Understanding Radiation Dr Jeremy Whitlock Canadian Nuclear Laboratories

Understanding Radiation Dr. Jeremy Whitlock Canadian Nuclear Laboratories jeremy. whitlock@cnl. ca
CHALK RIVER LABS Chalk River Laboratories 2

Outline � What is radiation? � Natural background radiation � Man-made sources of radiation � Effects of radiation � Medical uses of radiation � Nuclear fuel cycle 3

What is radiation? � Possibly the most misunderstood element of our natural world 4

He’s grown a foot since I saw him last… 5

“Radiation” is an important part of our world � It is energy flowing through the environment. � It is how we see the world around us � It is how we sense heat � It is largely how we communicate with each other � It is how we cook our food � It is the life-giving energy from our sun � It is how we kill cancer and fight disease � It is natural energy 6

Radiation is everywhere Low Energy High Energy The Electromagnetic Spectrum 7
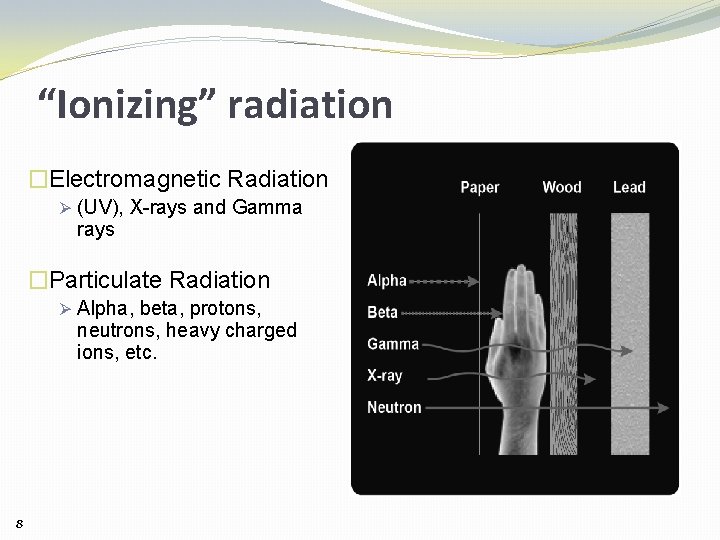
“Ionizing” radiation �Electromagnetic Radiation Ø (UV), X-rays and Gamma rays �Particulate Radiation Ø Alpha, beta, protons, neutrons, heavy charged ions, etc. 8

“Nuclear” ionizing radiation “Alpha” particle Uranium atom (ionizing radiation) (actually its “nucleus”) 12 million uranium atoms disappear this way every second in 1 kg of Uranium Thorium atom 9 (12 million uranium atoms = 0. 00000005 grams of uranium)

Measuring radiation � Radioactive Decay: 1 Bequerel (1 Bq) = 1 decay per second � “Absorbed Dose”: Energy deposited per kg of material: “Gray” (Gy) � “Equivalent Dose”: Factor in biological effect of different types of radiation: “Sievert” (Sv) Ø 1 Sv of neutrons = 1 Sv of alpha particles = 1 Sv of gammas � Average natural background radiation dose: around 0. 002 Sv per year (or 2 “millisievert”, 2 m. Sv) � Public annual limit from nuclear operations: 1 m. Sv 10 (excludes medical)

Regulating radiation � Canadian Nuclear Safety Commission (2000 – replaced Atomic Energy Control Board est. 1946) � Regulates the use of nuclear energy and materials to protect health, safety, security and the environment � Implements Canada's international commitments on the peaceful use of nuclear energy � Disseminates objective scientific, technical and regulatory information to the public. � Independent tribunal - arms length from gov’t; no ties to industry nuclearsafety. gc. ca 11
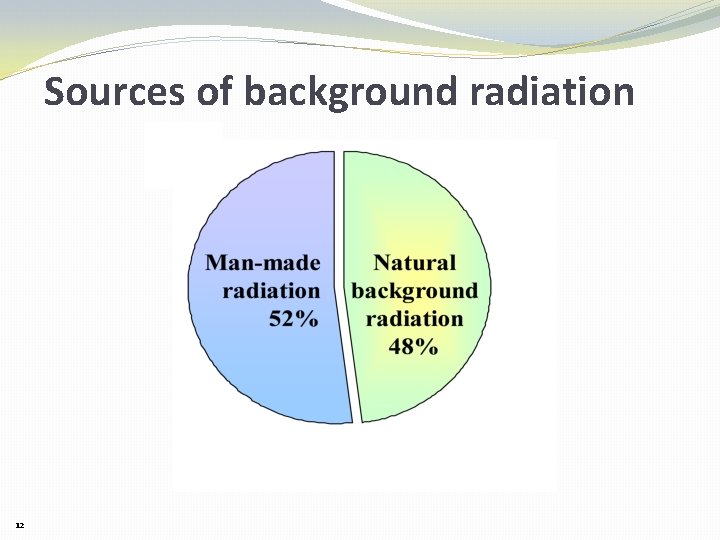
Sources of background radiation 12

