Understanding Specialty Distribution Copyright 2019 by the HDA

Understanding Specialty Distribution Copyright © 2019 by the HDA Research Foundation

About this Deck • Understanding Specialty Distribution was developed as a turn-key presentation that includes an explanation of the pharmaceutical supply chain players and a discussion on the economics of specialty pharmaceutical distribution. • It is composed of a Power. Point module with speaking points, as well as a glossary of terms based on the Specialty Pharmaceutical Distribution: Facts, Figures and Trends (2018) data, as well as other secondary sources. • It is intended to be used by manufacturer, distributor and pharmacy presenters/trainers to “on board” new personnel, provide data for a rich explanation of processes that support how specialty pharmaceutical medication moves from the manufacturer (finished product) to the customer (pharmacy/hospital/clinic). Copyright © 2019 by the HDA Research Foundation

About the Slides • The information appearing on each slide is intended to be used “as is. ” However, the deck has been developed to encourage the presenter to pick and choose the number and order of the slides to be presented. • The “notes” feature of this deck provides speaking points for each slide and allows the presenter to add or subtract concepts, thus enabling fine tuning of each presentation to suit targeted audience needs. Copyright © 2019 by the HDA Research Foundation
About this Product Legal Terms and Conditions Copyright infringement is against the law. If you believe this copy of this e-presentation you are reading infringes on the author’s copyright, please notify the publisher at: pfri@hda. org Copyright © 2019 by the HDA Research Foundation

About the Foundation The HDA Research Foundation is a 501(c)(3) non-profit charitable organization affiliated with the Healthcare Distribution Alliance (HDA). The HDA Research Foundation Mission To conduct research and disseminate information that will enhance the knowledge base, efficiency and effectiveness of the total healthcare supply chain. To provide thought leadership to further enhance the safety and security of the healthcare supply chain through future focused study and programming. Foundation Staff Contact Information Perry Fri, Executive Vice President & Chief Operating Officer pfri@hda. org Copyright © 2019 by the HDA Research Foundation

Specialty Pharmaceuticals 101 US Specialty Pharmaceutical Supply Chain Overview Manufacturer - Distributor Partnership and Operations Distributor – Provider Partnership and Operations Payer Influence on the Supply Chain Regulatory Influence Copyright © 2019 by the HDA Research Foundation

What is a specialty pharmaceutical drug? The HDA Research Foundation, in collaboration with its specialty pharmaceutical task force and industry thought leaders, defines specialty pharmaceuticals as products that treat chronic, complex and rare diseases; and have a minimum of four out of seven additional characteristics related to distribution, care, delivery and/or cost of the medicine. Medicines Must Exhibit Four of These Seven Criteria to be Considered Specialty: Are high in cost (~$6000 or more per year) Initiated/maintained by a specialist Require administration by another individual or healthcare professional (i. e. , not self-administered) Require special handling in the supply chain (e. g. , refrigerated, frozen, chemo precautions, biohazard) Require patient payment assistance Distributed through non-traditional channels Medication has significant side effects that require additional monitoring/counseling (including, but not limited to REMS programs) and/or disease requires additional monitoring of therapy (e. g. , monitoring of blood/cell counts to assess effectiveness/side effects of therapy) Copyright © 2019 by the HDA Research Foundation

Specialty pharmaceuticals differ from common therapies in a variety of aspects Traditional Specialty Type of Condition Common Acute Common Chronic Complex Chronic Rare Disease U. S. Patient Population Millions Affects >50 million Affects ~ 20 K Duration of Therapy About 10 days/ episode Ongoing (maintenance) Lifelong Cost of Therapy ~ $100/episode $1, 000+/year $17, 000+/year $250, 000/year Medication Examples Anti-infective Lipitor® Enbrel Cerezyme Indication Examples Acute bacterial infection Cholesterol reduction Rheumatoid arthritis Gaucher’s disease Handling Requirements No special requirements Refrigeration training Refrigeration/ mixing/pumps/ central line training ® ® Source: IQVIA Copyright © 2019 by the HDA Research Foundation 7
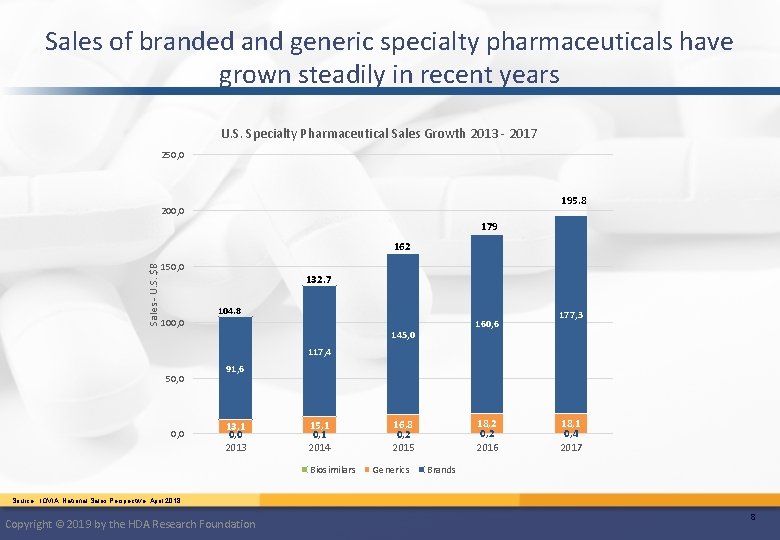
Sales of branded and generic specialty pharmaceuticals have grown steadily in recent years U. S. Specialty Pharmaceutical Sales Growth 2013 - 2017 250, 0 195. 8 200, 0 179 Sales - U. S. $B 162 150, 0 100, 0 132. 7 104. 8 160, 6 145, 0 177, 3 117, 4 50, 0 91, 6 13, 1 0, 0 2013 15, 1 0, 1 2014 Biosimilars 18, 2 0, 2 2016 16, 8 0, 2 2015 Generics 18, 1 0, 4 2017 Brands Source: IQVIA, National Sales Perspective, April 2018 Copyright © 2019 by the HDA Research Foundation 8

The top 20 branded specialty drugs account for 43% of the $195. 8 B US specialty drug market Humira® Enbrel® Harvoni® Remicade® Neulasta® Copaxone® Rituxan® Stelara® Genvoya® Tecfidera® Opdivo® Ibrance® Avastin® Truvada® Herceptin® Epclusa® Triumeq® Keytruda® Gilenya® Imbruvica® Top 20 All Other Total U. S. Specialty 16. 3 7. 9 5. 5 4. 2 4. 1 4. 0 3. 7 3. 6 3. 1 2. 9 2. 8 2. 7 2. 5 2. 2 84. 9 110. 8 195. 8 8% 4% 3% 3% 2% 2% 1% 1% 43% 57% 100% 2. 8 0. 3 -3. 8 0. 2 -0. 0 -0. 1 1. 1 2. 0 0. 1 0. 5 0. 8 -0. 1 -0. 2 1. 6 0. 7 1. 5 0. 2 0. 6 8. 4 16. 8 21% 4% -41% 3% 0% -2% 4% 43% 126% 4% 17% 39% -5% -7% 6% 149% 36% 204% 9% 37% 11% 8% 9% Source: IQVIA National Sales Perspectives, April 2018 Copyright © 2019 by the HDA Research Foundation 9
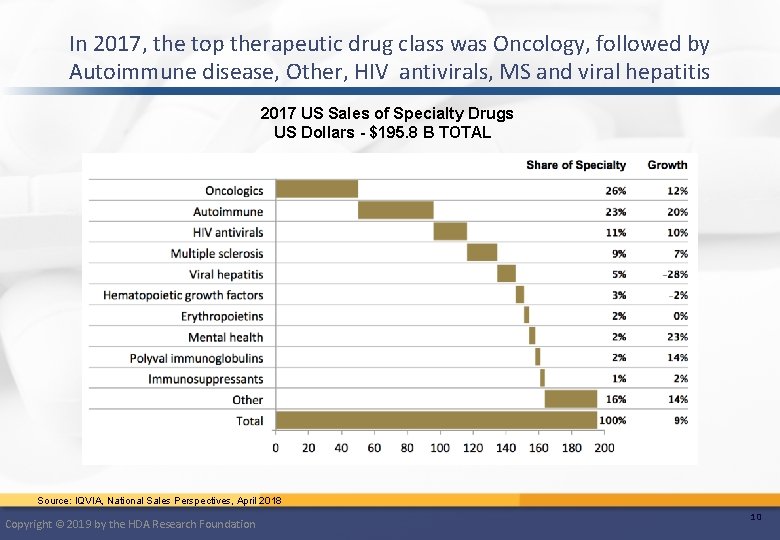
In 2017, the top therapeutic drug class was Oncology, followed by Autoimmune disease, Other, HIV antivirals, MS and viral hepatitis 2017 US Sales of Specialty Drugs US Dollars - $195. 8 B TOTAL Source: IQVIA, National Sales Perspectives, April 2018 Copyright © 2019 by the HDA Research Foundation 10

