Understanding the Periodic Table Key Questions What does

Understanding the Periodic Table Key Questions: What does atomic structure have to do with the periodic table?
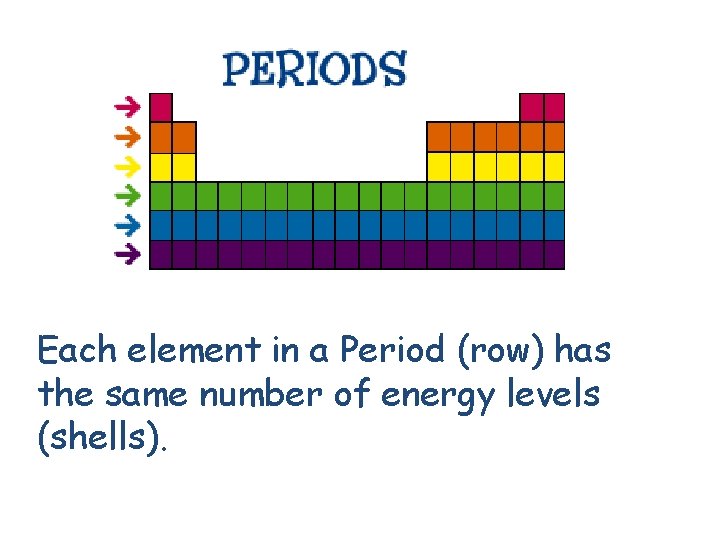
Each element in a Period (row) has the same number of energy levels (shells).


Groups • Each Group (column) of elements has the same number of valence electrons (electrons in the outer shell).

Families • Elements are divided up into families based on their chemical properties and how they react with the outside world.

What are Valence Electrons? Lithium atom One valence electron Helium atom Two valence electrons

What does atomic structure have to do with the periodic table? • Elements are arranged in the periodic table based on both their chemical and physical properties.

Energy Levels, Shells and Orbitals Energy Level Shell Letter (Principal Quantum Number) Maximum Electron Capacity Suborbitals 1 K 2 s 2 2 L 8 s 2, p 6 3 M 18 s 2, p 6, d 10 4 N 32 s 2, p 6, d 10, f 14 5 O 32 s 2, p 6, d 10, f 14 6 P 18 s 2, p 6, d 10 7 Q 2 s 2 http: //books. google. com/books? id=yb 9 x. Tj 72 v. NAC&pg=PA 15&lpg=PA 15&dq=energy+level+7+shell+letter&source=bl&ots=iw. PSu. Wv. J 0 e&sig=Y 4 LDYy. OBr 16 q. N 3 Zzk. Han. XL 6 J 66 A&hl=en&sa=X&ei=Sh 4 r. VIew. FMTLgg. T 574 HIDw&ved=0 CDc. Q 6 AEw. BA#v=onepage&q&f=false

Resources • http: //books. google. com/books? id=yb 9 x. Tj 72 v. NAC&pg=PA 15&lpg=PA 15&dq=energy+level+7+shell+letter&source =bl&ots=iw. PSu. Wv. J 0 e&sig=Y 4 LDYy. OBr 16 q. N 3 Zzk. Han. XL 6 J 66 A&hl=en&sa=X&ei=Sh 4 r. VIew. FMTLgg. T 574 HIDw&v ed=0 CDc. Q 6 AEw. BA#v=onepage&q&f=false
- Slides: 9