Natural background contributes about 50% of lifetime radiation exposure 10% Cosmic Radiation 90% Terrestrial • 70% Radon • 10% External • 10% Internal 13
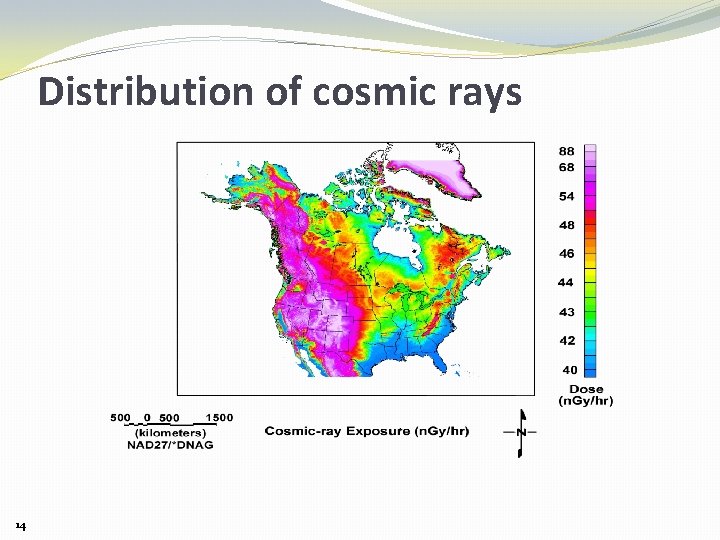
Distribution of cosmic rays 14
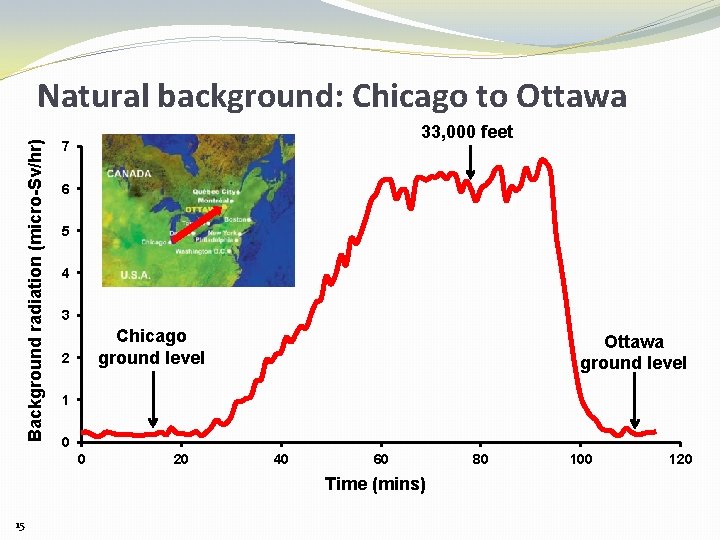
Background radiation (micro-Sv/hr) Natural background: Chicago to Ottawa 33, 000 feet 7 6 5 4 3 Chicago ground level 2 Ottawa ground level 1 0 0 20 40 60 Time (mins) 15 80 100 120

Your natural background radiation dose depends on where you are Geographic Location Guarapari Beach, Brazil Ramsar, Iran SW France Kerala Beach, India Sweden Evacuation threshold – Fukushima U. S. Rocky Mountain States Evacuated land near Chernobyl World Average U. S. Gulf States 16 Annual Dose (m. Sv) Up to 800 Up to 700 Up to 88 Up to 35 Up to 18 20 6 -12 6 2. 4 0. 8 -1. 2

Ramsar Iran: 200 -700 m. Sv/yr 17
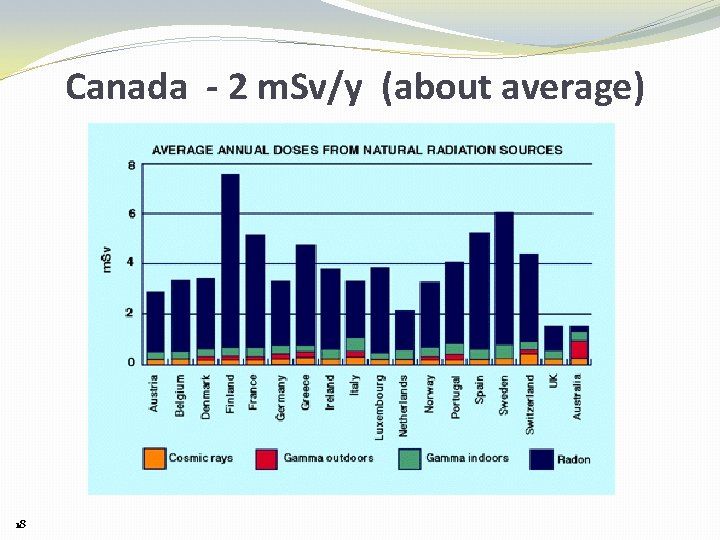
Canada - 2 m. Sv/y (about average) 18

Some buildings are more radioactive than others… Grand Central Station 1. 2 m. Sv/a 19

Some buildings are more radioactive than others… U. S. Capitol Approx. 1 m. Sv/a to workers 20