Specialty Pharmaceuticals 101 US Specialty Pharmaceutical Supply Chain Overview Manufacturer - Distributor Partnership and Operations Distributor – Provider Partnership and Operations Payer Influence on the Supply Chain Regulatory Influence Copyright © 2019 by the HDA Research Foundation
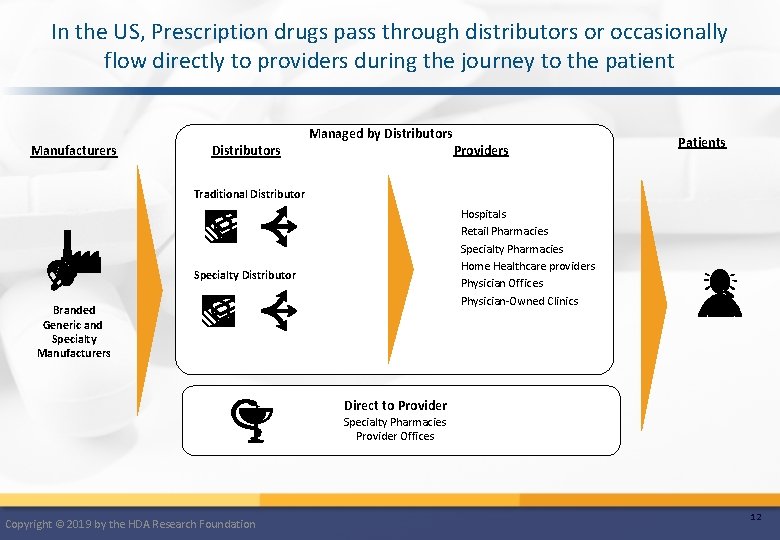
In the US, Prescription drugs pass through distributors or occasionally flow directly to providers during the journey to the patient Manufacturers Distributors Managed by Distributors Providers Patients Traditional Distributor Hospitals Retail Pharmacies Specialty Pharmacies Home Healthcare providers Physician Offices Physician-Owned Clinics Specialty Distributor Branded Generic and Specialty Manufacturers Direct to Provider Specialty Pharmacies Provider Offices Copyright © 2019 by the HDA Research Foundation 12
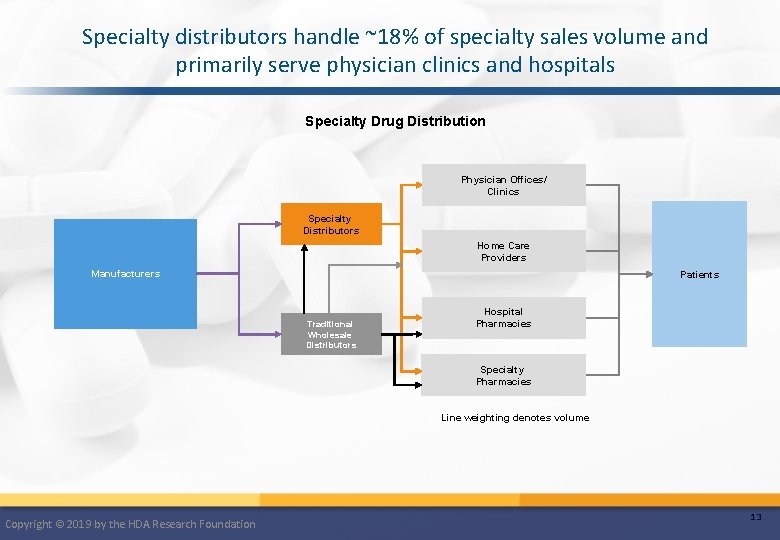
Specialty distributors handle ~18% of specialty sales volume and primarily serve physician clinics and hospitals Specialty Drug Distribution Physician Offices/ Clinics Specialty Distributors Home Care Providers Manufacturers Patients Traditional Wholesale Distributors Hospital Pharmacies Specialty Pharmacies Line weighting denotes volume Copyright © 2019 by the HDA Research Foundation 13
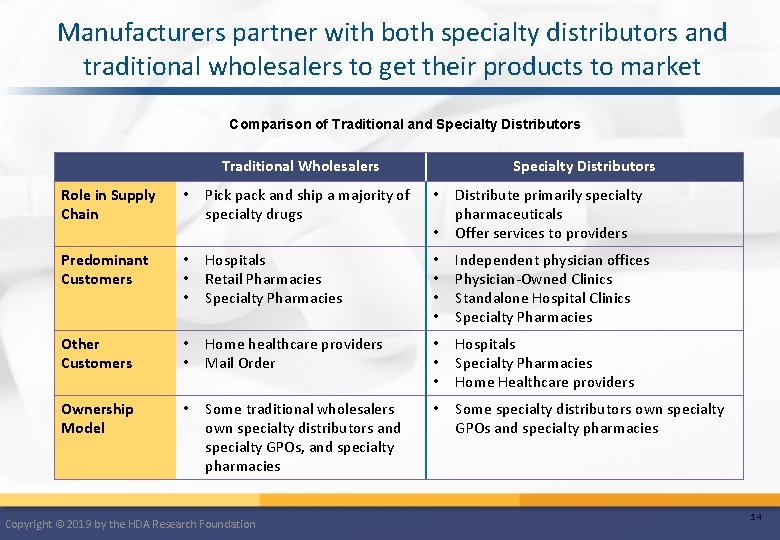
Manufacturers partner with both specialty distributors and traditional wholesalers to get their products to market Comparison of Traditional and Specialty Distributors Traditional Wholesalers Role in Supply Chain • Predominant Customers Specialty Distributors Pick pack and ship a majority of specialty drugs • • Distribute primarily specialty pharmaceuticals Offer services to providers • • • Hospitals Retail Pharmacies Specialty Pharmacies • • Independent physician offices Physician-Owned Clinics Standalone Hospital Clinics Specialty Pharmacies Other Customers • • Home healthcare providers Mail Order • • • Hospitals Specialty Pharmacies Home Healthcare providers Ownership Model • Some traditional wholesalers own specialty distributors and specialty GPOs, and specialty pharmacies • Some specialty distributors own specialty GPOs and specialty pharmacies Copyright © 2019 by the HDA Research Foundation 14

Specialty pharmacies receive products primarily from distributors then dispense prescriptions to providers or patients Specialty Pharmacies Role in Supply Chain • • • Location Type Location type varies significantly, for example: • Standalone entity focusing just on specialty drugs • Within a hospital • Within a traditional retail pharmacy • Mail order Ownership • • Order and receive medications primarily from distributors Licensed to dispense medications / fulfill prescriptions Provide patient counseling and support Vary from independently owned to owned by PBMs, payers, drugstore chains pharmacies, specialty distributors, traditional wholesalers, provider practices, GPO Traditional retail pharmacies are entering the specialty pharmacy space – causing a blurring of the distinctions between retail and specialty pharmacies Copyright © 2019 by the HDA Research Foundation 15

Specialty Group Procurement Organizations (GPOs) play an important role in supporting providers in procuring products from manufacturers Specialty GPOs What is a GPO? • • Role in Supply Chain • • An entity which specializes in certain pharmaceutical procurement activities on behalf of its provider members Establish contracts with pharmaceutical manufacturers, and leverage contracted volume to negotiate better pricing for provider members Support provider members in procurement activities including developing strategy, designing / executing contracts, conducting price negotiations Do not distribute or dispense drugs Customers GPO Provider Members: • Independent physician offices • Physician-Owned Clinics • Hospitals • Specialty Pharmacies • Home Healthcare providers Ownership • Several of the largest GPOs are owned by specialty distributors or traditional wholesalers; One is owned by a PBM Copyright © 2019 by the HDA Research Foundation 16

Specialty drugs are unique in that they often require special handling to ensure product viability Special Handling: Products § Many specialty drugs have special handling requirements – temperatures may need to be held within a certain range to ensure product viability Cold Chain: Temperature Control § Products requiring cold chain handling may include, but are not limited to vaccines, biologics, hormones, insulin, and certain antibiotics § Distribution centers have separate areas for products requiring special handling Warehouse Handling § Cold chain products are stored in separate chilled areas within the warehouse to ensure tight control over temperature ranges § Cold chain products are packed in special qualified containers or boxes containing cold packs, and may have temperature monitors enclosed in the package In-Transit Handling Copyright © 2019 by the HDA Research Foundation 17
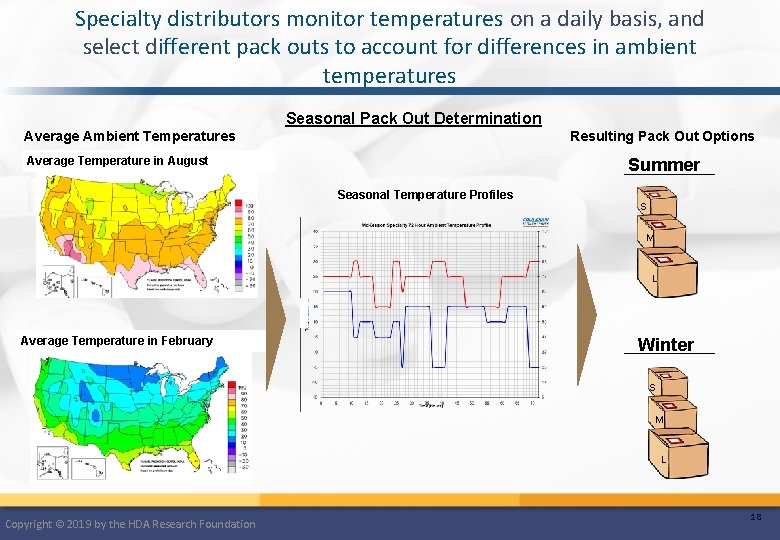
Specialty distributors monitor temperatures on a daily basis, and select different pack outs to account for differences in ambient temperatures Seasonal Pack Out Determination Resulting Pack Out Options Average Ambient Temperatures Average Temperature in August Summer Seasonal Temperature Profiles S M L Average Temperature in February Winter S M L Copyright © 2019 by the HDA Research Foundation 18

