Unit 1 Basics of Chemistry Matter Anything that

Unit 1 Basics of Chemistry

Matter – Anything that has mass and takes up space Atoms – The basic unit of all matter

3 Subatomic Particles (Smaller parts) Protons Neutrons Electrons Each of these particles has a charge, a mass, and location in the atom
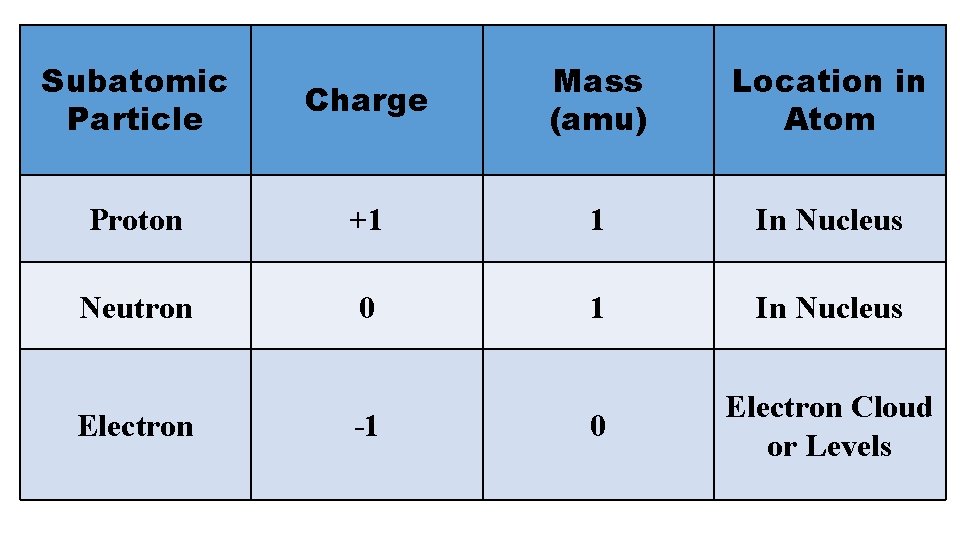
Subatomic Particle Charge Mass (amu) Location in Atom Proton +1 1 In Nucleus Neutron 0 1 In Nucleus 0 Electron Cloud or Levels Electron -1

The nucleus (Protons and Neutrons) make up more than 99% of the atom’s mass A specific type of atom is called an element The Periodic Table shows what elements (or types of atoms) exist
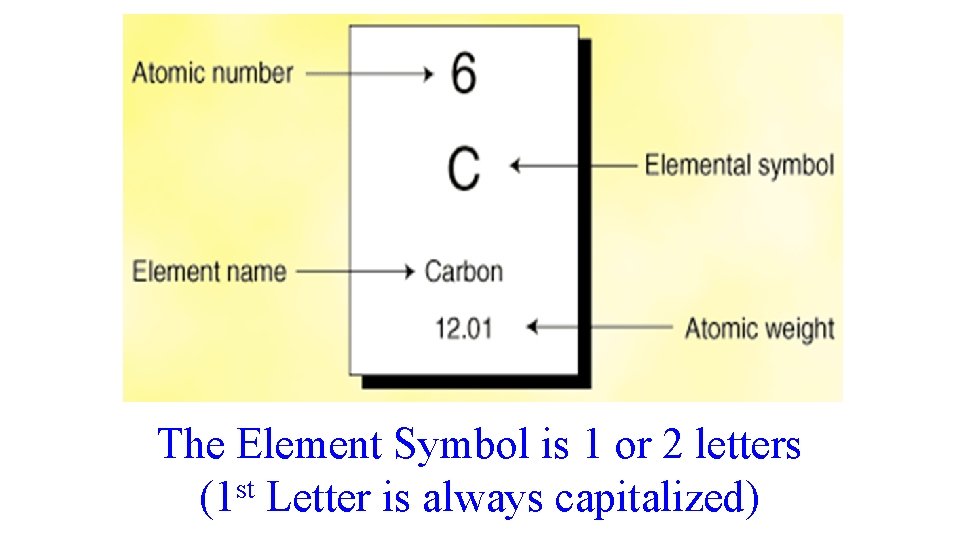
The Element Symbol is 1 or 2 letters (1 st Letter is always capitalized)

Atomic Number – Tells you the number of Protons an atom has The number of Protons will tell you what kind of atom (or element) you have (Build examples on the board)

The number of electrons and neutrons an element has can change but not the number of protons Don’t mess with Protons!

