • Cohesion = attraction between molecules of the same substance…water sticks to itself! • Gives water its surface tension!

• Adhesion = attraction between molecules of different substances. • ie) water creeping up the side of a glass beaker causing a meniscus

Capillary Action • Adhesion pulls the water molecule up a small tube…against gravity. • Cohesion keeps the water together as it rises.

p. H • Water can break down to form hydrogen ions (H+) and hydroxide ions (OH-) • p. H scale measures the concentration of H+ ions

p. H Scale • Scale = 0 - 14 • Pure water is neutral at 7. There is an equal amount of H+ and OH • Acids are below 7 on the scale and have a higher concentration of H+. • Bases are above 7 on the scale and have a higher concentration of OH-

p. H Scale
- Slides: 17