Specialty pharmaceutical manufacturers carefully choose their distribution model so that it meets several primary goals Manufacturer Goals § Maximize patient and provider access to the specialty pharmaceutical product § Provide clinical and educational support for drugs with complex dosing regimens § Ensure support exists in the event of adverse events § Optimize patient and provider reimbursement for the products, including securing financial assistance for patients, when needed § Minimize supply chain costs and maximize supply chain efficiencies § Ensure product integrity and reduce the risk of counterfeiting or tampering § Meet regulatory requirements, such as complying with FDA REMS mandates Copyright © 2019 by the HDA Research Foundation 19

Manufacturers consider drug characteristics, regulatory requirements and reimbursement norms when choosing the distribution model Sample of Key Questions Considered When Choosing a Distribution Model Copyright © 2019 by the HDA Research Foundation 20
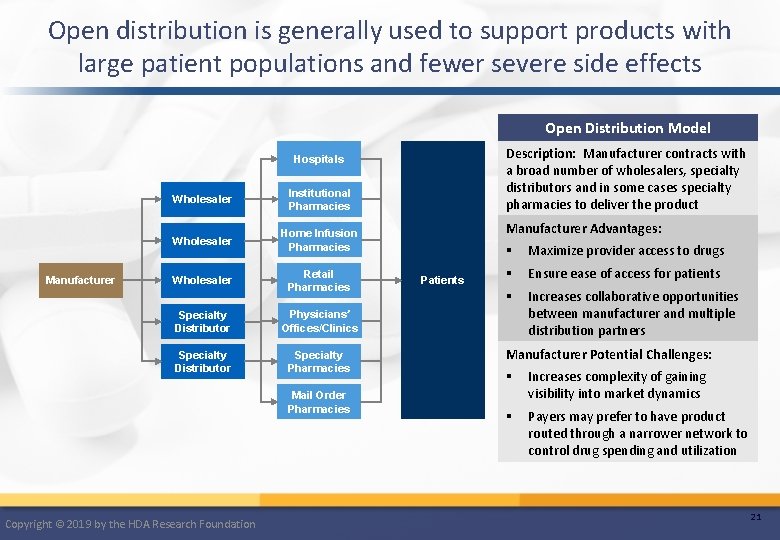
Open distribution is generally used to support products with large patient populations and fewer severe side effects Open Distribution Model Description: Manufacturer contracts with a broad number of wholesalers, specialty distributors and in some cases specialty pharmacies to deliver the product Hospitals Manufacturer Wholesaler Institutional Pharmacies Wholesaler Home Infusion Pharmacies Wholesaler Retail Pharmacies Specialty Distributor Physicians’ Offices/Clinics Specialty Distributor Specialty Pharmacies Mail Order Pharmacies Copyright © 2019 by the HDA Research Foundation Manufacturer Advantages: Patients § Maximize provider access to drugs § Ensure ease of access for patients § Increases collaborative opportunities between manufacturer and multiple distribution partners Manufacturer Potential Challenges: § Increases complexity of gaining visibility into market dynamics § Payers may prefer to have product routed through a narrower network to control drug spending and utilization 21
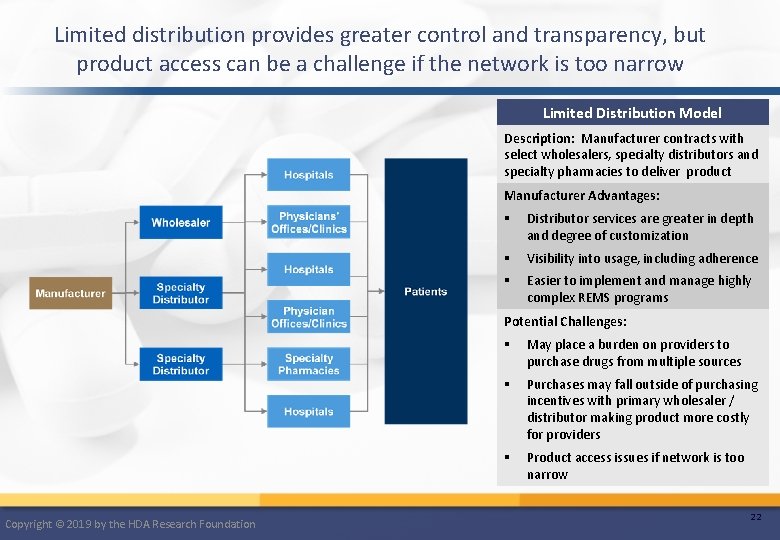
Limited distribution provides greater control and transparency, but product access can be a challenge if the network is too narrow Limited Distribution Model Description: Manufacturer contracts with select wholesalers, specialty distributors and specialty pharmacies to deliver product Manufacturer Advantages: § Distributor services are greater in depth and degree of customization § Visibility into usage, including adherence § Easier to implement and manage highly complex REMS programs Potential Challenges: Copyright © 2019 by the HDA Research Foundation § May place a burden on providers to purchase drugs from multiple sources § Purchases may fall outside of purchasing incentives with primary wholesaler / distributor making product more costly for providers § Product access issues if network is too narrow 22
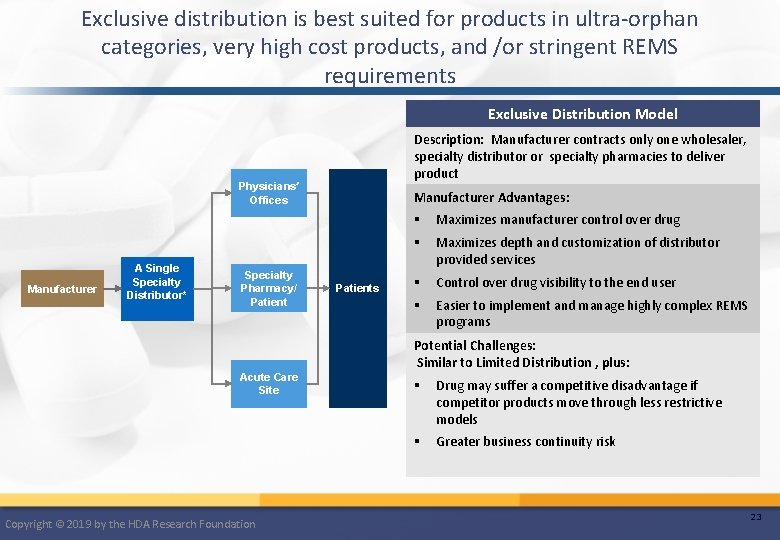
Exclusive distribution is best suited for products in ultra-orphan categories, very high cost products, and /or stringent REMS requirements Exclusive Distribution Model Description: Manufacturer contracts only one wholesaler, specialty distributor or specialty pharmacies to deliver product Physicians’ Offices Manufacturer A Single Specialty Distributor* Specialty Pharmacy/ Patient Manufacturer Advantages: Patients § Maximizes manufacturer control over drug § Maximizes depth and customization of distributor provided services § Control over drug visibility to the end user § Easier to implement and manage highly complex REMS programs Potential Challenges: Similar to Limited Distribution , plus: Acute Care Site Copyright © 2019 by the HDA Research Foundation § Drug may suffer a competitive disadvantage if competitor products move through less restrictive models § Greater business continuity risk 23

Specialty Pharmaceuticals 101 US Specialty Pharmaceutical Supply Chain Overview Manufacturer - Distributor Partnership and Operations Distributor – Provider Partnership and Operations Payer Influence on the Supply Chain Regulatory Influence Copyright © 2019 by the HDA Research Foundation
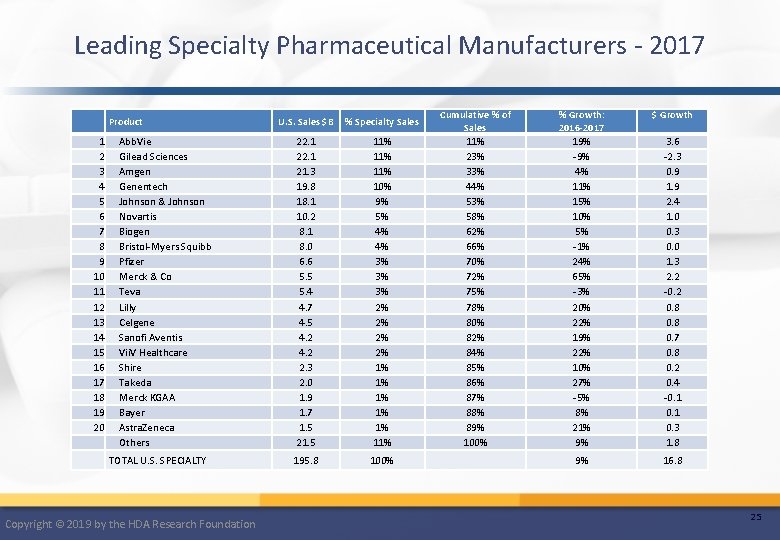
Leading Specialty Pharmaceutical Manufacturers - 2017 Product 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Abb. Vie Gilead Sciences Amgen Genentech Johnson & Johnson Novartis Biogen Bristol-Myers Squibb Pfizer Merck & Co Teva Lilly Celgene Sanofi Aventis Vii. V Healthcare Shire Takeda Merck KGAA Bayer Astra. Zeneca Others TOTAL U. S. SPECIALTY Copyright © 2019 by the HDA Research Foundation U. S. Sales $B % Specialty Sales 22. 1 21. 3 19. 8 18. 1 10. 2 8. 1 8. 0 6. 6 5. 5 5. 4 4. 7 4. 5 4. 2 2. 3 2. 0 1. 9 1. 7 1. 5 21. 5 11% 11% 10% 9% 5% 4% 4% 3% 3% 3% 2% 2% 1% 1% 11% 195. 8 100% Cumulative % of Sales 11% 23% 33% 44% 53% 58% 62% 66% 70% 72% 75% 78% 80% 82% 84% 85% 86% 87% 88% 89% 100% % Growth: 2016 -2017 19% -9% 4% 11% 15% 10% 5% -1% 24% 65% -3% 20% 22% 19% 22% 10% 27% -5% 8% 21% 9% $ Growth 9% 16. 8 3. 6 -2. 3 0. 9 1. 9 2. 4 1. 0 0. 3 0. 0 1. 3 2. 2 -0. 2 0. 8 0. 7 0. 8 0. 2 0. 4 -0. 1 0. 3 1. 8 25

