UNIT 1 a Syllabus Expectations and Lab Safety

UNIT 1 a. Syllabus Expectations and Lab Safety b. Scientific Measurement, Accuracy, Precision c. Scientific Notation and Sig Figs d. Calculations and Conversions

Bell Work- Lab Safety DO THIS WORK IN THE BACK HALF OF YOUR LABORATORY NOTEBOOK – MARK THE PAGE 1. Which of the following are safety equipment found in the laboratory: a. Fire extinguisher b. Graduated Cylinder c. Erlenmeyer Flask d. Safety goggles e. Eye Wash Station 3. Where can you find important safety information? a. NFPA diamond b. MSDS Sheets c. The red folder in the back of the room d. Reagent Labels e. The FDA website 4. What is the cell phone policy in our Chemistry lab? _____________________________________

Introduction to Accuracy & Precision https: //youtu. be/h. RAFPd. Dppzs
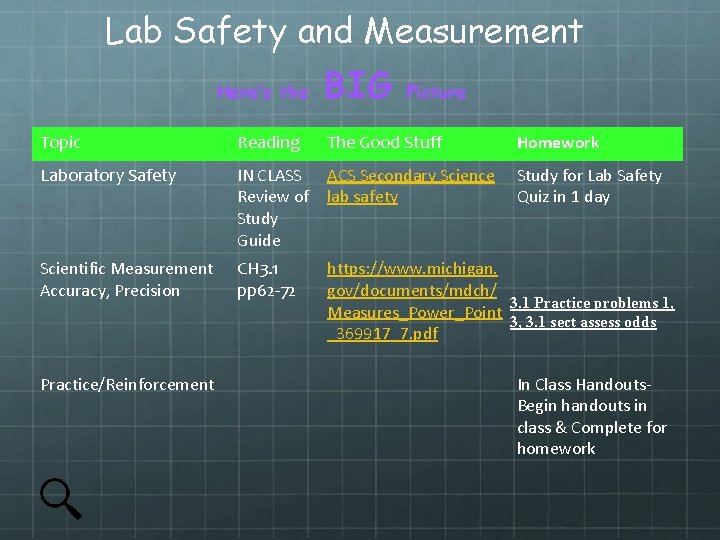
Lab Safety and Measurement Here’s the BIG Picture Topic Reading The Good Stuff Homework Laboratory Safety IN CLASS Review of Study Guide ACS Secondary Science lab safety Study for Lab Safety Quiz in 1 day Scientific Measurement Accuracy, Precision CH 3. 1 pp 62 -72 https: //www. michigan. gov/documents/mdch/ 3. 1 Practice problems 1, Measures_Power_Point 3, 3. 1 sect assess odds _369917_7. pdf Practice/Reinforcement In Class Handouts. Begin handouts in class & Complete for homework

Unit 1 b: Measurements in Science • Measurement is one of the most basic foundations of good laboratory technique. • Today, we will be practicing measuring objects and solutions using those techniques in the measurement lab. • But first…. What is good technique? • Accuracy • Precision

What is the difference between accuracy and precision? Can something be both precise and accurate? Can something be neither accurate nor precise?
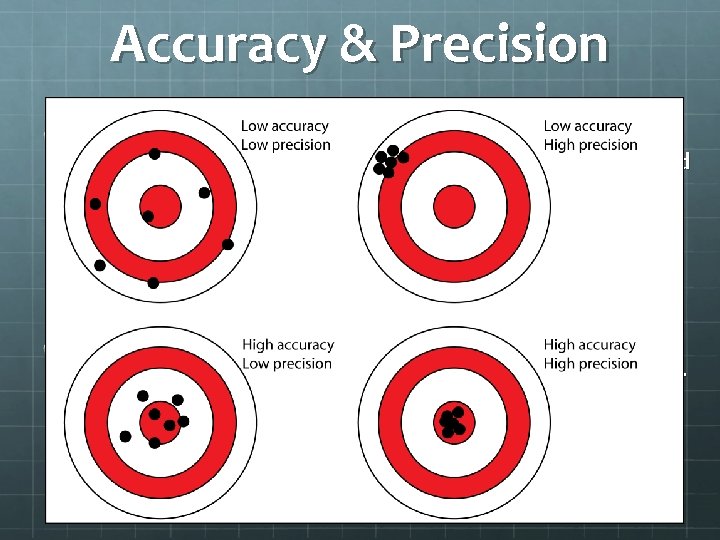
Accuracy & Precision The degree to which the result of a measurement or calculation conforms to the correct value or a standard. A measurement or calculation as represented by the consistency of results. How close you are to the actual answer. How close together your results are to one another regardless of the actual answer.

Taking Measurements When taking measurements, you always measure one digit past what you can see. This is called the “limit of precision. ”
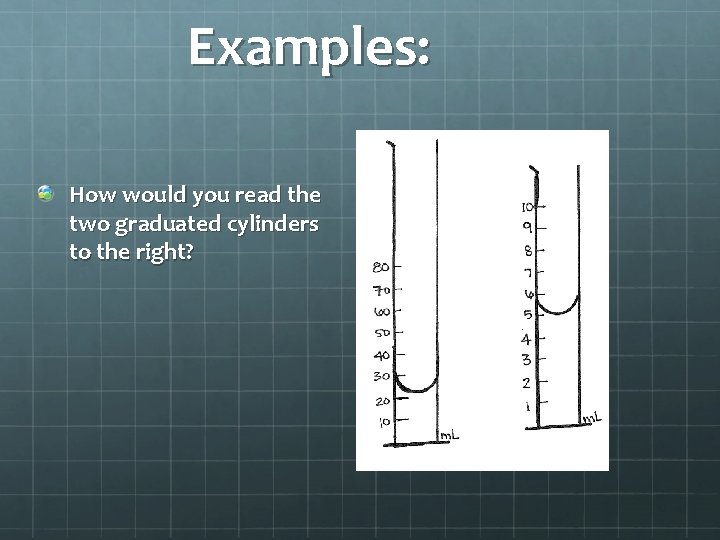
Examples: How would you read the two graduated cylinders to the right?

