Understanding United States Pharmacopeia USP compounding standards and










![JC FAQs • USP <797> interpretations Jointcommission. org. (2018). Standards Interpretation FAQs. [online] Available JC FAQs • USP <797> interpretations Jointcommission. org. (2018). Standards Interpretation FAQs. [online] Available](https://slidetodoc.com/presentation_image_h2/12a3cd078b93287b8edcd8bf798bcaf4/image-12.jpg)






























- Slides: 45

Understanding United States Pharmacopeia (USP) compounding standards and applying to daily clinical practice Bridget Gegorski, Pharm. D, MS Medication Safety Officer University Hospitals October 12 th, 2018

Objectives • Define USP <797> and <800> standards • Identify hospital practices and recognize necessary changes for USP compliance • Explain how USP <797> and <800> general chapter changes will affect practices in the pharmacy and on the practice units • Recognize applicability to your own area of practice 2

Why is this topic important to clinical pharmacists? • Sterile compounding regulations and enforcement have changed • Proposed changes to bedside compounding requirements and definitions have proposed changes • Changes to how hazardous drugs are defined • Definition of hazardous drug amended and handling requirements increased 3

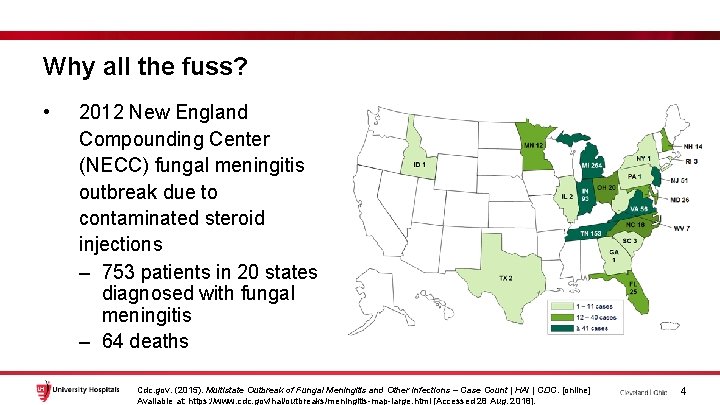
Why all the fuss? • 2012 New England Compounding Center (NECC) fungal meningitis outbreak due to contaminated steroid injections – 753 patients in 20 states diagnosed with fungal meningitis – 64 deaths Cdc. gov. (2015). Multistate Outbreak of Fungal Meningitis and Other Infections – Case Count | HAI | CDC. [online] Available at: https: //www. cdc. gov/hai/outbreaks/meningitis-map-large. html [Accessed 28 Aug. 2018]. 4

Tragedy sparks reform • US Department of Justice named the NECC fungal meningitis outbreak largest US public health crisis caused by a pharmaceutical product in recent history • 2013 Drug Quality and Security Act established 503(b) compounding pharmacies – Establishes FDA direct oversight of these pharmacy types • Increased oversight for all compounding practices from regulatory bodies Justice. gov. (2017). Owner of New England Compounding Center Sentenced for Racketeering Leading to Nationwide Fungal Meningitis Outbreak. [online] Available at: https: //www. justice. gov/opa/pr/owner-new-england-compounding-center-sentenced-racketeering-leading-nationwide-fungal [Accessed 28 Aug. 2018]. Congress. gov. (2013). H. R. 3204 - 113 th Congress (2013 -2014): Drug Quality and Security Act. [online] Available at: https: //www. congress. gov/bill/113 th-congress/house-bill/3204 5

United States Pharmacopeia (USP) • Not a government entity • Develops and publishes standards for drug substances, drug products, excipients, and dietary supplements in the United States Pharmacopeia–National Formulary (USP–NF) – USP general chapters are legally recognized in the Federal Food, Drug and Cosmetic (FD&C) Act since it was first enacted in 1938 • No role in enforcement Usp. org. (2018). Legal Recognition of USP Standards | USP. [online] Available at: http: //www. usp. org/about/legalrecognition [Accessed 28 Aug. 2018]. 6

USP <797> Pharmaceutical compounding sterile preparations 7

USP <797> Pharmaceutical compounding sterile preparations • Standard written with intent to prevent patient harm – Microbial contamination – Excessive bacterial endotoxins – Variability in intended strength that exceed monograph limits – Use of ingredients of inappropriate quality – Unintended physical or chemical contaminants • Establishes minimum practice and quality standards for compounded sterile preparations (CSPs) <797> Pharmaceutical compounding sterile preparations. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 39 -84. 8