Manufacturers may utilize a wide array of supply chain, financial and information services provided by their specialty distribution partners Core Specialty Distributor Services Manufacturers Supply Chain: Financials: § Order management and fulfilment § Ownership of credit risk (receivables) § New product launch support § Chargeback administration § Inventory management Data / Information: § Reverse logistics / returns § Data services: basic; e. g. sales, inventory, returns § Recalls § Data services: enhanced; e. g. clinical performance § Security § Web based portal solutions § Special handling services Regulatory § Disaster preparedness § Business continuity risk management § Support compliance with federal and state regulations § Packaging and re-packaging services § REMS support § Product compliance support services Copyright © 2019 by the HDA Research Foundation 26

Distributors are also offering manufacturers additional services to support the use of specialty pharmaceutical products Other Specialty Distributor Services Manufacturers Patient & Provider Support Reimbursement Support: § Adherence management programs § Reimbursement Services/Consulting § Provide 3 rd party support services (contract review, reimbursement support) § Medication preauthorization § Utilization Management § Claims / co-payment collection § Patient Counselling Marketing: § Refill reminders § Promotional material distribution § Disease management § Customer access and knowledge § Disease state advocacy Hub Services § Provider advocacy § Customer call centers § Patient assistance program management § Treatment initiation and education Copyright © 2019 by the HDA Research Foundation 27

Manufacturer - Distributor operations ensure that drug inventory is readily available for distribution to providers 1 Manufacturers create forecasts using historical shipment data received from distributors, as well as latest market intelligence 2 Distributors order drugs from manufacturers using EDI technology 3 Manufacturers ship drugs to distributor warehouses 4 Distributors receive and put away goods from manufacturers – High degree of automation and WMS technology utilized to ensure that goods are received, and put away in the warehouse – Bar-code scanning, RFID – Inventory tracking systems 5 Distributors send manufacturers reports and data to provide transparency into inventory levels and shipments Copyright © 2019 by the HDA Research Foundation 28
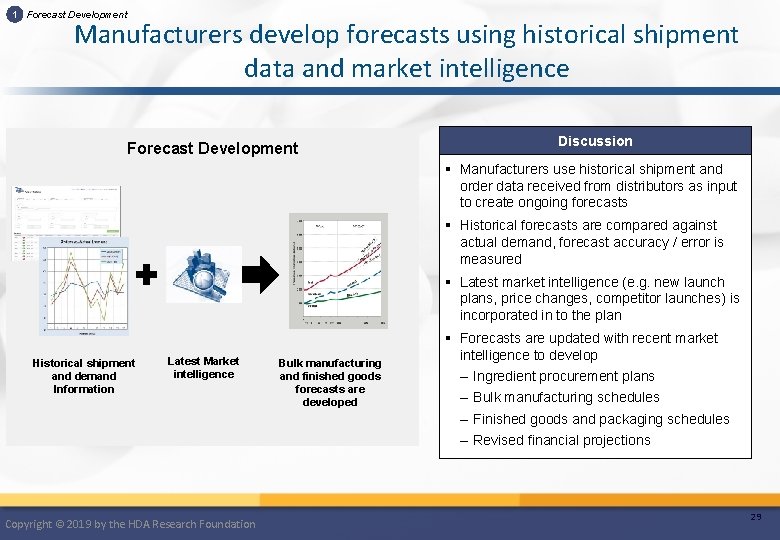
1 Forecast Development Manufacturers develop forecasts using historical shipment data and market intelligence Forecast Development Discussion § Manufacturers use historical shipment and order data received from distributors as input to create ongoing forecasts § Historical forecasts are compared against actual demand, forecast accuracy / error is measured § Latest market intelligence (e. g. new launch plans, price changes, competitor launches) is incorporated in to the plan Historical shipment and demand Information Latest Market intelligence Copyright © 2019 by the HDA Research Foundation Bulk manufacturing and finished goods forecasts are developed § Forecasts are updated with recent market intelligence to develop – Ingredient procurement plans – Bulk manufacturing schedules – Finished goods and packaging schedules – Revised financial projections 29
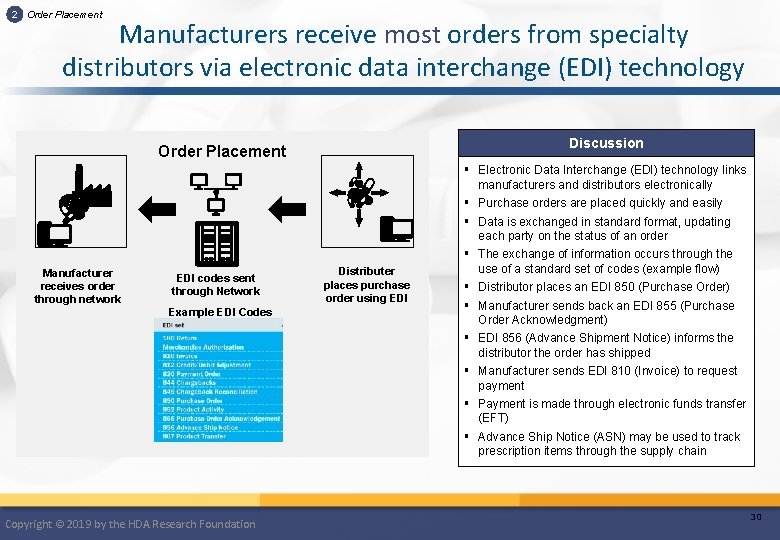
2 Order Placement Manufacturers receive most orders from specialty distributors via electronic data interchange (EDI) technology Discussion Order Placement Manufacturer receives order through network EDI codes sent through Network Example EDI Codes Copyright © 2019 by the HDA Research Foundation Distributer places purchase order using EDI § Electronic Data Interchange (EDI) technology links manufacturers and distributors electronically § Purchase orders are placed quickly and easily § Data is exchanged in standard format, updating each party on the status of an order § The exchange of information occurs through the use of a standard set of codes (example flow) § Distributor places an EDI 850 (Purchase Order) § Manufacturer sends back an EDI 855 (Purchase Order Acknowledgment) § EDI 856 (Advance Shipment Notice) informs the distributor the order has shipped § Manufacturer sends EDI 810 (Invoice) to request payment § Payment is made through electronic funds transfer (EFT) § Advance Ship Notice (ASN) may be used to track prescription items through the supply chain 30
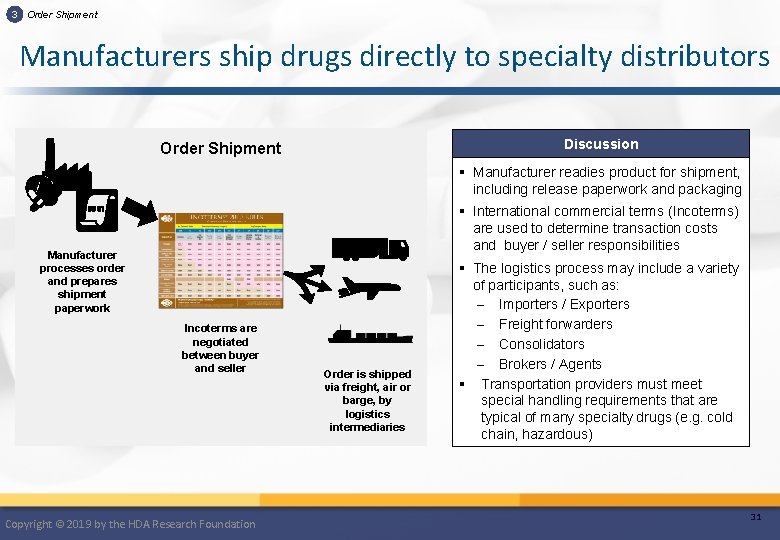
3 Order Shipment Manufacturers ship drugs directly to specialty distributors Discussion Order Shipment § Manufacturer readies product for shipment, including release paperwork and packaging § International commercial terms (Incoterms) are used to determine transaction costs and buyer / seller responsibilities Manufacturer processes order and prepares shipment paperwork Incoterms are negotiated between buyer and seller Copyright © 2019 by the HDA Research Foundation Order is shipped via freight, air or barge, by logistics intermediaries § The logistics process may include a variety of participants, such as: ‒ Importers / Exporters ‒ Freight forwarders ‒ Consolidators ‒ Brokers / Agents § Transportation providers must meet special handling requirements that are typical of many specialty drugs (e. g. cold chain, hazardous) 31
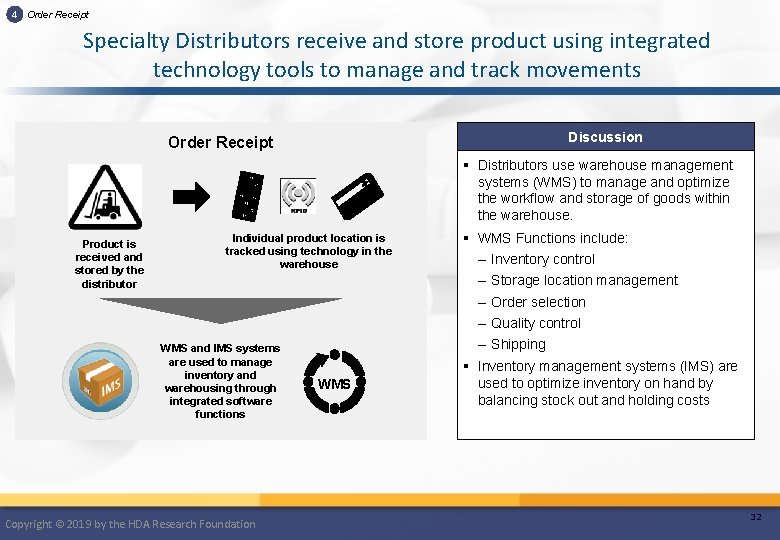
4 Order Receipt Specialty Distributors receive and store product using integrated technology tools to manage and track movements Discussion Order Receipt § Distributors use warehouse management systems (WMS) to manage and optimize the workflow and storage of goods within the warehouse. Product is received and stored by the distributor Individual product location is tracked using technology in the warehouse WMS and IMS systems are used to manage inventory and warehousing through integrated software functions Copyright © 2019 by the HDA Research Foundation WMS § WMS Functions include: – Inventory control – Storage location management – Order selection – Quality control – Shipping § Inventory management systems (IMS) are used to optimize inventory on hand by balancing stock out and holding costs 32
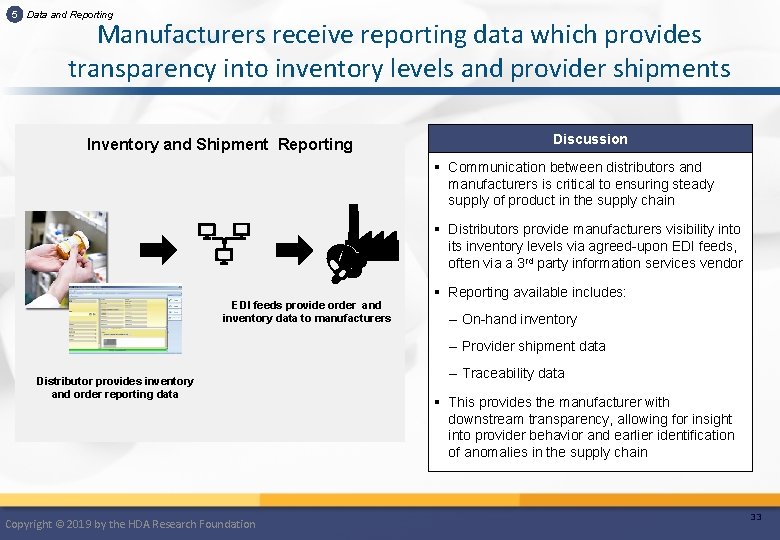
5 Data and Reporting Manufacturers receive reporting data which provides transparency into inventory levels and provider shipments Inventory and Shipment Reporting Discussion § Communication between distributors and manufacturers is critical to ensuring steady supply of product in the supply chain § Distributors provide manufacturers visibility into its inventory levels via agreed-upon EDI feeds, often via a 3 rd party information services vendor EDI feeds provide order and inventory data to manufacturers § Reporting available includes: – On-hand inventory – Provider shipment data Distributor provides inventory and order reporting data Copyright © 2019 by the HDA Research Foundation – Traceability data § This provides the manufacturer with downstream transparency, allowing for insight into provider behavior and earlier identification of anomalies in the supply chain 33