REMEMBER UNITS!!! All numbers must be followed by a unit Liters = L Milliliters = m. L Grams = g Naked numbers have no meaning in science! Thus will receive no credit!

Accuracy Precision and Error

Accuracy Precision and Error

b. Socrative You will now take the quizlet entitled ACCURACY & PRECISION https: //b. socrative. com/login/student/

Measurement Lab Purpose: Practice taking measurements and appropriately calculating results. Nuts and Bolts: Work with your lab partners in your group YES YOU MUST WEAR YOUR SAFETY GOGGLES Done? Answer the summary questions on the last page and prepare for your safety quiz!

Homework/Reminders/Etc. BRING A CALCULATOR TO CLASS Homework: Topic Reading Laboratory Safety Homework Due today 3. 1 Practice problems 1, 3, 3. 1 Sectiont Assessment, odds Hold for text CH 3. 2 pp 73 -79 3. 2 Section Assessment, evens Hold for text Scientific Measurement CH 3. 1 pp 62 -72 Accuracy & Precision Practice/Reinforcement HANDOUT SIG FIGS
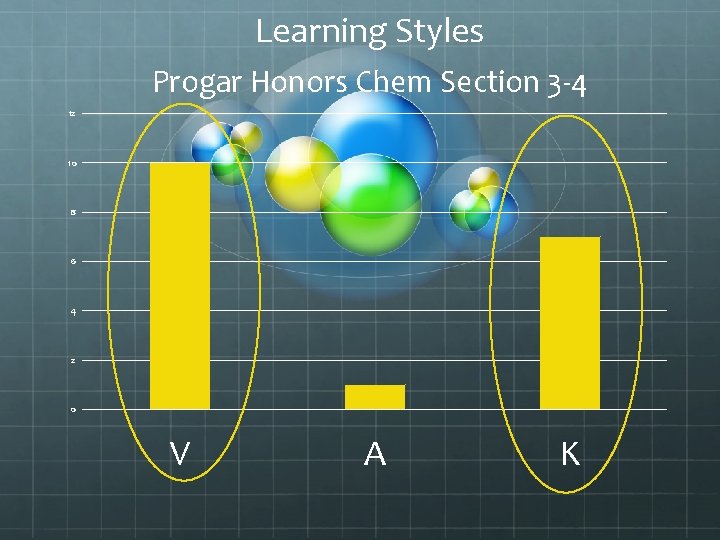
Learning Styles Progar Honors Chem Section 3 -4 12 10 8 6 4 2 0 V A K
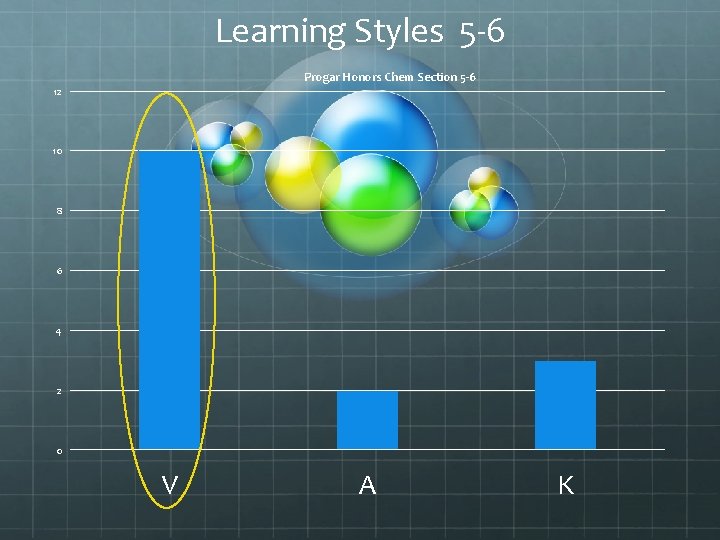
Learning Styles 5 -6 Progar Honors Chem Section 5 -6 12 10 8 6 4 2 0 V A K
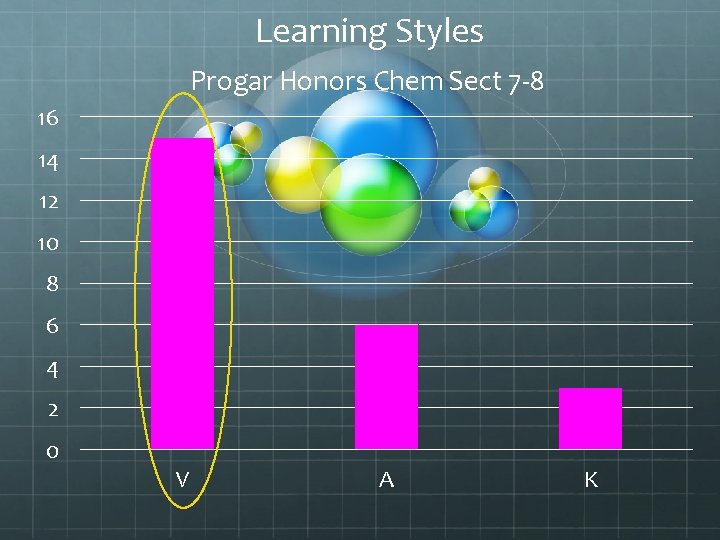
Learning Styles Progar Honors Chem Sect 7 -8 16 14 12 10 8 6 4 2 0 V A K
- Slides: 18