UNIT 1 a Syllabus Expectations and Lab Safety

UNIT 1 a. Syllabus Expectations and Lab Safety b. Scientific Measurement, Accuracy, Precision c. Scientific Notation and Sig Figs d. Calculations and Conversions

c. Scientific Notation and Significant Figures

Bell Work- Scientific Notation and Sig Figs 1. Which of the following are safety equipment found in the laboratory: a. Fire extinguisher b. Graduated Cylinder c. Erlenmeyer Flask d. Safety goggles e. Eye Wash Station 2. What are the 2 components of a Risk Assessment: 1. ______ and 2. _____ 3. Where can you find important safety information? a. NFPA diamond b. MSDS Sheets c. The purple binder in the back of the room d. Reagent Labels e. The FDA website 4. What is the cell phone policy in lab? __________________________________________________________________________ Answer these questions in your lab notebook (back half) and I will review their completeness after roll

Bell Work 1/18 - Scientific Notation, Sig Figs, Conversion How many significant figures? 1. 0. 005 5, 050 1 3 2. 0. 0008 1 3. 1, 000. 4 Convert 1. 1. 255 mm to m 2. 6. 2 L to m. L 3. 5. 75 cm to m 4. 10. 5 kg to cg 5. 2 days to sec 0. 001255 6200 0. 0575 1050000 172800 Answer these questions in your lab notebook (back half) - we will have a quiz on Friday from The bell work you completed Last week and Wed and Thurs this week

Learning Inventory Learning inventory sheet (paper)

Rounding Number adjacent to the number of places you wish to have. Is it a 5? Is it greater than 5? If yes, round the last sig fig in your number UP. If not, keep it the same.

Scientific Notation and Sig Figs Here’s the BIG Picture Topic The Good Stuff Scientific Notation What is Scientific Notation 13 https: //youtu. be/Dme-G 4 rc 6 NI Multiply and Divide sig figs 9: 30 https: //youtu. be/ior. Zdz 4 ds. BU Add and Subtract Sig figs https: //youtu. be/x. Hg. Pt. FUb. Ae. U 8: 58 International System of Units The power of 10 https: //youtu. be/v. Rj. Gar. ICal 4 Conversion Problems Dimensional Analysis 11: 23 https: //youtu. be/h. Qp. Q 0 hx. VNT g Read 3. 3 pp 80 -88 3. 8 Sect Assess, odds Reinforcement Handout Complete remainder of handout for homework Look for these: t 5: 26 Most Important Real World Evidence Homework Work on handout in class, complete remainder at home Read 3. 2 pp 73 -79, Sect Assessment evens

Scientific Notation Why Scientific Notation? Scientists sometimes have to deal in very large or very small quantities, and scientific notation is a way of expressing that number. Remember the Power of 10? The Power of 10 Video

How To: Scientific Notation Rules to Follow: A positive exponent indicates that the number is greater than 1. Example: 1, 500, 000. 0 = 1. 5 x 106 Standard Notation A negative exponent indicates that the number is less than 1. Example: 0. 000025 = 2. 5 x 10 -5 Scientific Notation: Coefficient must be between 1 and 9

What Is Scientific Notation?










Scientific Notation Practice! Convert the following to scientific notation: 1. 0. 005 2. 5, 050 5 x 10 -3 5. 05 x 103 8 x 10 -4 3. 0. 0008 1. 000 x 103 4. 1, 000. 1 x 106

Do all numbers matter? Devices for measurement have a ‘limit of precision Not all numbers in a value are “significant. ” Significant figures show the accuracy and precision of a measurement How many meniscus m. L are in this graduated cylinder?

Counting Significant Figures 4 Rules to Remember: Non-zero Numbers are ALWAYS Significant Sandwiched Zeros are ALWAYS Significant Leading Zeros are NEVER Significant Trailing Zeros are SOMETIMES Significant