USP <797> timeline Published 11/2003 Official 1/2004 Revision released 12/2007 Revision official 6/2008 2 nd • USP received >10, 000 public comments • Public comment period open until 11/30/2018 revision released 9/2015 revision re -release 7/2018 2 nd revision intended official date 12/2019 9

USP <797> standard enforcement • FDA and other government authorities – Federal Food, Drug, and Cosmetics Act – Insanitary conditions draft guidance (September 2018) • State Boards of Pharmacy – Ohio Administrative Code 4729 -16 -03 • Centers for Medicare and Medicaid (CMS) – Added as part of the Conditions of Participation in 2015 Insanitary Conditions at Compounding Facilities Guidance for Industry [internet]. U. S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research Office of Compliance. [Revision Sept. 2018, cited 2018 Sept 27], Available from www. fda. gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm 514666. pdf. 4729 -16 -03 Drugs compounded in a pharmacy. Ohio Laws and Rules. [Effective 2016 Feb 15; cited 2018 Jan 16], Available from http: //codes. ohio. gov/oac/4729 -16 -03 10

USP <797> standard enforcement • Joint Commission (JC) – Statement issued October 2017 • “While activities associated with sterile compounding are currently assessed during JC surveys, accredited organizations performing sterile compounding should now expect increased attention on these processes. ” Reducing Risk Associated with Sterile Medication Compounding. (2017). The Joint Commission Perspectives, 37(10), pp. 12 -13. 11
![JC FAQs USP 797 interpretations Jointcommission org 2018 Standards Interpretation FAQs online Available JC FAQs • USP <797> interpretations Jointcommission. org. (2018). Standards Interpretation FAQs. [online] Available](https://slidetodoc.com/presentation_image_h2/12a3cd078b93287b8edcd8bf798bcaf4/image-12.jpg)
JC FAQs • USP <797> interpretations Jointcommission. org. (2018). Standards Interpretation FAQs. [online] Available at: https: //www. jointcommission. org/standards_information/jcfaq. aspx [Accessed 21 Aug. 2018]. 12

Joint Commission survey experiences • IV hoods/rooms • Compounding level – low, medium, or high risk • Paperwork review – Equipment certifications – Environmental viable sampling (air and surface) – Competency documentations 13

USP <800> Hazardous drugs – Handling in health care settings 14

USP <800> Hazardous drugs – Handling in health care settings • Standard written with intent to prevent unnecessary employee exposure to hazardous drugs (HDs) • Official December 1, 2019 • Establishes practice and quality standards for the handling of HDs throughout the medication use life cycle Receive Transport Store Mix Administer <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. Dispose 15

Occupational HD exposure • Healthcare workers exposed to HDs by absorption through skin, mucous membranes, inadvertent ingestion, inhalation, or injection • Studies have detected levels of HDs in the urine of pharmacists and nurses • Oncology Nursing Society (ONS) cites extensive research on the subject Polovich, M. and Olsen, M. Safe handling of hazardous drugs. 3 rd ed. Pittsburgh: Oncology Nursing Society. 2018; pages 8 -17. 16

NIOSH List of HDs The National Institute for Occupational Safety and Health (NIOSH) • Table 1: Antineoplastics • Table 2: Non-antineoplastics • Table 3: Reproductive-only hazards Cdc. gov. (2016). NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings, 2016. [online] Available at: https: //www. cdc. gov/niosh/docs/2016 -161/pdfs/2016 -161. pdf? id=10. 26616/NIOSHPUB 2016161 [Accessed 28 Aug. 2018]. 17

Personnel to consider • • Receiving Transport Pharmacy technicians Pharmacists Nursing Procedural personnel – Surgical services – Emergency department – Obstetrics Physicians Medical office personnel Kienle PC et al. Perform an assessment of risk to comply with USP <800>[Internet]. Pharmacy Purchasing & Products. 2017; 14: 34. [Cited 2017 May 8]. Available from: https: //www. pppmag. com/article/2012. 18

CDC and NIOSH recommended controls Cdc. gov. (2018). CDC - Hierarchy of Controls - NIOSH Workplace Safety and Health Topic. [online] Available at: https: //www. cdc. gov/niosh/topics/hierarchy/ [Accessed 28 Aug. 2018]. 19

USP <800> HD list and HD handling • Entity specific HD list is required • Handling Option 1 – Use the NIOSH list in its entirety and handle every drug in every dosage form with all the containment strategies and work practices listed in USP <800> – Not practical for workflow and not always possible with infrastructure • Handling Option 2 – Perform an assessment of risk (Ao. R) on NIOSH listed drugs to determine alternative containment strategies for some drugs/dosage forms – Drugs that qualify include oral chemotherapy and NIOSH tables 2 and 3 drugs Kienle PC et al. Perform an assessment of risk to comply with USP <800>[Internet]. Pharmacy Purchasing & Products. 2017; 14: 34. [Cited 2017 May 8]. Available from: https: //www. pppmag. com/article/2012. 20