Low dose radioactivity in food Water Tritium – Naturally 1 -7 Bq/L (Health Canada “drinking water standard: 7500 Bq/L) Banana Smoothie Potassium – 40 100 -150 Bq/L Veggie Shake Carbon – 14 21 200 -300 Bq/L *Bq is a decay per second
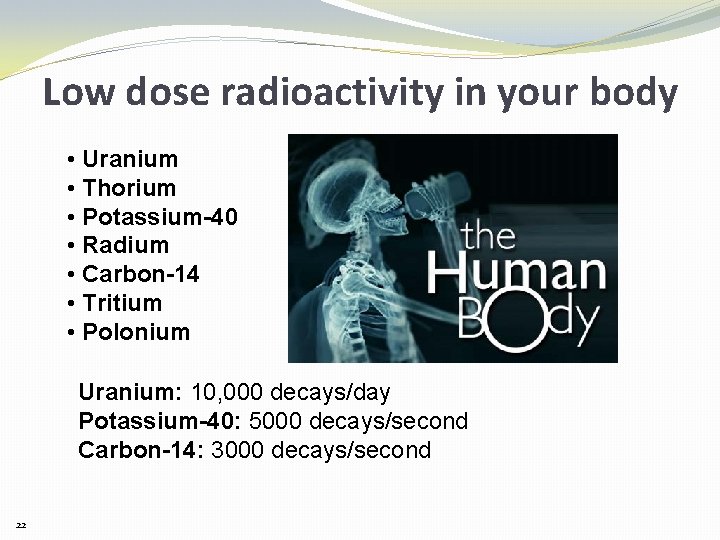
Low dose radioactivity in your body • Uranium • Thorium • Potassium-40 • Radium • Carbon-14 • Tritium • Polonium Uranium: 10, 000 decays/day Potassium-40: 5000 decays/second Carbon-14: 3000 decays/second 22
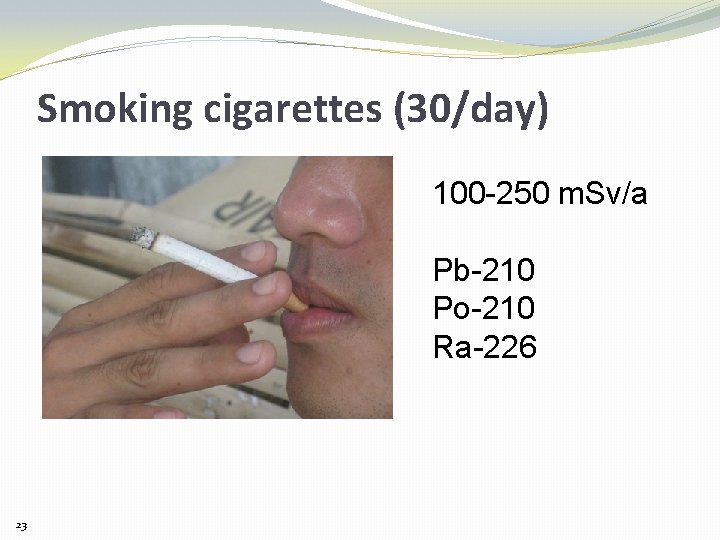
Smoking cigarettes (30/day) 100 -250 m. Sv/a Pb-210 Po-210 Ra-226 23