NON-ZERO NUMBERS Value Sig Figs 8 mm 1 42 lbs ? 678 mm ? 9 lbs ?
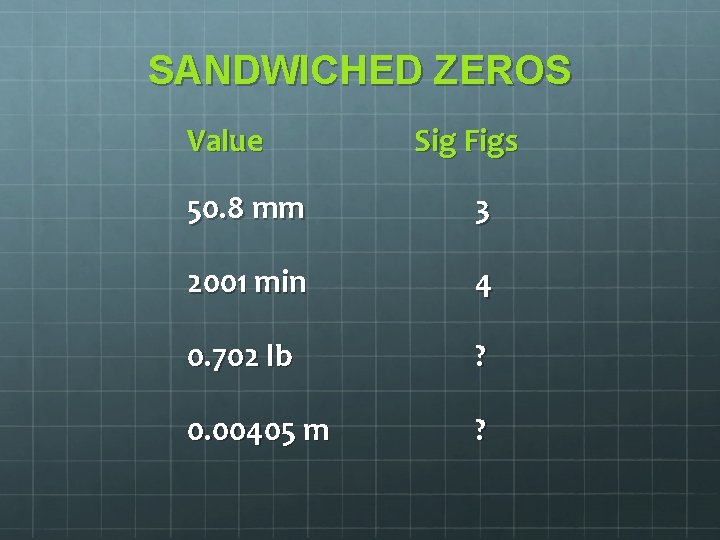
SANDWICHED ZEROS Value Sig Figs 50. 8 mm 3 2001 min 4 0. 702 lb ? 0. 00405 m ?

LEADING ZEROS Value Sig Figs 0. 008 mm 1 0. 0156 oz 3 0. 0042 lb ? 0. 000262 m. L ?

TRAILING ZEROS When a decimal is PRESENT: Trailing Zeros ARE Significant When a decimal is ABSENT: Trailing Zeros are NOT Signficiant Value Sig Figs 25, 000 in. 2 200. yr 3 48, 600 gal ? 25, 000. g ?

Calculations Using Sig Figs There are different rules for different mathematical operations: RULE: When multiplying and dividing the answer has the same number of significant digits as the value with the fewest. Example: 23. 0 cm x 432 cm x 19 cm = The calculated answer is 188, 784 cm 3 The reported answer is rounded to two sig figs because 19 cm has the least amount in the problem. The answer is 190, 000 cm 3

Calculations Using Sig Figs There are different rules for different mathematical operations: RULE: When adding and subtracting the answer is limited not by the significant digits, but the limit of precision! Example: 123. 25 m. L + 46. 0 m. L + 86. 257 m. L = The calculated answer is 255. 507 m. L The reported answer is rounded to the tenths place because 46. 0 m. L has the lowest limit of precision. The answer is 255. 5 m. L

Practice Makes Permanent! The Dice Game

d. Calculations: Units and Dimensional Analysis
![The Metric System Base Units [BU] Liters Meters Grams Multiples of 10 1 Kilo The Metric System Base Units [BU] Liters Meters Grams Multiples of 10 1 Kilo](http://slidetodoc.com/presentation_image_h2/0b8261d967a6c6a033943ff738ce12a3/image-31.jpg)
The Metric System Base Units [BU] Liters Meters Grams Multiples of 10 1 Kilo = 1000 [BU] 1 [BU] = 100 centi = 1000 milli =1, 000 micro = 1, 000, 000 nano

Units of Measurement Ø SI Units Ø What are they? Ø Why are they important? Ø SI Base Units for the Quantities of length, mass, (volume), time, temperature, and amount of substance (counted number).

Quantity SI Base Unit Abbreviation Length meter m Mass kilogram kg Volume Liter L Time second s Temperature Kelvin K Amount mole mol

What is a Conversion Factor? Ø A conversion factor is a comparison of two different units that allows us to convert from one unit to the other. Ø Conversion factors may be arranged differently. 1 meter = 100 centimeters -OR 1 meter 100 centimeters

Why use a conversion factor? Your units are clearly indicated. Reduces confusion on whether to multiply or divide. Prepares you to complete more complex problems.

Metric Conversion Base Units Ø The metric conversion base units are almost the same as the SI base units with the exception of mass. Ø Mass = gram = g Ø Volume = liter = L Ø Length = meter = m