USP <800> requirements • Creation of a HD list – JC requirement MM 01. 03 • Training, competency, and documentation needs for staff – HD handling, PPE use, and spill control • HD communication program – A signed employee statement acknowledging that they have been informed about hazardous drug exposure risks <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. 21

USP <800> requirements • Describes acceptable facility designs and environmental controls • Necessitates designated areas for receipt, storage, and nonsterile and sterile compounding for HD use only • Defines decontamination/deactivation frequency and product requirements • Requires closed system transfer devices (CSTDs) for administration of HDs <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. 22

Components of an assessment of risk (Ao. R) • Type of HD • Specific dosage form • Risk of exposure • Packaging • Type of manipulation performed • Alternative containment strategy to reduce the risk of exposure Kienle PC et al. Perform an assessment of risk to comply with USP <800>[Internet]. Pharmacy Purchasing & Products. 2017; 14: 34. [Cited 2017 May 8]. Available from: https: //www. pppmag. com/article/2012. 23

USP <800> receipt and storage facility requirements • Handled under conditions that promote patient safety, worker safety, and environmental protection • Received in an area that is negative or neutral pressure to adjacent areas • HD storage: – Externally vented – Physically separate – Negative pressure – Minimum 12 air changes per hour (ACPH) <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. 24

USP <800> Engineering controls for sterile HD compounding and storage Configuration IV hood requirement IV room requirements Cleanroom suite: ISO Class 7 buffer room with an ISO Class 7 anteroom Externally vented • Example biological safety cabinet (BSC) or contained aseptic compounding isolator (CACI) • • • Externally vented 30 ACPH Negative pressure 0. 010. 03 inches or water column relative to adjacent areas Unclassified: Containment segregated compounding area (CSCA) Externally vented • Example BSC or CACI • • • Externally vented 12 ACPH Negative pressure 0. 010. 03 inches or water column relative to adjacent areas <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. 25

HD administration • Closed system transfer device (CSTD) www. BD. com www. ICUmedical. com www. Equashield. com <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. 26

HD administration • Don PPE according to NIOSH Table 5 recommendations • Gloves must be worn for handling all HDs • American Society of Testing and Materials (ASTM) requirements • D 6978 (or its successor) • “Chemotherapy gloves” • Optional PPE based on dosage form: • Impenetrable gown • Eye/face protection • Respiratory protection 27

HD administration • All doses and storage areas labeled denoting HD • Clean area with a decontamination/deactivation agent • Spill kits and spill response planning 28

Examples of NIOSH listed drugs • Table 2 (non-antineoplastic HD): – Azathioprine – Chloramphenicol – Cidofovir – Conjugated Estrogens – Cyclosporine – Dexrazoxane – Fosphenytoin – Ganciclovir – Mycophenolate mofetil – Palifermin – Phenytoin – Progesterone – Tacrolimus – Zidovudine • Table 3 (reproductive risk only): – Cetrorelix – Choriogonadotropin – Fluconazole – Icatibant – Mentropins – Methylgonovine – Pamidronate – Plerixafor – Telavancin – Testosterone – Valproic Acid – Voriconazole – Ziprasidone – Zoledronic acid Cdc. gov. (2016). NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings, 2016. [online] Available at: https: //www. cdc. gov/niosh/docs/2016 -161/pdfs/2016 -161. pdf? id=10. 26616/NIOSHPUB 2016161 [Accessed 28 Aug. 2018]. 29

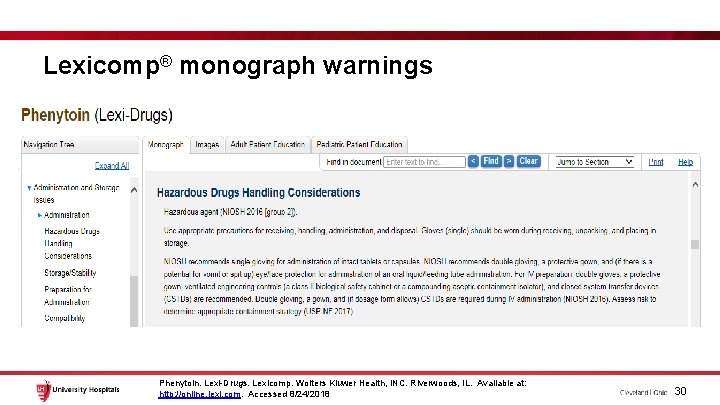
Lexicomp® monograph warnings Phenytoin. Lexi-Drugs. Lexicomp. Wolters Kluwer Health, INC. Riverwoods, IL. Available at: http: //online. lexi. com. Accessed 8/24/2018 30