Uranium decay – it doesn’t stop at Thorium. . . “Alpha” particle (ionizing radiation) Uranium atom 12 million uranium atoms disappear this way every second in 1 kg of Uranium Thorium atom ? 24 (12 million uranium atoms = 0. 00000005 grams of uranium)
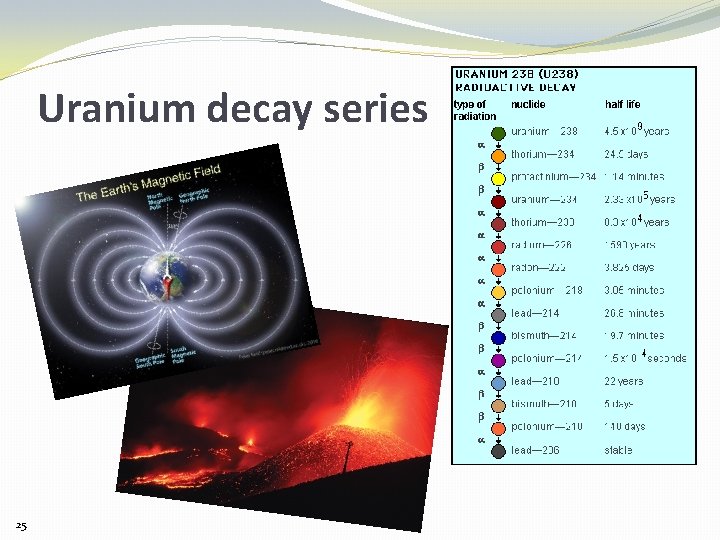
Uranium decay series 25
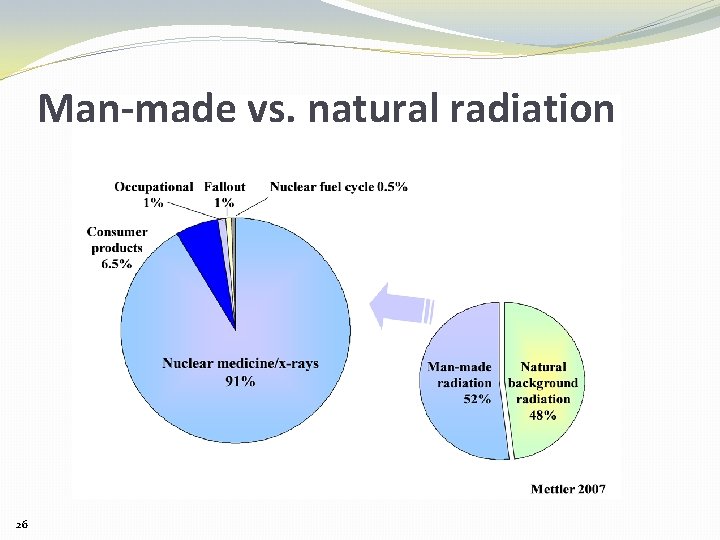
Man-made vs. natural radiation 26
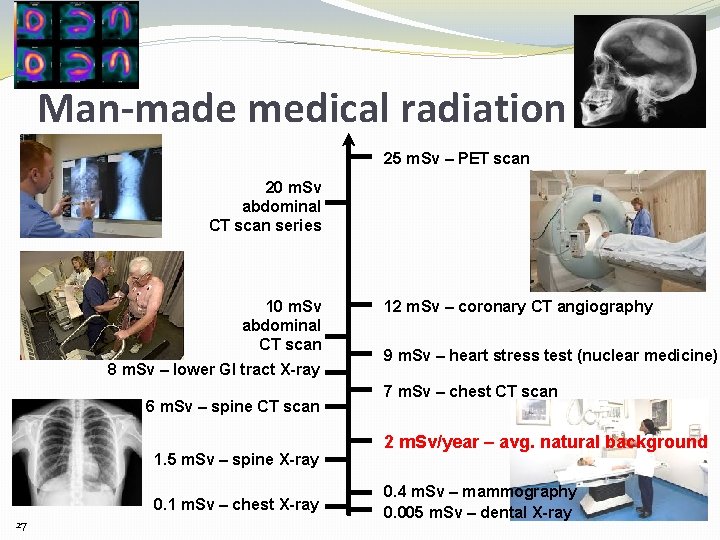
Man-made medical radiation 25 m. Sv – PET scan 20 m. Sv abdominal CT scan series 10 m. Sv abdominal CT scan 8 m. Sv – lower GI tract X-ray 6 m. Sv – spine CT scan 1. 5 m. Sv – spine X-ray 0. 1 m. Sv – chest X-ray 27 12 m. Sv – coronary CT angiography 9 m. Sv – heart stress test (nuclear medicine) 7 m. Sv – chest CT scan 2 m. Sv/year – avg. natural background 0. 4 m. Sv – mammography 0. 005 m. Sv – dental X-ray
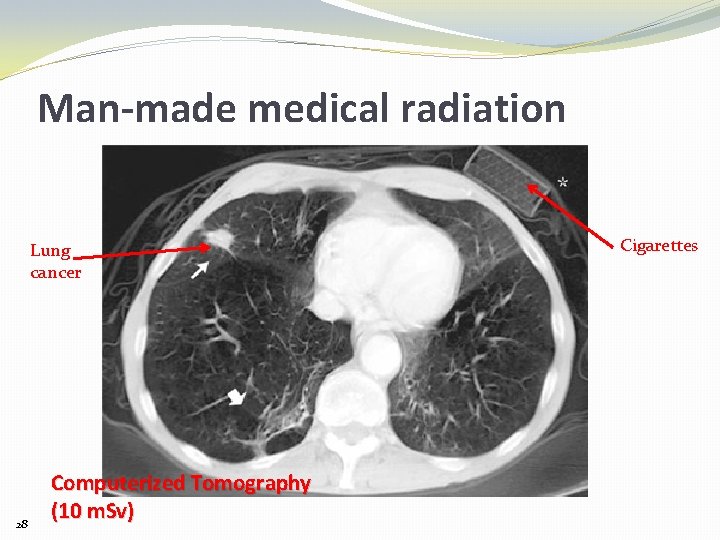
Man-made medical radiation Lung cancer 28 Computerized Tomography (10 m. Sv) Cigarettes