Specialty Pharmaceuticals 101 US Specialty Pharmaceutical Supply Chain Overview Manufacturer - Distributor Partnership and Operations Distributor – Provider Partnership and Operations Payer Influence on the Supply Chain Regulatory Influence Copyright © 2019 by the HDA Research Foundation

Providers elect to work with specialty distribution partners who ensure safe, timely supply and may provide many other services Core Specialty Distributor Services Providers Supply Chain: Financials: § Order management and fulfilment § Payer economic modelling and negotiation support § Inventory management § Financial management / access to credit § Reverse logistics / returns Data / Information: § Recalls § Pharmacy management systems and services § Security § Web based portal solutions § Special handling services Regulatory § Disaster preparedness § Business continuity risk management § Support compliance with federal and state regulations § Packaging and re-packaging services § REMS support § Product compliance support services Copyright © 2019 by the HDA Research Foundation 35

Providers also benefit from additional services provided by distributors on behalf of manufacturers Additional Specialty Distributor Services Patient & Provider Support Reimbursement Support: § Adherence management programs § Reimbursement Services/Consulting § Provide 3 rd party support services (contract review, § Medication preauthorization reimbursement support) § Patient assistance program management § Utilization Management § Claims / co-payment collection Providers § Patient Counselling Hub Services § Refill reminders § Customer Call Centers § Disease management § Disease state advocacy § Provider advocacy § Treatment initiation and education Copyright © 2019 by the HDA Research Foundation 36