Prefixes That Make Conversion Base Units LARGER or smaller kilo- Base Unit deci- centi- millik m, g, L Makes the base unit LARGER d c m Make the base unit smaller
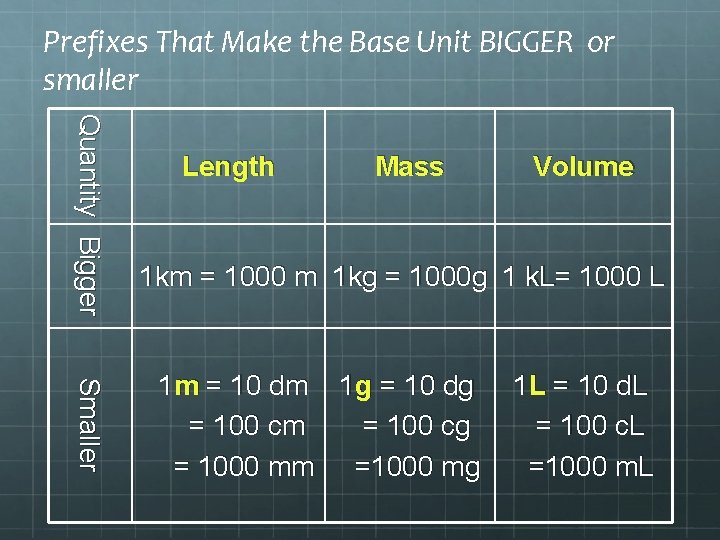
Prefixes That Make the Base Unit BIGGER or smaller Quantity Bigger Length Mass Volume 1 km = 1000 m 1 kg = 1000 g 1 k. L= 1000 L Smaller 1 m = 10 dm 1 g = 10 dg = 100 cm = 100 cg = 1000 mm =1000 mg 1 L = 10 d. L = 100 c. L =1000 m. L

How many minutes are in 2. 5 hours? What do you know about the problem? 2. 5 hours 1 hour = 60 minutes 2. 5 hours X 60 minutes 1 hour Units must cancel! = 150 minutes Sig Figs: Conversion factors are defined numbers

Practice Problem You have $7. 25 in your pocket in quarters. How many quarters do you have? Identify what you know You have $7. 25 $1 = 4 quarters $7. 25 X 4 quarters $1 = 29. 0 quarters

Practice Problem 3 How many days old are you on your 16 th birthday? Identify what you know 16 years 1 year = 365 days 16 yrs X 365 days 1 year = 5840 days Significant Figures? 5800 days

Practice Problem 4 How many seconds are in 1. 4000 days? What do you know? 1. 4000 days 1 day = 24 hours 1 hour = 60 minutes 1 minute = 60 seconds 1. 400 days x 24 hr 1 day x ? ?

Practice Problem 4 1. 4000 days x 24 hours 1 day x 60 minutes 1 hour 60 sec x = 1 minute What will your answer be? 120960 sec

Practice Problem 4 What is wrong with the following setup? 1. 4 day x 1 day 24 hr x 60 min x 60 sec 1 hr 1 min

Practice Problem 5 An adult human has 4. 65 L of blood. How many gallons of blood is that? What do you know: 1 quart = 0. 946 L 1 gallon = 4 quarts 4. 65 L x 1 qt 0. 946 L x 1 gal 4 qt = 1. 23 gal

Your Turn! Independent or with small group of 2 -3 people.
![[INSERT VIDEO HERE] [INSERT VIDEO HERE]](http://slidetodoc.com/presentation_image_h2/0b8261d967a6c6a033943ff738ce12a3/image-47.jpg)
[INSERT VIDEO HERE]

Ø When converting from one metric unit to another, if the known unit or the unknown unit is not a metric base unit (g, m, L), you must first convert the known to the base unit and then to the unknown unit. This will require 2 conversion factors instead of 1. 5. 5 km to mm. 5. 5 km →m → mm

Ø Use the 3 step process to make the following conversions: 1. 2. 7 m to km 2. 35 mm to cm 3. 62. 5 g to mg 4. 5. 50 mol S to g S 5. 500. k. Pa to atm






Practice: Do the Following Conversions 1. 255 mm to m 2. 6. 2 L to m. L 3. 5. 75 cm to m 4. 10. 5 kg to cg 5. 1050 k. Pa to mm Hg

Practice Problem 6 How many centigrams are in 605 grams? What do you know? 605 grams 1 gram = 100 centigrams 605 grams x 100 centigrams 1 gram = 60500 centigrams

Practice Problem 7 How many meters are in 14 nanometers? What do you know? 14 nanometers 1 meter = 1, 000, 000 nanometers or 109 nm 14 nm x 1 m = 1, 000, 000 nm 1. 4 x 10 -8 m Or 0. 000000014 m

Practice Problem 8 How many meters are in 42. 0 feet? What do you know? 42. 0 feet 1 foot = 12 inches 1 inch = 2. 54 cm 100 cm = 1 m 42. 0 feet x 12 inches 1 foot x 2. 54 cm 1 inch x 1 meter 100 cm = 12. 8 m

Squares and Cubes Use the conversion factors you already know, but when you square or cube the unit, don’t forget to cube the number also! Best way: Square or cube the ENTIRE conversion factor Example: Convert 4. 3 cm 3 to mm 3 4. 3 cm 3 ( ) 10 mm 1 cm 3 or 4. 3 cm 3 = 4300 mm 3 103 mm 3 13 cm 3