Same Dose Rate per Hour (0. 004 m. Sv/h) Handling fresh nuclear fuel Flying = 1 metre 30, 000 ft (500 hours at either activity would give your annual background dose) 29
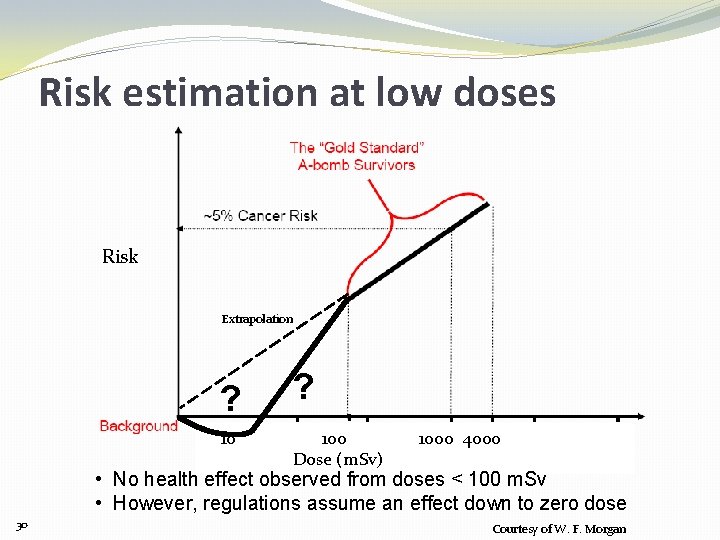
Risk estimation at low doses Risk Extrapolation ? 100 Dose (m. Sv) 1000 4000 • No health effect observed from doses < 100 m. Sv • However, regulations assume an effect down to zero dose 30 Courtesy of W. F. Morgan
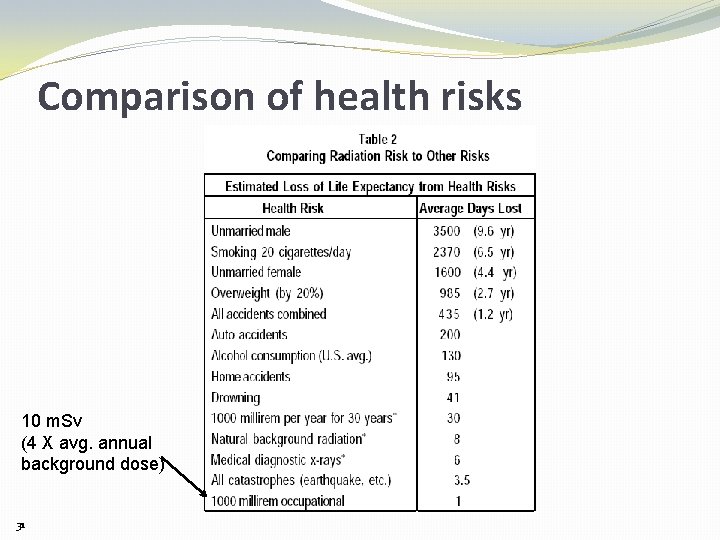
Comparison of health risks 10 m. Sv (4 X avg. annual background dose) 31

Radiation from cobalt-60: a weapon to fight cancer since 1951 Harold Johns 1951 University of Saskatchewan 32

Designs have changed since then… Gamma “knife” 33

A Canadian Gift to the World: Cancer Therapy and Nuclear Medicine 34
NRX reactor (1947 -1995) NRU reactor (1957 -present) Chalk River Laboratories 35