The provider order fulfillment process is highly efficient, ensuring safe, secure delivery of drugs in a timely manner A Day in the Life of a Provider Pharmaceutical Order 1 Provider places an order with their specialty distribution partner (EDI, online ordering, phone or fax) 2 Specialty Distributor receives the order, which is routed to the distribution center 3 Sophisticated WMS systems are used to enable the pick-pack-ship operations to run smoothly and efficiently (examples of systems, and capabilities of sophisticated WMS systems) 4 Orders are picked using different technology based on inventory velocity and product handling requirements 5 6 Order is packed and shipped via UPS, Courier or a Delivery Truck, depending on the size of the order Order is received at the provider, in some cases, as early as the same day of placement Copyright © 2019 by the HDA Research Foundation
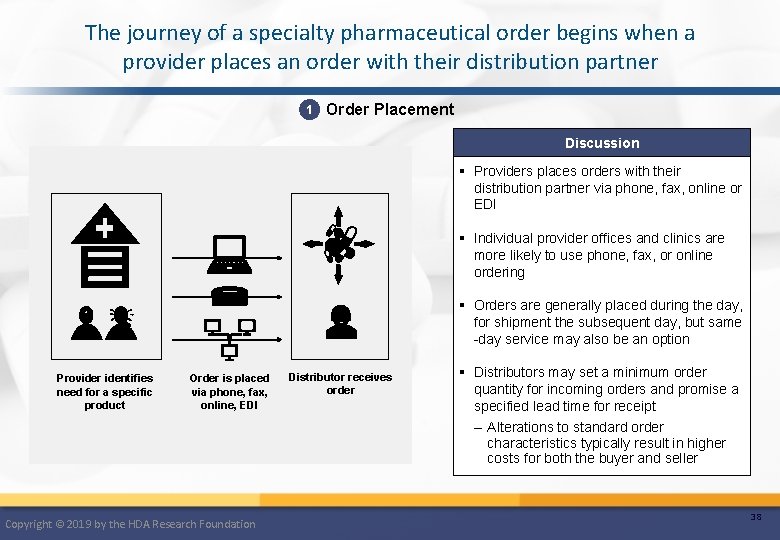
The journey of a specialty pharmaceutical order begins when a provider places an order with their distribution partner 1 Order Placement Discussion § Providers places orders with their distribution partner via phone, fax, online or EDI § Individual provider offices and clinics are more likely to use phone, fax, or online ordering § Orders are generally placed during the day, for shipment the subsequent day, but same -day service may also be an option Provider identifies need for a specific product Order is placed via phone, fax, online, EDI Copyright © 2019 by the HDA Research Foundation Distributor receives order § Distributors may set a minimum order quantity for incoming orders and promise a specified lead time for receipt – Alterations to standard order characteristics typically result in higher costs for both the buyer and seller 38
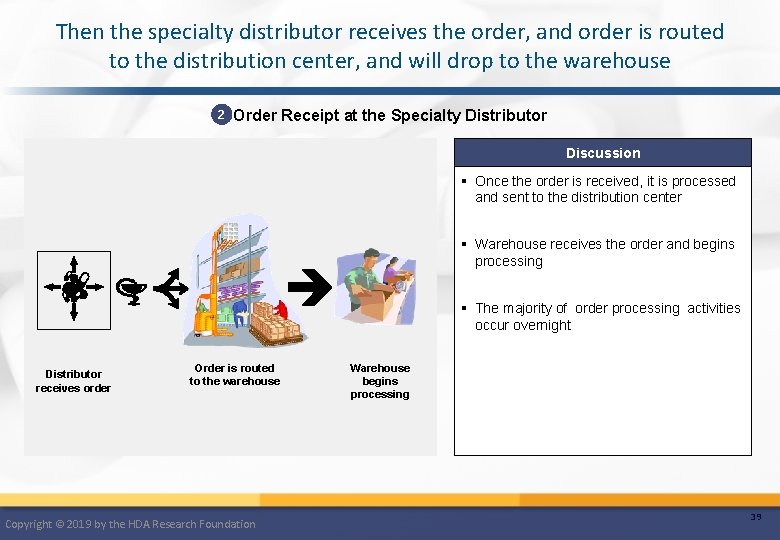
Then the specialty distributor receives the order, and order is routed to the distribution center, and will drop to the warehouse 2 Order Receipt at the Specialty Distributor Discussion § Once the order is received, it is processed and sent to the distribution center § Warehouse receives the order and begins processing § The majority of order processing activities occur overnight Distributor receives order Order is routed to the warehouse Copyright © 2019 by the HDA Research Foundation Warehouse begins processing 39
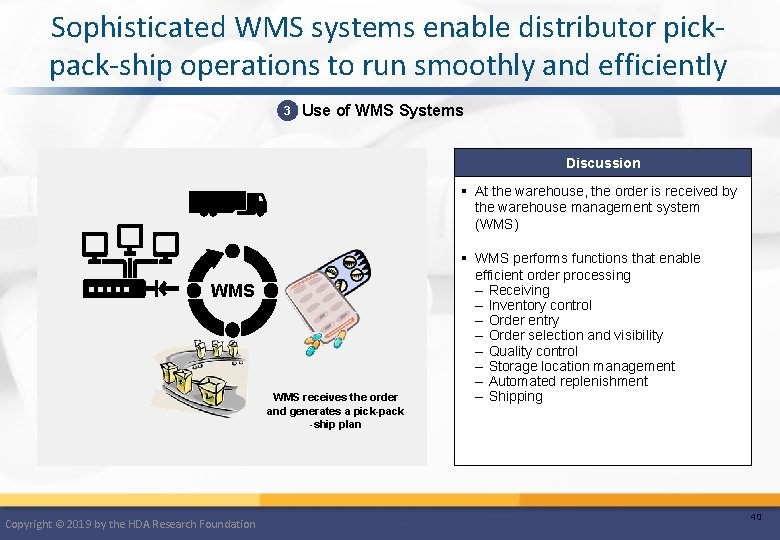
Sophisticated WMS systems enable distributor pickpack-ship operations to run smoothly and efficiently 3 Use of WMS Systems Discussion § At the warehouse, the order is received by the warehouse management system (WMS) WMS receives the order and generates a pick-pack -ship plan Copyright © 2019 by the HDA Research Foundation § WMS performs functions that enable efficient order processing – Receiving – Inventory control – Order entry – Order selection and visibility – Quality control – Storage location management – Automated replenishment – Shipping 40
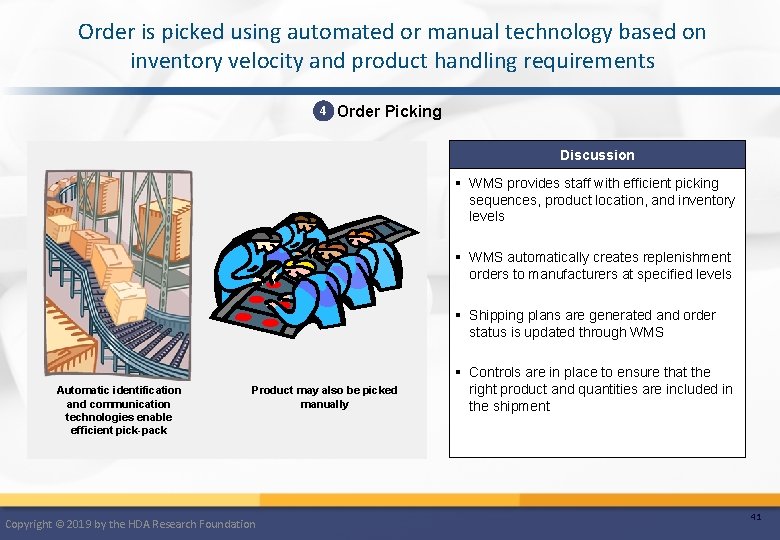
Order is picked using automated or manual technology based on inventory velocity and product handling requirements 4 Order Picking Discussion § WMS provides staff with efficient picking sequences, product location, and inventory levels § WMS automatically creates replenishment orders to manufacturers at specified levels § Shipping plans are generated and order status is updated through WMS Automatic identification and communication technologies enable efficient pick-pack Product may also be picked manually Copyright © 2019 by the HDA Research Foundation § Controls are in place to ensure that the right product and quantities are included in the shipment 41
When the full order has been picked and packed, it will be sent to the provider 5 Order Packing and Shipping Discussion § Warehouse readies order for shipment, including paperwork and packaging § Manufacturers may mandate that distributors use certain packing types for some products requiring specialty handling Warehouse prepares order for shipment Copyright © 2019 by the HDA Research Foundation Order is shipped via most appropriate transportation method § Orders will be shipped in different modes depending on order size and service level agreements including – Small parcel shipments – Courier delivery – Less than truckload (LTL) – for large hospital systems or large pharmacies 42
Order is received at the provider, as early as the same day of order placement depending on service level agreements 6 Order Receipt by Provider Discussion § Typically, orders are placed in one day, then shipped and received the next § Depending on urgency or service level agreements, product may be ordered and delivered on the same day § Provider receives the product as well as administrative tracing information. § Provider stores drugs in appropriate conditions upon receipt (e. g. freezer, chiller room temperature) Order is delivered to provider Provider receives order with appropriate paperwork Copyright © 2019 by the HDA Research Foundation 43
Hospitals rely on specialty distributors for order fulfillment, and commonly leverage GPO contracts to obtain better pricing on drugs Distribution Characteristics Provider Characteristics Hospitals § Formulary-based ordering practices § Administrative support, ordering, billing, systems Shipment/ Receipt § Direct shipments, with moderate inventory levels § Cold chain requirements § Medium shipment quantities to individual drop locations Service § Ancillary service needs § RFID enabled Inventory Cabinet systems, Software § High service level agreements Financial § Leverage buying power with Group Purchasing Organizations § Variable depending on hospital size § Large quantity discounts Ordering/ Forecasting § Regional networks and standalone institutions § Commonly participate in Group Purchasing Organizations (GPO) to leverage purchasing power of a group Copyright © 2019 by the HDA Research Foundation Distributor Services 44
Individual provider offices require high service levels for small orders, and may need additional administrative and financial support Distribution Characteristics Provider Offices / Clinics Ordering/ Forecasting Shipment/ Receipt § Small, individual offices § Require specialty services and cold chain capabilities § May require more financial support due to the high cost of specialty drugs Copyright © 2019 by the HDA Research Foundation Service Financial Distributor Services § Smaller order quantities § Technology capabilities vary § Administrative support § May leverage distributor ordering systems § Limited storage space § Cold chain requirements § Small order quantities § Complex delivery network § Specialty product handling § Rapid order fulfillment needs § Reimbursement support needs § High service levels § Rapid order responsiveness § Potential for longer payment schedule § Payment terms § Financial credit § Consignment inventory 45
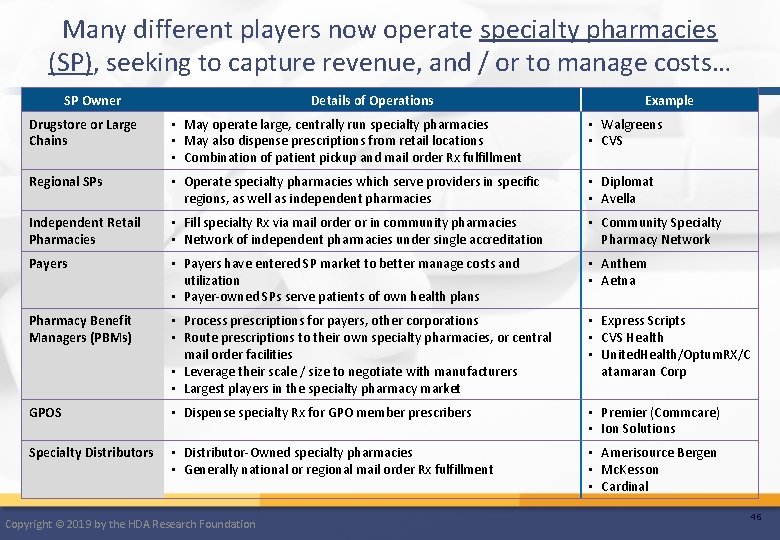
Many different players now operate specialty pharmacies (SP), seeking to capture revenue, and / or to manage costs… SP Owner Details of Operations Example Drugstore or Large Chains • May operate large, centrally run specialty pharmacies • May also dispense prescriptions from retail locations • Combination of patient pickup and mail order Rx fulfillment • Walgreens • CVS Regional SPs • Operate specialty pharmacies which serve providers in specific regions, as well as independent pharmacies • Diplomat • Avella Independent Retail Pharmacies • Fill specialty Rx via mail order or in community pharmacies • Network of independent pharmacies under single accreditation • Community Specialty Pharmacy Network Payers • Payers have entered SP market to better manage costs and utilization • Payer-owned SPs serve patients of own health plans • Anthem • Aetna Pharmacy Benefit Managers (PBMs) • Process prescriptions for payers, other corporations • Route prescriptions to their own specialty pharmacies, or central mail order facilities • Leverage their scale / size to negotiate with manufacturers • Largest players in the specialty pharmacy market • Express Scripts • CVS Health • United. Health/Optum. RX/C atamaran Corp GPOS • Dispense specialty Rx for GPO member prescribers • Premier (Commcare) • Ion Solutions Specialty Distributors • Distributor-Owned specialty pharmacies • Generally national or regional mail order Rx fulfillment • Amerisource Bergen • Mc. Kesson • Cardinal Copyright © 2019 by the HDA Research Foundation 46

