Understanding the Periodic Table PERIODS The periodic table

Understanding the Periodic Table: PERIODS The periodic table can help you use your imagination to visualize the nano-world of atomic structure.

Look at your periodic table How many rows?
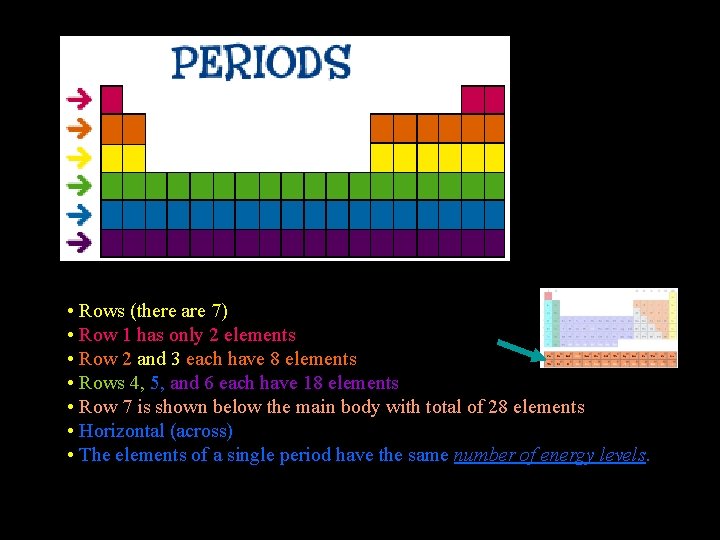
• Rows (there are 7) • Row 1 has only 2 elements • Row 2 and 3 each have 8 elements • Rows 4, 5, and 6 each have 18 elements • Row 7 is shown below the main body with total of 28 elements • Horizontal (across) • The elements of a single period have the same number of energy levels.
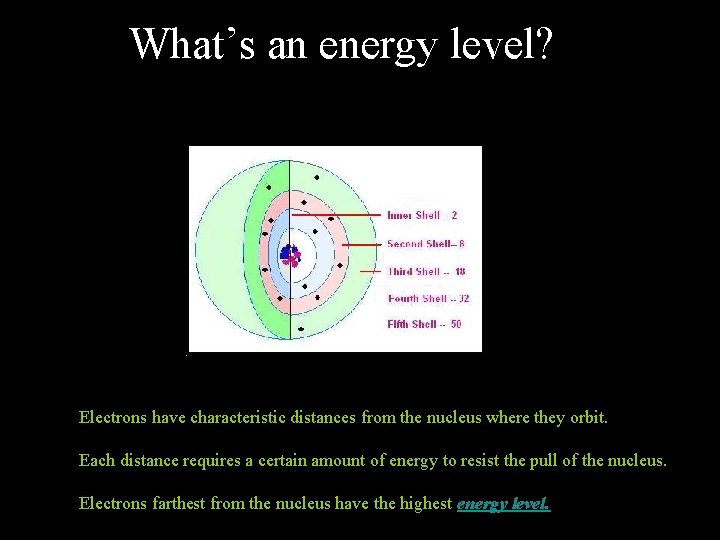
What’s an energy level? Electrons have characteristic distances from the nucleus where they orbit. Each distance requires a certain amount of energy to resist the pull of the nucleus. Electrons farthest from the nucleus have the highest energy level.
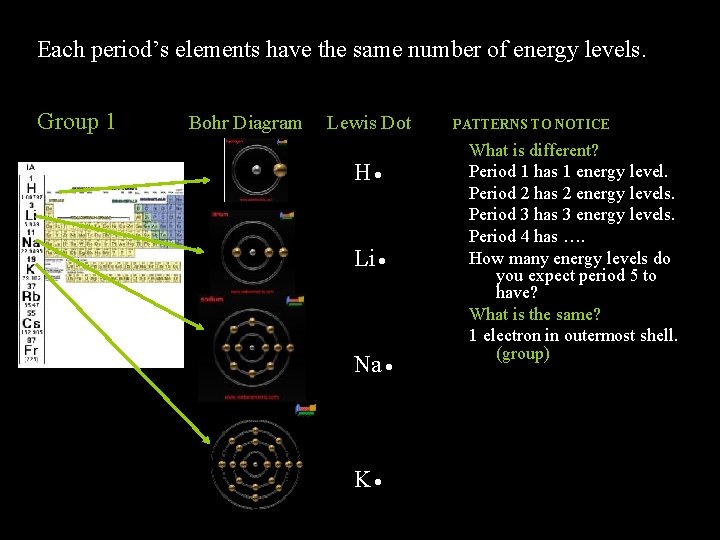
Each period’s elements have the same number of energy levels. Group 1 Bohr Diagram Lewis Dot H Li Na K PATTERNS TO NOTICE What is different? Period 1 has 1 energy level. Period 2 has 2 energy levels. Period 3 has 3 energy levels. Period 4 has …. How many energy levels do you expect period 5 to have? What is the same? 1 electron in outermost shell. (group)
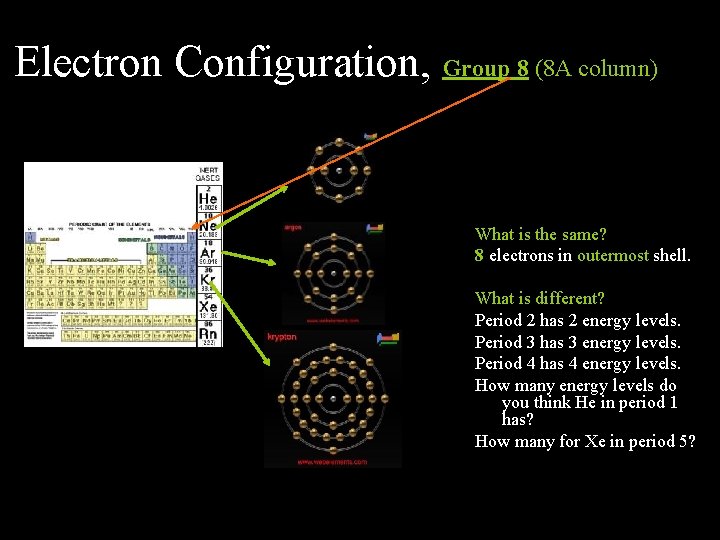
Electron Configuration, Group 8 (8 A column) What is the same? 8 electrons in outermost shell. What is different? Period 2 has 2 energy levels. Period 3 has 3 energy levels. Period 4 has 4 energy levels. How many energy levels do you think He in period 1 has? How many for Xe in period 5?
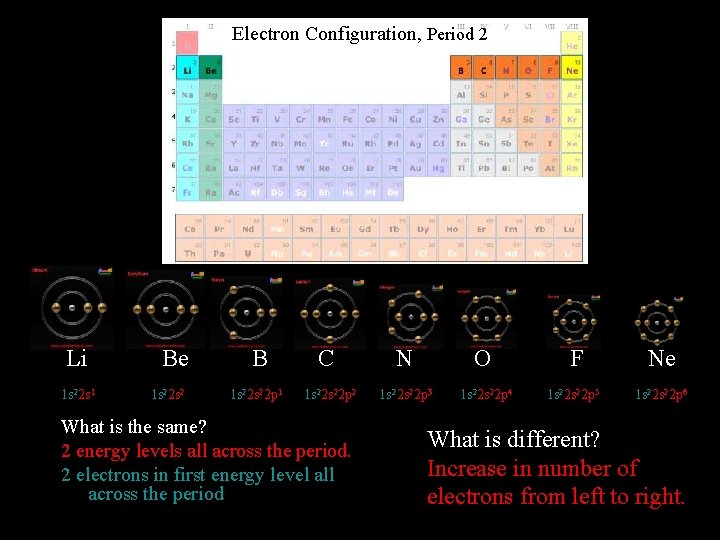
Electron Configuration, Period 2 Li 1 s 22 s 1 Be 1 s 22 s 2 B 1 s 22 p 1 C 1 s 22 p 2 What is the same? 2 energy levels all across the period. 2 electrons in first energy level all across the period N O F Ne 1 s 22 p 3 1 s 22 p 4 1 s 22 p 5 1 s 22 p 6 What is different? Increase in number of electrons from left to right.