Atomic Mass Number – Tell you the average mass (weight) of the element To find the Mass of an atom add the mass of the Protons and Neutrons (# of Protons + # of Neutrons = Mass) (Build examples on the board)

Isotopes – Atoms of the same element that have a different number of neutrons Isotopes are named according to their weight (Element name + weight) Example: Carbon-12 or Carbon-14
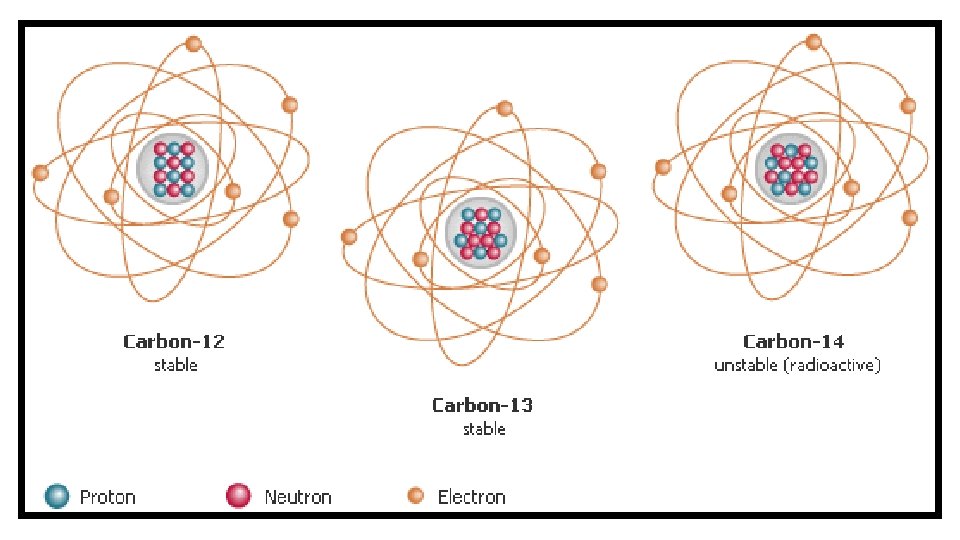

Most atoms are neutral (Which means the number of protons and electrons are the same) The Electron Cloud Model shows the most likely place an electron will be found Electrons are found in areas called levels or shells (certain distances from the nucleus)

How do I know where the electrons go? 1. Determine how many electrons you have 2. Place them in the lowest levels first The 1 st Level can hold 2 electrons All other levels can hold 8 electrons

Valence Electron: Any electron in the outer most (or highest) level Valence electrons are important because they are the electrons which form chemical bonds

A horizontal row is known as a Period The period tells you what level is the outer most level (valence shell)

Each column is called a Group Elements within a group have similar properties because they have the same number of valence electron The “A” Groups indicate how many valence electrons are in the outer most shell/level
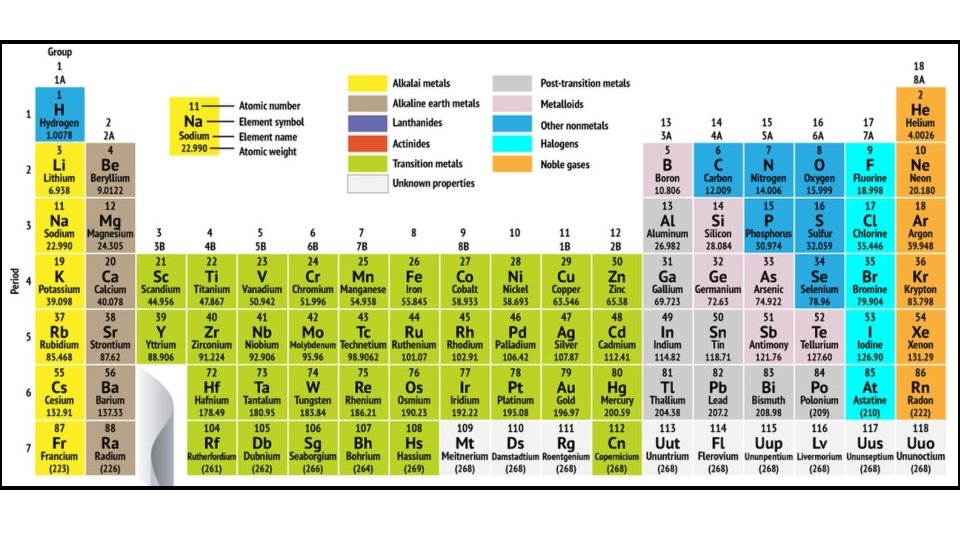

If the outer most level is full of electrons it is stable If the outer most level is not full of electrons it is unstable All atoms (except for Hydrogen and Helium) need 8 valence electrons to be stable

Atoms will do what it takes to become stable Atoms will give away electrons, take electrons, or share electrons to become stable