Specialty Pharmaceuticals 101 US Specialty Pharmaceutical Supply Chain Overview Manufacturer - Distributor Partnership and Operations Distributor – Provider Partnership and Operations Payer Influence on the Supply Chain Regulatory Influence Copyright © 2019 by the HDA Research Foundation
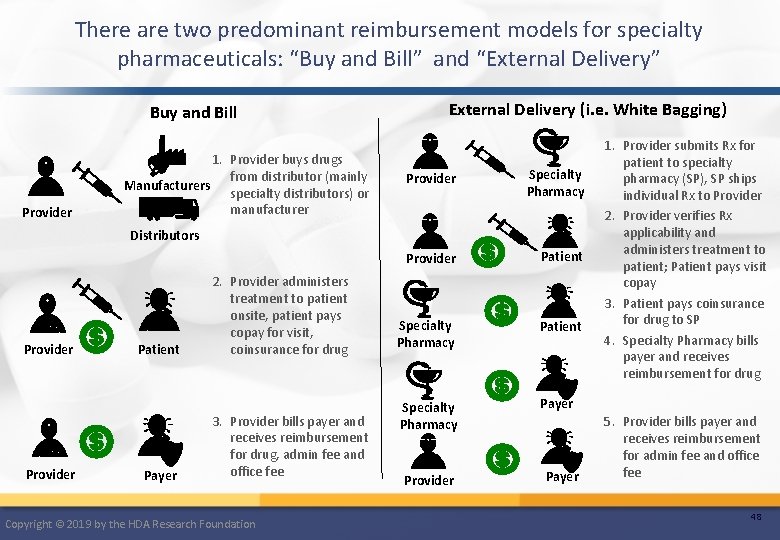
There are two predominant reimbursement models for specialty pharmaceuticals: “Buy and Bill” and “External Delivery” Buy and Bill Provider 1. Provider buys drugs from distributor (mainly Manufacturers specialty distributors) or manufacturer External Delivery (i. e. White Bagging) Provider Specialty Pharmacy Provider Patient Specialty Pharmacy Payer Distributors Provider Patient Payer 2. Provider administers treatment to patient onsite, patient pays copay for visit, coinsurance for drug 3. Provider bills payer and receives reimbursement for drug, admin fee and office fee Copyright © 2019 by the HDA Research Foundation Provider Payer 1. Provider submits Rx for patient to specialty pharmacy (SP), SP ships individual Rx to Provider 2. Provider verifies Rx applicability and administers treatment to patient; Patient pays visit copay 3. Patient pays coinsurance for drug to SP 4. Specialty Pharmacy bills payer and receives reimbursement for drug 5. Provider bills payer and receives reimbursement for admin fee and office fee 48
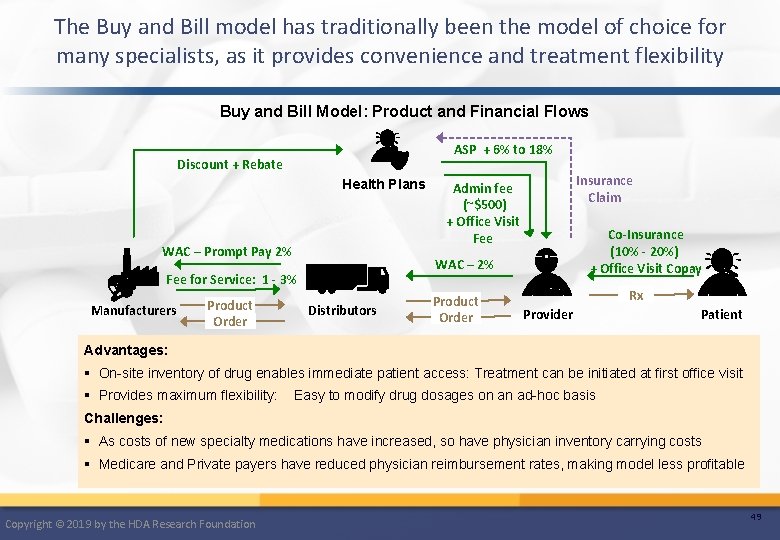
The Buy and Bill model has traditionally been the model of choice for many specialists, as it provides convenience and treatment flexibility Buy and Bill Model: Product and Financial Flows ASP + 6% to 18% Discount + Rebate Health Plans WAC – Prompt Pay 2% Manufacturers Co-Insurance (10% - 20%) + Office Visit Copay WAC – 2% Fee for Service: 1 - 3% Product Order Insurance Claim Admin fee (~$500) + Office Visit Fee Distributors Product Order Rx Provider Patient Advantages: § On-site inventory of drug enables immediate patient access: Treatment can be initiated at first office visit § Provides maximum flexibility: Easy to modify drug dosages on an ad-hoc basis Challenges: § As costs of new specialty medications have increased, so have physician inventory carrying costs § Medicare and Private payers have reduced physician reimbursement rates, making model less profitable Copyright © 2019 by the HDA Research Foundation 49
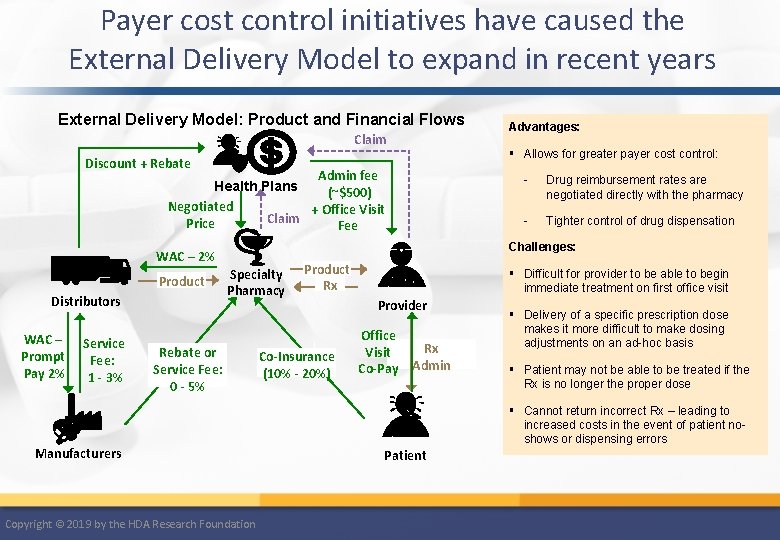
Payer cost control initiatives have caused the External Delivery Model to expand in recent years External Delivery Model: Product and Financial Flows Claim § Allows for greater payer cost control: Discount + Rebate Admin fee Health Plans (~$500) Negotiated + Office Visit Claim Price Fee Distributors WAC – Prompt Pay 2% Service Fee: 1 - 3% - Drug reimbursement rates are negotiated directly with the pharmacy - Tighter control of drug dispensation Challenges: WAC – 2% Product Advantages: Specialty Pharmacy Rebate or Service Fee: 0 - 5% Manufacturers Copyright © 2019 by the HDA Research Foundation Product Rx Co-Insurance (10% - 20%) § Difficult for provider to be able to begin immediate treatment on first office visit Provider Office Visit Co-Pay Rx Admin § Delivery of a specific prescription dose makes it more difficult to make dosing adjustments on an ad-hoc basis § Patient may not be able to be treated if the Rx is no longer the proper dose § Cannot return incorrect Rx – leading to increased costs in the event of patient noshows or dispensing errors Patient

Observations on “Buy and Bill” versus “External Delivery” distribution models • • Both Buy and Bill and External Delivery have a place within the current system Both models are likely to continue to thrive as the specialty market grows Buy and bill is viewed by providers as: – Offering the most efficiency and patient-centered flexibility – Being particularly well-suited for specialty medications where dosage may need to be adjusted for the patient on the day of treatment External delivery can offer benefits to providers as well, particularly small providers who prefer not to bear the cost of carrying high-cost specialty medications in inventory Copyright © 2019 by the HDA Research Foundation

The 340 B Drug Pricing programs provides access to drugs at discounted prices for the indigent or uninsured Federal 340 B Pricing Program • Introduced in 1992, with the goal of expanding drug access to indigent or uninsured patient populations at reduced costs • Requires pharmaceutical manufacturers to provide outpatient drugs, including specialty pharmaceuticals to qualifying providers /entities at significantly discounted prices • Qualifying parties may be: – “Qualifying eligible providers” - individual providers – “Covered Entities” – hospitals, clinics, long-term care facilities or other healthcare provider business entities • To qualify, a provider or entity must demonstrate that it provides a significant amount of care to indigent or uninsured patient populations Copyright © 2019 by the HDA Research Foundation