Atomic Radius • As the period increases, the size of the electron cloud increases • Looking across a period, the size of the electron cloud decreases. • Looking across a period, each next atom has one additional proton which pulls the electrons in tighter. 1 2 3 4 5 6
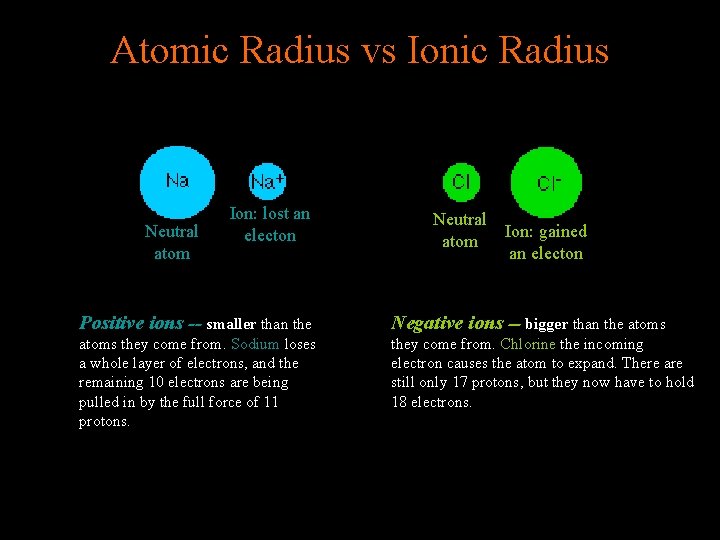
Atomic Radius vs Ionic Radius Neutral atom Ion: lost an electon Neutral atom Ion: gained an electon Positive ions -- smaller than the Negative ions -- bigger than the atoms they come from. Sodium loses a whole layer of electrons, and the remaining 10 electrons are being pulled in by the full force of 11 protons. they come from. Chlorine the incoming electron causes the atom to expand. There are still only 17 protons, but they now have to hold 18 electrons.