NRU Research Reactor (CNL Chalk River) 36

The Nuclear Fuel Cycle 37
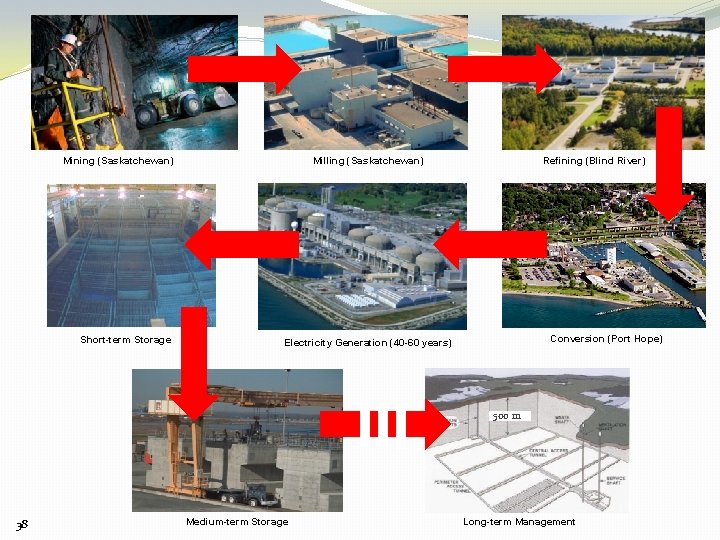
Mining (Saskatchewan) Short-term Storage Refining (Blind River) Milling (Saskatchewan) Conversion (Port Hope) Electricity Generation (40 -60 years) 500 m 38 Medium-term Storage Long-term Management
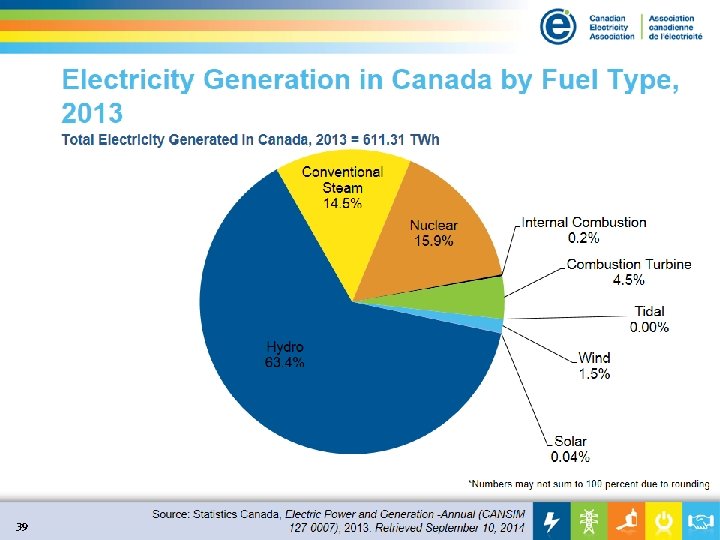
39
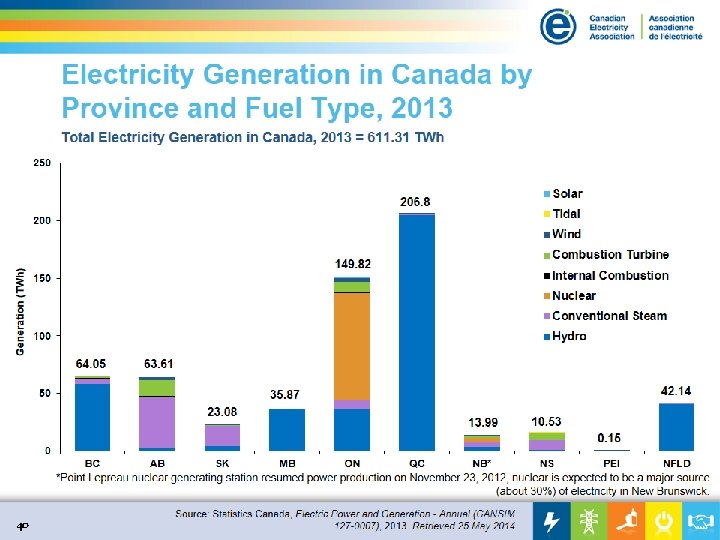
40
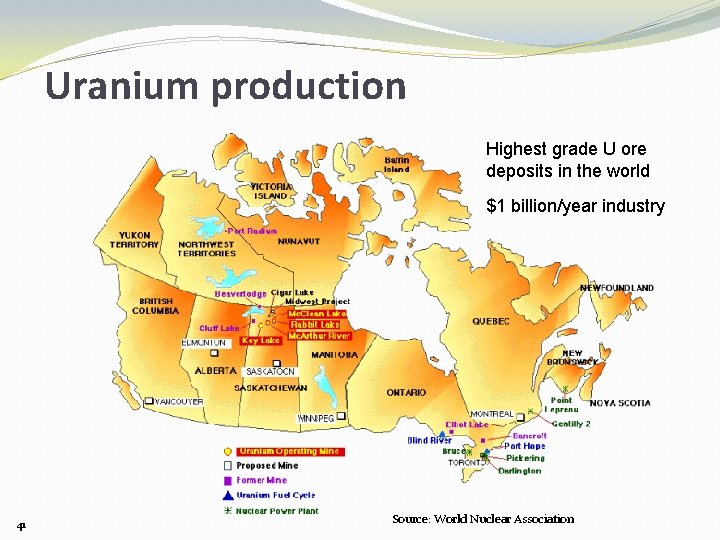
Uranium production Highest grade U ore deposits in the world $1 billion/year industry 41 Source: World Nuclear Association
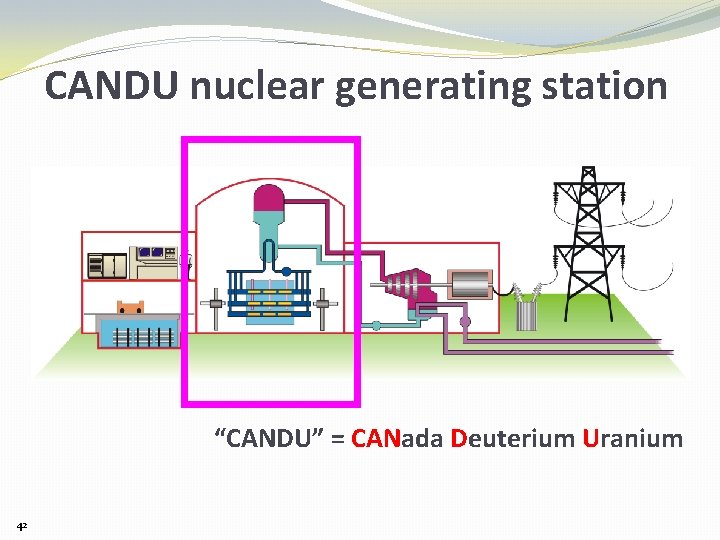
CANDU nuclear generating station “CANDU” = CANada Deuterium Uranium 42
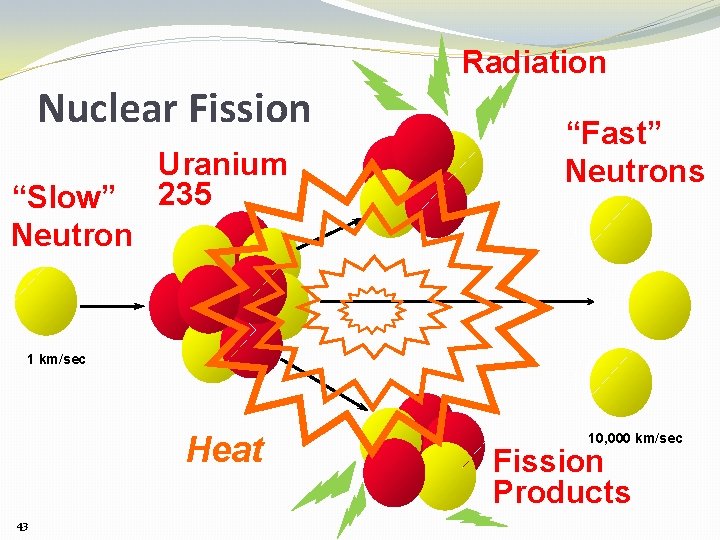
Nuclear Fission “Slow” Neutron Uranium 235 Radiation “Fast” Neutrons 1 km/sec Heat 43 10, 000 km/sec Fission Products
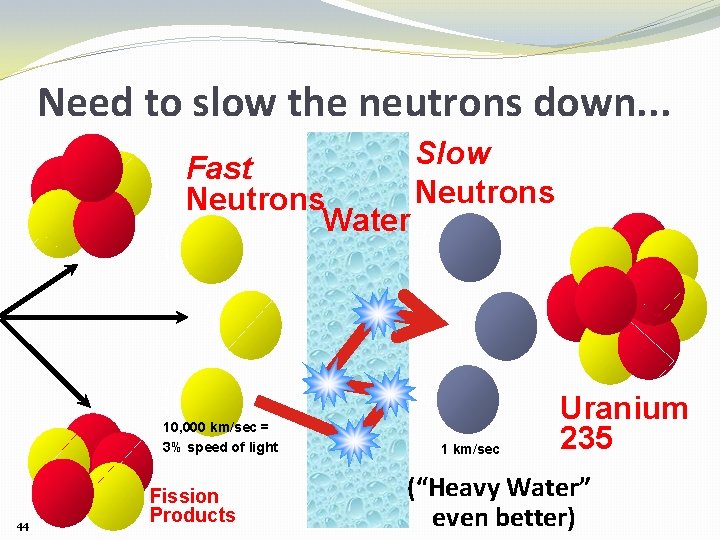
Need to slow the neutrons down. . . Slow Fast Neutrons Water 10, 000 km/sec = 3% speed of light 44 Fission Products 1 km/sec Uranium 235 (“Heavy Water” even better)