Ions are formed when electrons are given or taken away from an atom Ions: Positively or negatively charged atoms
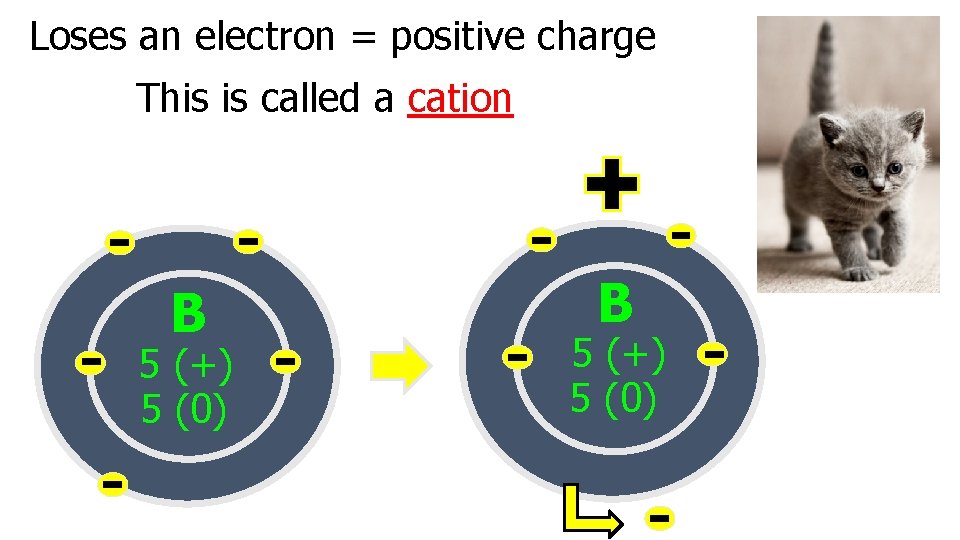
Loses an electron = positive charge This is called a cation B 5 (+) 5 (0)
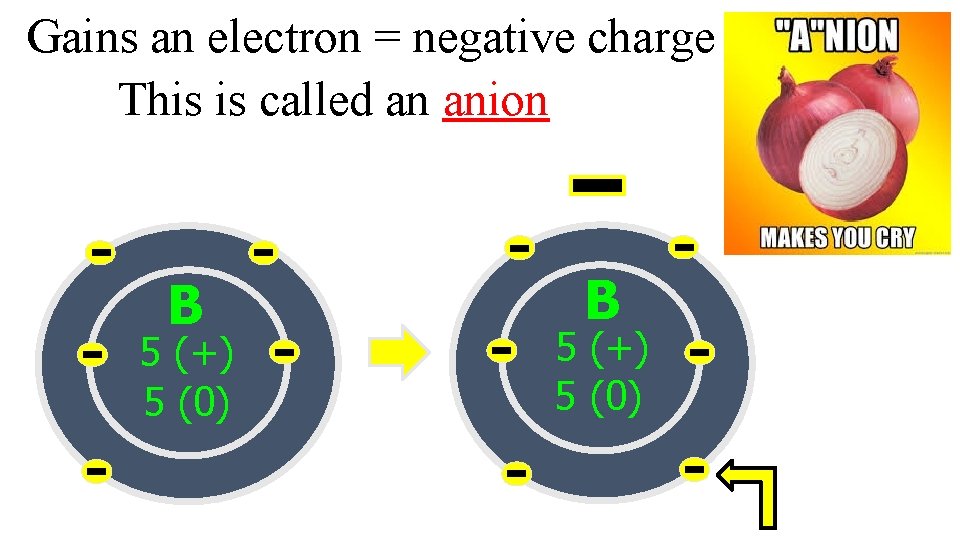
Gains an electron = negative charge This is called an anion B 5 (+) 5 (0)