340 B Eligible Entities Federal Grantees Comprehensive Hemophilia Treatment Centers Federally Qualified Health Centers/Look-Alikes Urban/638 Health Center Ryan White HIV/AIDS programs Sexually Transmitted Disease/Tuberculosis Clinics Title X Family Planning Clinics Black Lung Clinics Native Hawaiian Health Centers Tribal/Urban Indian Health Centers Hospital Types Disproportionate Share Hospitals Children’s Hospitals* Critical Access Hospitals* Freestanding Cancer Hospitals* Rural Referral Centers* Sole Community Hospitals* Source: “ 340 B Eligibility, ” Health Resources and Services Administration (HRSA), last modified May 2018, https: //www. hrsa. gov/opa/eligibility-andregistration/index. html. *340 B Eligible via Section 7101 of the ACA Copyright © 2019 by the HDA Research Foundation
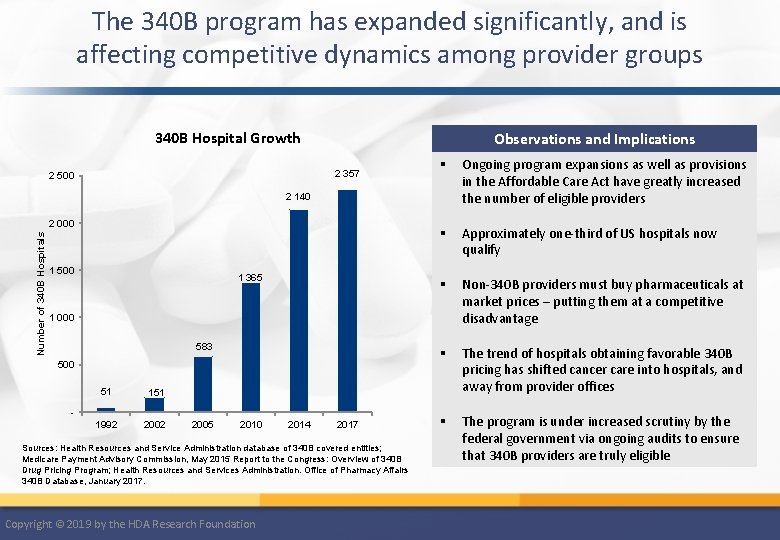
The 340 B program has expanded significantly, and is affecting competitive dynamics among provider groups 340 B Hospital Growth Observations and Implications 2 357 2 500 § Ongoing program expansions as well as provisions in the Affordable Care Act have greatly increased the number of eligible providers § Approximately one-third of US hospitals now qualify § Non-340 B providers must buy pharmaceuticals at market prices – putting them at a competitive disadvantage § The trend of hospitals obtaining favorable 340 B pricing has shifted cancer care into hospitals, and away from provider offices § The program is under increased scrutiny by the federal government via ongoing audits to ensure that 340 B providers are truly eligible 2 140 Number of 340 B Hospitals 2 000 1 500 1 365 1 000 583 500 51 1992 2005 2010 2014 2017 Sources: Health Resources and Service Administration database of 340 B covered entities; Medicare Payment Advisory Commission, May 2015 Report to the Congress: Overview of 340 B Drug Pricing Program; Health Resources and Services Administration. Office of Pharmacy Affairs 340 B Database, January 2017. Copyright © 2019 by the HDA Research Foundation

Specialty Pharmaceuticals 101 US Specialty Pharmaceutical Supply Chain Overview Manufacturer - Distributor Partnership and Operations Distributor – Provider Partnership and Operations Payer Influence on the Supply Chain Regulatory Influence Copyright © 2019 by the HDA Research Foundation

The FDA requires that manufacturers develop a Risk Evaluation and Mitigation Strategy (REMS) for certain drugs • • • The Food and Drug Administration Amendments Act of 2007 gave FDA the authority to require a Risk Evaluation and Mitigation Strategy (REMS) from manufacturers REMS are “required risk management plans that use risk mitigation strategies beyond professional labeling to ensure that the benefits of a drug or biological product outweigh its risks” REMS programs vary in complexity, depending on the drug, but can be comprised of one or more of the following components • • Medication Guide – package insert with specific, FDA approved information and guidance for patient and provider Communication Plan – to distribute safety information to providers and patients Elements to Ensure Safe Use – customized program which may in include patient registries, provider registries, and verification of certain patient conditions Implementation System – plan to certify, train and/or audit REMS program execution on an ongoing basis Copyright © 2019 by the HDA Research Foundation

REMS programs vary in complexity Approved REMS Programs by Level of Complexity July 2014 – 74 drugs § 20 15 10 5 0 MG = Medication Guide § CP = Communication Plan § ETASU = Elements to Assure Safe Use § IS = Implementation System 19 16 9 7 MG ONLY CP ONLY MG + CP 11 5 MG + ETASU CP + ETASU 4 3 1 ETASU + IS CP + ETASU + IS MG + CP + ETASU + IS Simple Complex Simple § Medication guides § Specific package insert language § Communication plan to disseminate safety information § “Dear healthcare providers” letters (DHCP) § Educational material packages Moderate Level 1 plus the following: § Prescriber training / certification § Pharmacist verification of lab test results prior to dispensing § Patient monitoring § Registration of prescribers § Patient enrollment into Registries § Patient acknowledgement forms (e. g. , Acknowledgement of Risks) Complex Level 2 plus the following: § Certification and training of pharmacists and physicians § Recertification and product release approval at each dispense § Regular audits of prescribers and dispense locations § Implementation systems § Authorization codes § Limited access to registered customers Note: This captures the ONLY the elements of approved REMS programs – as listed by FDA. gov – products may also have medication guides, which are not associated with the REMS; Includes Individual REMS and Shared System REMS Copyright © 2019 by the HDA Research Foundation

HR 3204, The Drug Quality and Security Act is intended to create a national, unit level traceability system for pharmaceutical products Highlights of the Rule: • Voted into law in 2013 – a national standard law on traceability, which will supersede prior state-specific regulations (e. g. California e. Pedigree) • Requires that manufacturer products are uniquely identifiable at the package level, allowing for traceability • Establishes standards for the exchange of transaction documentation, which shall include transaction information, transaction history, and transaction statements • Requires that manufacturers, wholesale distributors, dispensers and repackagers ensure that all prior transaction information is provided at each transfer of ownership. • Requires manufacturers to provide the Secretary of Health and Human services with all applicable transaction documentation within a “reasonable” amount of time in the event of a recall Implications for stakeholders: • The emergence of a national law sets the foundation for a common, standard way to manage traceability, rather than a fragmented web of unique state requirements • In coming years, Manufacturers, Distributors and providers will need to invest to build the capabilities required to meet the new requirements Copyright © 2019 by the HDA Research Foundation

Glossary Copyright © 2019 by the HDA Research Foundation 59

Glossary of Terms These terms are used in the pharmaceutical distribution industry and may be found in the text of the 89 th Edition HDA Factbook (2018 -2019) or in 2018 Specialty Distribution Facts Figures and Trends. Words or phrases defined uniquely by the cited source are listed as table notes. 3 PL ACA ANDA API ASN ASP AWP Chain Drug Stores Chargeback Cost of Goods Sold CPFR CRM DC DEA Disease Management DTC EC EDI EMR Third-party Logistics The Patient Protection and Affordable Care Act, sometimes referred to as “Health Reform” or “Obamacare” Abbreviated New Drug Application Active Pharmaceutical Ingredient Advance Ship Notice Average Sales Price Average Wholesale Price Drug stores with four or more stores primarily in the business of retail sales of pharmaceuticals and OTC products The amount a distributor bills back to a manufacturer when a product is sold to a customer at a contract price that is less than the distributor’s cost [Beginning inventory + purchases (net of trade discounts & returns, freight, value of inventory shrinkage and sales or use taxes) ending inventory] Collaborative Planning Forecasting and Replenishment — frequent replenishment of product based on actual and forecasted demand Customer Relationship Management Distribution center Drug Enforcement Administration (U. S. ) A system of coordinated healthcare interventions and communications for patient populations with specific medical conditions that involve components of self-management Direct to Consumer — pharmaceutical advertising directed to the patient, rather than to the healthcare professional Electronic Commerce Electronic Data Interchange Electronic Medical Records Copyright © 2019 by the HDA Research Foundation

Glossary of Terms (cont’d) FDA FIFO FTE’s GDP GPO HBC HHC HMO HPC IMA Institutional Customers Independent Drug Stores Inside Sales Force ISD Food and Drug Administration (U. S. ) First In First Out — a method of valuing the cost of goods sold that uses the cost of the oldest items in inventory first Full-Time Employees, where the proportion of full-time hours worked is reported for those who do not work full-time Gross Domestic Product Group Purchasing Organization — negotiates buying contracts for healthcare products at discounted fees for hospitals and health plans Health and Beauty Care – products that do not treat a therapeutic class, (e. g. , toiletries and cosmetics) Home Healthcare products Health Maintenance Organization — provides, offers or arranges coverage of health services for a fixed, pre -paid premium Health and Personal Care (used interchangeably with HBC) Inventory Management Agreement Customers who purchase healthcare products without the primary intention of reselling them, including hospitals, nursing homes, prisons or institutions Drug stores with no more than one to three locations, and are not part of a larger chain Sales staff that work customer accounts from an office where calls are received for products or services Insufficient Data Copyright © 2019 by the HDA Research Foundation

Glossary of Terms (cont’d) NA NDC Order Fill Rate OTC PBM R&D Returns RFID Saleable Scorecarding SKU Specialty Distribution Specialty Pharmacy Not Applicable New Drug Application National Drug Code The percent of both total sales invoice lines and total sales dollars ordered that are shipped without error or backorder Over-the-Counter Pharmacy Benefit Management Research and Development Products unsaleable at the retail level (damaged, outdated, overstocked, etc. ) returned to a manufacturer or distributor for credit Radiofrequency Identification Product that may be sold or re-sold and re-placed into inventory An evaluation that assesses the order fulfillment process between trading partners Stock Keeping Unit — code that identifies each unique product form Handles specialty products that are generally defined as: All biologics except vaccines and insulin; and three or more of the following: typically high cost ($600 or more per month); require special handling, storage and delivery; dispensed to treat individuals with chronic or rare diseases; have complex treatment regimes that require ongoing clinical monitoring and patient education; frequently have limited or exclusive product availability and distribution Typically a pharmacy function that focuses on dispensing high-cost, high maintenance specialty drug products that treat chronic diseases; These products are nearly always biotech products and are often associated with complex reimbursement issues, require specialized shipping and handling, and require disease state management tools for patients to maximize therapeutic benefit and ensure patient compliance Copyright © 2019 by the HDA Research Foundation

Glossary of Terms (cont’d) Unit Dose Unit of Use Unsaleable VMI WAC The package level for a single dose of therapy The package level for one course of therapy Healthcare products retuned to distributors or manufacturers which cannot be sold, or re-placed into inventory and re-sold Vendor Managed Inventory — replenishment is managed by the supplier based on the trading partner agreement Wholesale Acquisition Cost Copyright © 2019 by the HDA Research Foundation
- Slides: 64