CANDU reactor core (“Calandria”) 45

Efficient use of resources = 1 CANDU fuel bundle (50 cm long, 23 kg) 46 Total electricity needs of a family for 100 years

Efficient use of resources 1 CANDU fuel bundle (50 cm long, 23 kg) 47 = 400 tonnes coal 1000 tonnes CO 2 1 tonne particulates 8 tonnes acid gas = 60, 000 gal oil = 10 million ft 3 natural gas 600 tonnes CO 2 1 tonne acid gas

Uranium fuel pellets Fuel Pellet: 8 of these provide one home’s electricity for a year 48
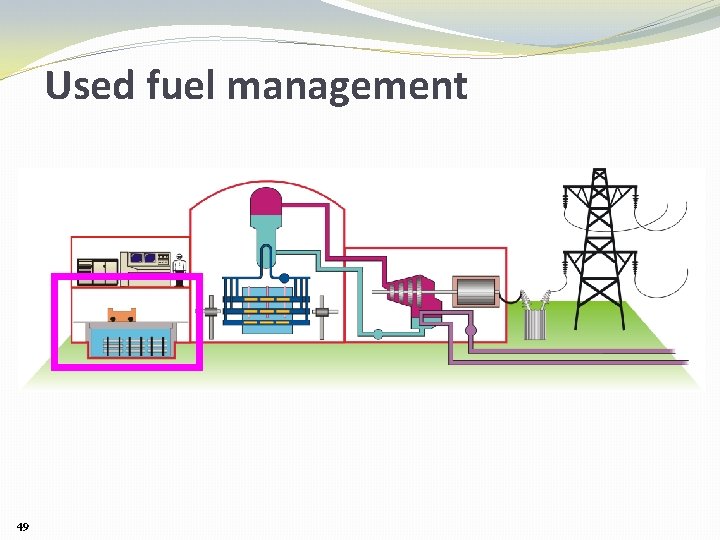
Used fuel management 49