Ionization Energy the amount of energy it takes to detach one electron from a neutral atom. (Electrostatic attraction is the attraction of opposite charges. )
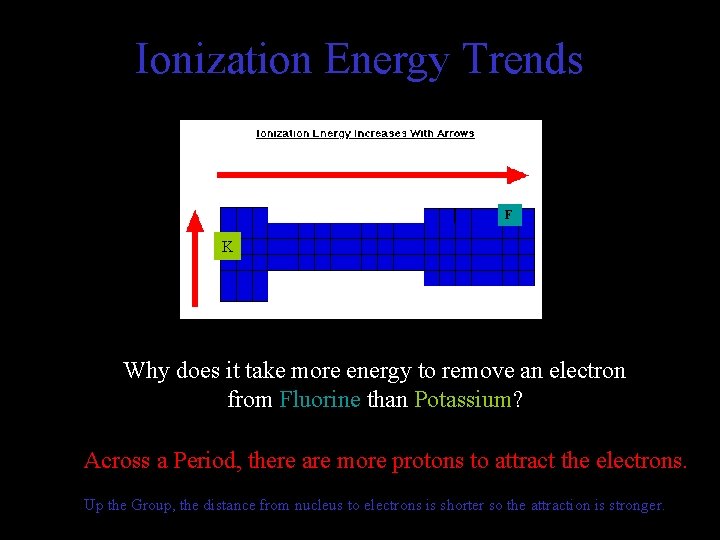
Ionization Energy Trends F K Why does it take more energy to remove an electron from Fluorine than Potassium? Across a Period, there are more protons to attract the electrons. Up the Group, the distance from nucleus to electrons is shorter so the attraction is stronger.

Electronegativity measures the tendency of an atom to attract a bonding pair of electrons. The Pauling scale is the most commonly used. Fluorine (the most electronegative element) is assigned a value of 4. 0, and values range down to caesium and francium which are the least electronegative at 0. 7.

Electronegativity No electronegativity difference between two atoms leads to a pure non-polar covalent bond. Electrons are shared evenly. A small electronegativity difference leads to a polar covalent bond. Electrons are drawn towards the stronger atom. A large electronegativity difference leads to an ionic bond. Electrons are drawn to the stronger atom.

Test Yourself 1. Which pairs of atoms will have electrostatic attraction? A C Carbon B Silicon Oxygen D Helium Sodium Chlorine ANSWER: D Sodium has 1 electron to give, Chlorine has a strong pull to gain 1 electron. Becoming ions, they attract each other.

Test Yourself 2. Which pairs of elements are in the same period? A C Boron Carbon B Silicon Oxygen D Helium Neon Hydrogen Chlorine ANSWER: A Elements in the same period have the same number of energy levels.

Test Yourself 3. Which atom would require the least energy to detach an electron from? A C Carbon (C) B Potassium (K) D Neon (Ne) Chlorine (Cl) ANSWER: C One single electron in the outermost shell is easiest to remove. Potassium has the lowest ionization energy of all the elements shown.

Test Yourself 4. What happens to the ionization energy of elements when moving from the bottom to the top of a group in the periodic table? A Ionization energy changes randomly B Ionization energy remains unchanged C Ionization energy decreases D Ionization energy increases ANSWER: D Atoms at the top of the periodic table have fewer electrons, therefore fewer energy levels. The closer the electrons are to the nucleus, the stronger the pull, and therefore the more energy required to escape the pull.
- Slides: 17