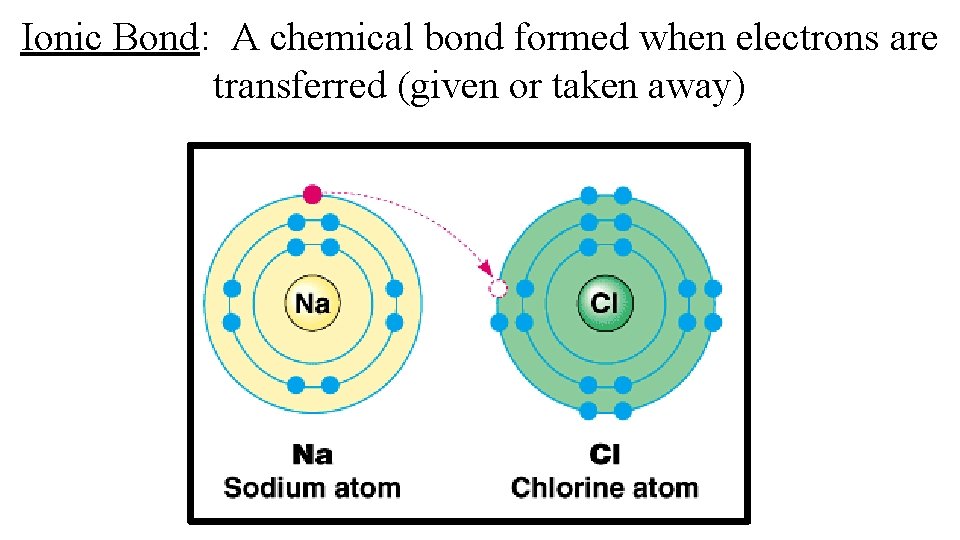
Ionic Bond: A chemical bond formed when electrons are transferred (given or taken away)
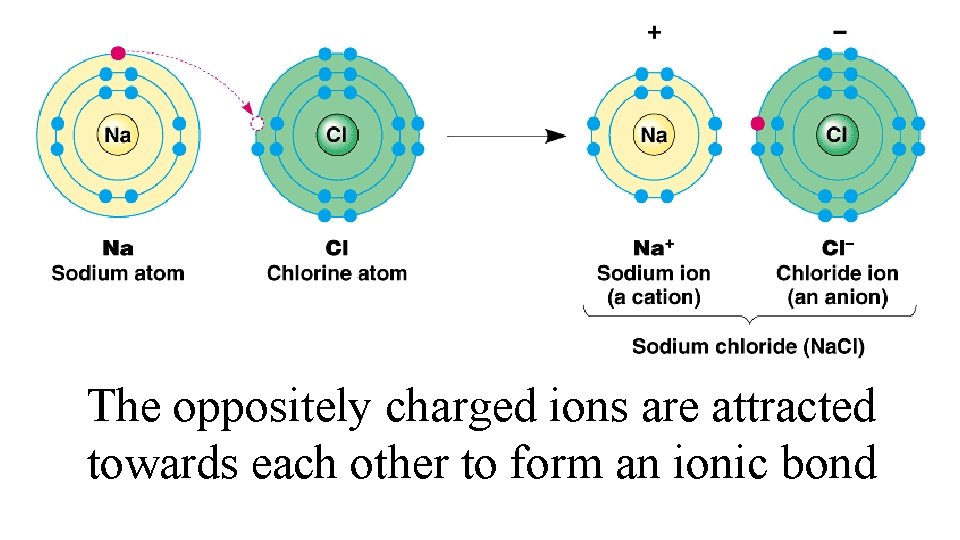
The oppositely charged ions are attracted towards each other to form an ionic bond
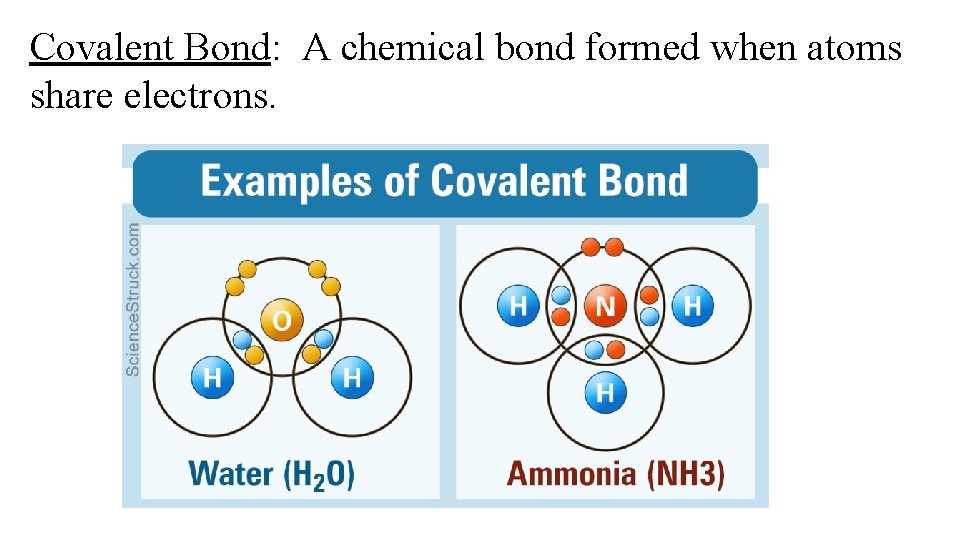
Covalent Bond: A chemical bond formed when atoms share electrons.
- Slides: 26