Used fuel management SHORT TERM (< 10 years): water-filled bays 50

Used fuel management INTERMEDIATE TERM: Air-cooled dry storage 51

Used fuel management LONG-TERM: Deep Geologic Repository Canada’s “Adaptive Phased Management” Program for used nuclear fuel is managed by the Nuclear Waste Management Organization: www. nwmo. ca 52
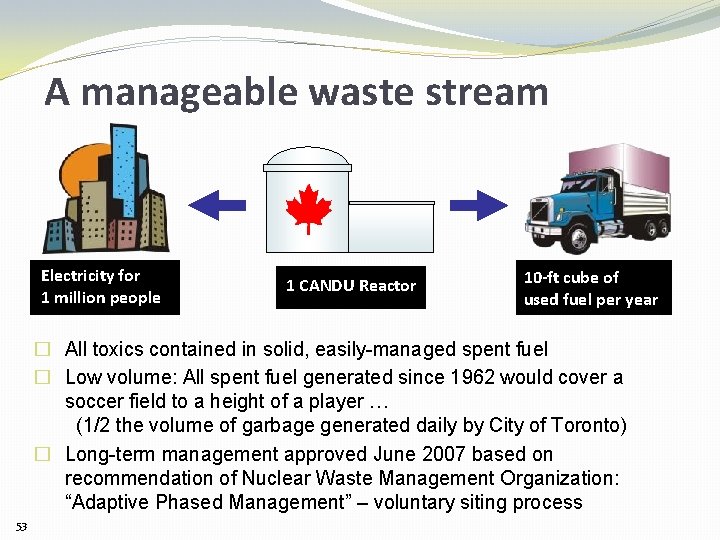
A manageable waste stream Electricity for 1 million people 1 CANDU Reactor 10 -ft cube of used fuel per year � All toxics contained in solid, easily-managed spent fuel � Low volume: All spent fuel generated since 1962 would cover a soccer field to a height of a player … (1/2 the volume of garbage generated daily by City of Toronto) � Long-term management approved June 2007 based on recommendation of Nuclear Waste Management Organization: “Adaptive Phased Management” – voluntary siting process 53
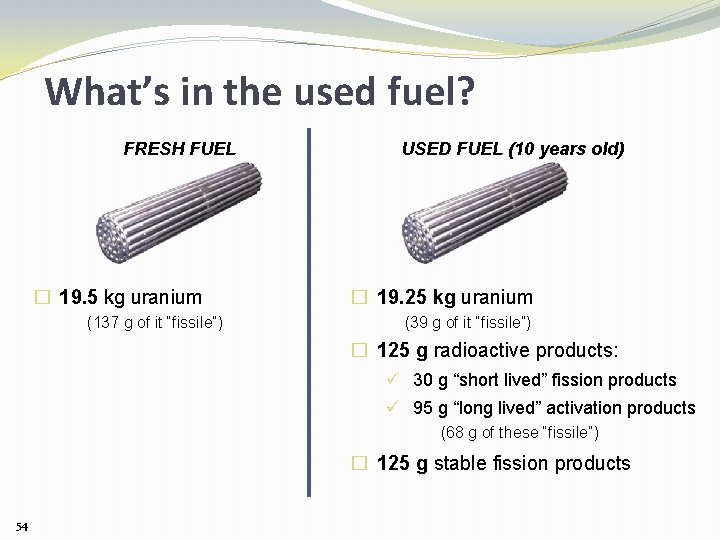
What’s in the used fuel? FRESH FUEL � 19. 5 kg uranium (137 g of it “fissile”) USED FUEL (10 years old) � 19. 25 kg uranium (39 g of it “fissile”) � 125 g radioactive products: ü 30 g “short lived” fission products ü 95 g “long lived” activation products (68 g of these “fissile”) � 125 g stable fission products 54

Decay of a used fuel bundle 54, 000 m. Sv/h 1, 150 m. Sv/h 360 m. Sv/h 37 m. Sv/h 1 55 100 200 0. 8 m. Sv/h 300 Years 400 500 0. 009 m. Sv/h 1, 000 Risk after ~500 years mainly from internal exposure (ingestion, inhalation, absorption)
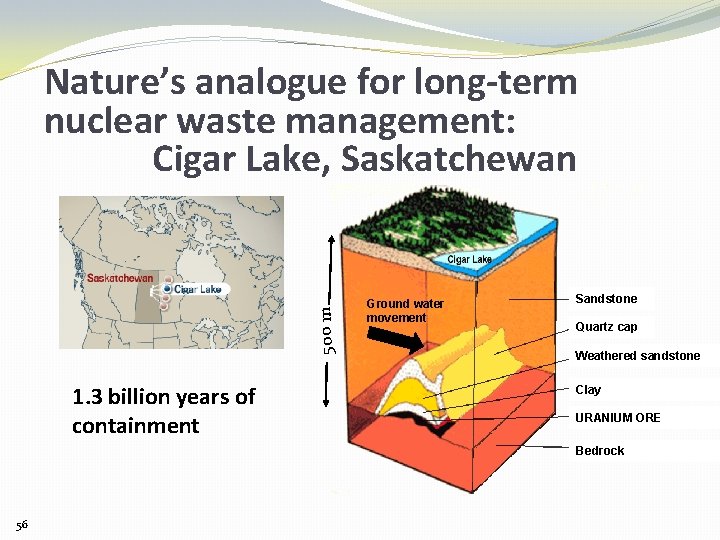
500 m Nature’s analogue for long-term nuclear waste management: Cigar Lake, Saskatchewan 1. 3 billion years of containment Ground water movement Sandstone Quartz cap Weathered sandstone Clay URANIUM ORE Bedrock 56

Questions? Dr. Jeremy Whitlock Canadian Nuclear Laboratories jeremy. whitlock@cnl. ca “The Canadian Nuclear FAQ”: www. nuclearfaq. ca
- Slides: 57