Learning Check A Nalgene water bottle holds 1000 cm 3 of dihydrogen monoxide (DHMO). How many cubic decimeters is that? 1 dm = 10 cm

Cubic Practice 1000 cm 3 ( or 1000 1 dm 10 cm 3 1 dm cm 3 1000 3 10 3 cm 3 or cm 3 ) 3 1 dm 3 1, 000 cm 3 = 1 dm 3

Cubic - Example How many cubic meters are in 50. cubic centimeters? What do you know? 50. cm 3 100 3 cm 3 = 1 3 m 3 50. cm 3 3 1 m 3 3 3 100 cm = 5. 0 x 10 -5 m 3

Practice Time! Work with the people around you- this means supporting your lab mates if you understand this! Resources: http: //www. youtube. com/watch? v=IIQPHC 5 g. ZT 8 (7: 58) http: //www. youtube. com/watch? v=PYTp 75 sry. WA (7: 12) http: //www. youtube. com/watch? v=ci. FOlirz 4 Js (5: 30) http: //www. youtube. com/watch? v=7 b 60 RZqut 0 U (7: 37) http: //www. youtube. com/watch? v=2 e. XC 6 s 9 X 6 Wc (4: 43) http: //www. youtube. com/watch? v=o 5 Bmo. MDJLRY (4: 31)
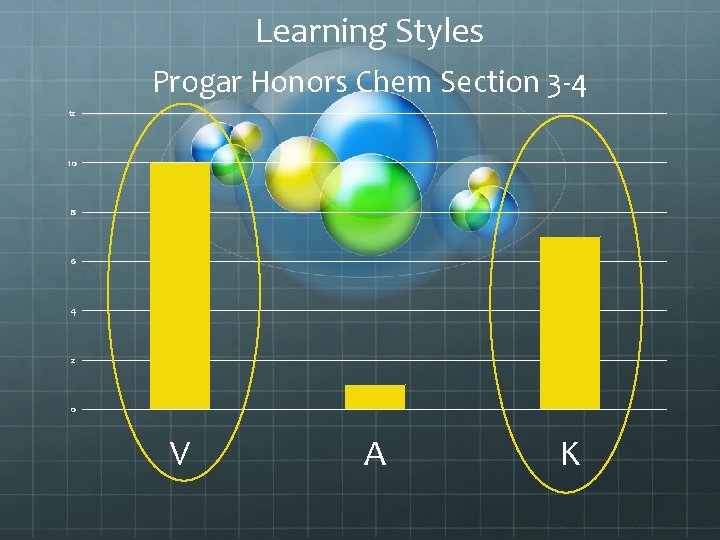
Learning Styles Progar Honors Chem Section 3 -4 12 10 8 6 4 2 0 V A K
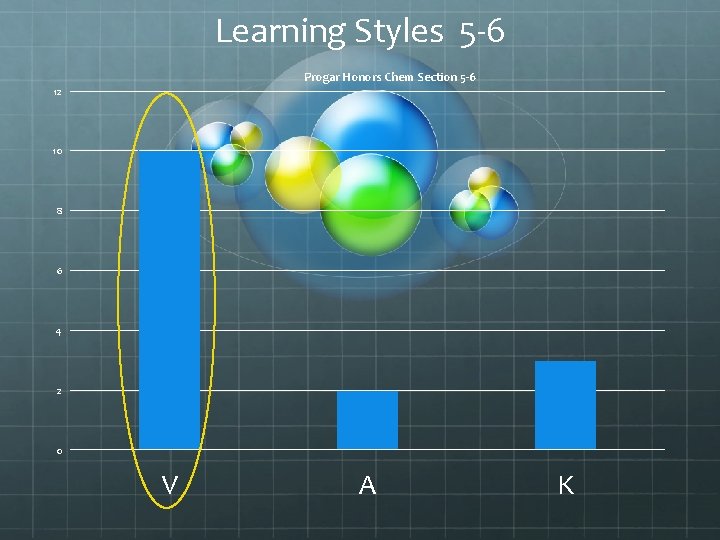
Learning Styles 5 -6 Progar Honors Chem Section 5 -6 12 10 8 6 4 2 0 V A K
Learning Styles Progar Honors Chem Sect 7 -8 16 14 12 10 8 6 4 2 0 V A K

Homework/Reminders/Etc. BRING A CALCULATOR TO CLASS Homework:
- Slides: 67