USP <797> Revised 31

USP <797> revision 7/29/18 11/30/19 6/1/19 12/1/19 • Draft standard published • Public comment period closes • Official publication • Standard will be enforceable Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 32

Biggest changes • Definition of compounding vs administration • Defined restrictions for immediate use compounding • Low, Medium, High Risk compounding categories are replaced with Category 1 and 2 compounds that are based on IV preparation location design • New competency requirements and frequencies • Increased frequency for environmental sampling required Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 33

CSPs in scope • Injections, including infusions • Irrigations for internal body cavities – Mouth, rectal cavity, and sinus cavity are excluded • Ophthalmic dosage forms • Preparations for pulmonary inhalations • Baths and soaks for live organs and tissues • Implants Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 34

USP <797> revisions • Compounding definition: • “Combining, admixing, diluting, pooling, reconstituting, repackaging, or otherwise altering a drug or bulk drug substance to create a sterile medication” • Exclusions • “Preparing a conventionally manufactured sterile product in accordance with the directions contained in approved labeling provided by the product’s manufacturer is not compounding as long as the product is prepared for an individual patient” • Withdrawal of doses from a vial is considered part of administration Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 35

Proprietary bag and vial systems • If used in accordance with manufacturer’s instructions for immediate administration to a patient, it is not considered compounding – Must be attached using aseptic technique http: //www. baxtermedicationdeliveryproducts. com/drugdelivery/minibag. html Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 36

Proprietary bag and vial systems • Docking of bag and vial for future activations must be performed in accordance to USP <797> – ISO 5 environment (IV hood) – Beyond use date (BUD) exception: BUD for proprietary bag and vials systems must not be longer than those specified in the manufacturer’s labeling 37

Immediate use CSP requirements • • Single patient dose Non-hazardous preparation Use only sterile products Administration must begin within 1 hour of the start of preparation Single dose vials discarded after preparation complete Aseptic technique Follow evidenced based information for physical and chemical compatibilities Procedures in place to prevent contact with nonsterile surface, particulate matter, biological fluids, and mix ups with other CSPs Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 38

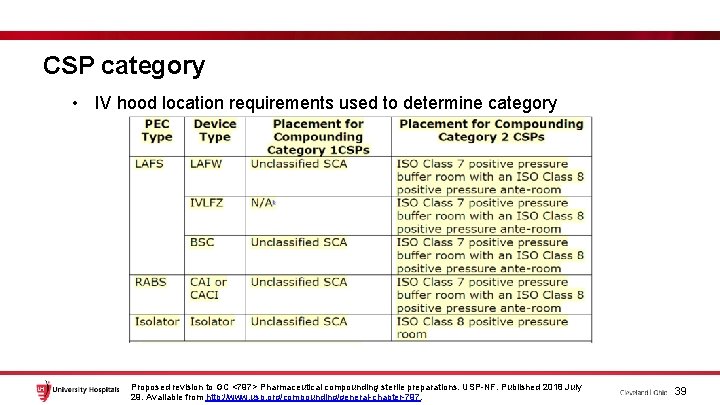
CSP category • IV hood location requirements used to determine category Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 39

IV hood and room acronyms • ISO- international organization for standardization • LAFS- Laminar airflow system – LAFW- Laminar airflow workbench – IVLFZ- Integrated vertical laminar flow zone – BSC- Biological Safety Cabinet • RABS- Restricted access barrier system – CAI- Compounding aseptic isolator – CACI- Containment aseptic compounding isolator Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 40

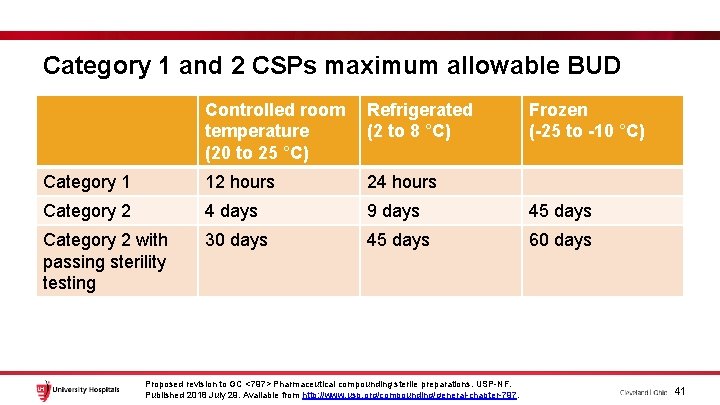
Category 1 and 2 CSPs maximum allowable BUD Controlled room temperature (20 to 25 °C) Refrigerated (2 to 8 °C) Category 1 12 hours 24 hours Category 2 4 days 9 days 45 days Category 2 with passing sterility testing 30 days 45 days 60 days Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. Frozen (-25 to -10 °C) 41

BUD vs expiration date Term Definition Applicability BUD • Either the date, or hour and date after which a CSP must not be used or administration must not begin The BUD is determined from the date/time that preparation of the CSP is initiated Applies to all CSPs Time during which a product can be expected to meet the requirements of the compendial monograph, if one exists, provided it is kept under the prescribed conditions Applies to all conventionally manufactured products, APIs, and excipients • Expiration Date • Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/general-chapter-797. 42

Summary • USP <800> will require increased precautions for medication storage, preparation, administration, and disposal on the floors • Ao. Rs must be completed to avoid requiring all NIOSH listed drugs to be treated the same as a chemotherapy product • USP <797> revision will expand the availability of the immediate use exemption for compounding on the floor • BUDs of medications at your facility may change based on pharmacy IV room design 43

Questions? 44

References • Cdc. gov. (2015). Multistate Outbreak of Fungal Meningitis and Other Infections – Case Count | HAI | CDC. [online] Available at: https: //www. cdc. gov/hai/outbreaks/meningitismap-large. html [Accessed 28 Aug. 2018 ]. • Justice. gov. (2017). Owner of New England Compounding Center Sentenced for Racketeering Leading to Nationwide Fungal Meningitis Outbreak. [online] Available at: https: //www. justice. gov/opa/pr/owner-new-england-compounding-center-sentenced-racketeering-leading-nationwide-fungal [Accessed 28 Aug. 2018]. • Congress. gov. (2013). H. R. 3204 - 113 th Congress (2013 -2014): Drug Quality and Security Act. [online] Available at: https: //www. congress. gov/bill/113 th-congress/housebill/3204 [Accessed 28 Aug. 2018 ]. • Usp. org. (2018). Legal Recognition of USP Standards | USP. [online] Available at: http: //www. usp. org/about/legal-recognition [Accessed 28 Aug. 2018]. • <797> Pharmaceutical compounding sterile preparations. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 3984. • Insanitary Conditions at Compounding Facilities Guidance for Industry [internet]. U. S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research Office of Compliance. [Revision Sept. 2018, cited 2018 Sept 27], Available from www. fda. gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm 514666. pdf. • 4729 -16 -03 Drugs compounded in a pharmacy. Ohio Laws and Rules. [Effective 2016 Feb 15; cited 2018 Jan 16], Available from http: //codes. ohio. gov/oac/4729 -16 -03 • Reducing Risk Associated with Sterile Medication Compounding. (2017). The Joint Commission Perspectives, 37(10), pp. 12 -13. • Jointcommission. org. (2018). Standards Interpretation FAQs. [online] Available at: https: //www. jointcommission. org/standards_information/jcfaq. aspx [Accessed 21 Aug. 2018]. • <800> Hazardous drugs – Handling in health care settings. Found in: USP Compounding Compendium. The United States Pharmacopeial Convention. Rockville, MD. c 2016: 85 -103. • Polovich, M. and Olsen, M. Safe handling of hazardous drugs. 3 rd ed. Pittsburgh: Oncology Nursing Society. 2018; pages 8 -17. • Cdc. gov. (2016). NIOSH List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings, 2016. [online] Available at: https: //www. cdc. gov/niosh/docs/2016161/pdfs/2016 -161. pdf? id=10. 26616/NIOSHPUB 2016161 [Accessed 28 Aug. 2018]. • Kienle PC et al. Perform an assessment of risk to comply with USP <800>[Internet]. Pharmacy Purchasing & Products. 2017; 14: 34. [Cited 2017 May 8]. Available from: https: //www. pppmag. com/article/2012. • Cdc. gov. (2018). CDC - Hierarchy of Controls - NIOSH Workplace Safety and Health Topic. [online] Available at: https: //www. cdc. gov/niosh/topics/hierarchy / [Accessed 28 Aug. 2018]. • Proposed revision to GC <797> Pharmaceutical compounding sterile preparations. USP-NF. Published 2018 July 29. Available from http: //www. usp. org/compounding/generalchapter-797. 45