Understanding Pressure and What It Has To Do










- Slides: 16

Understanding Pressure and What It Has To Do With the Atmosphere Activity: Hot air balloon: Activity: Molecules in a box Come inside and review observations.

Molecules of a gas behave in a very simple manner. They move in a straight line and at a high speed, until they bump into something.

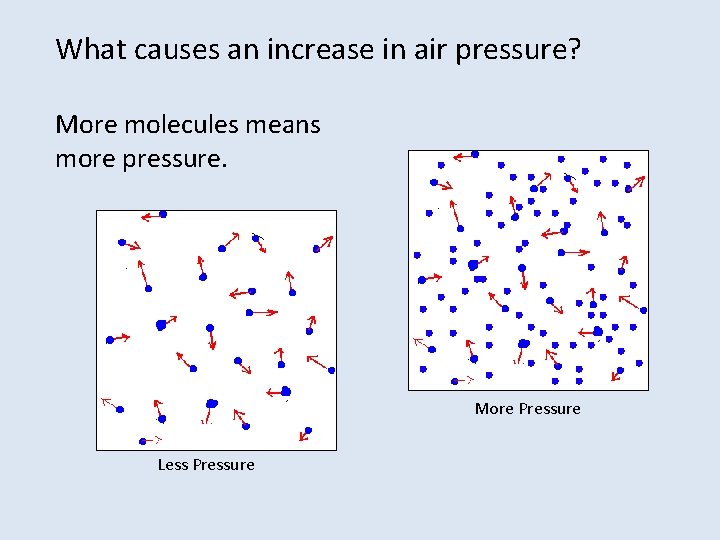
What causes an increase in air pressure? More molecules means more pressure. More Pressure Less Pressure

What causes an increase in air pressure? More molecules means more pressure. Higher temperature (meaning faster molecules!) means more pressure. HOT HOT

What causes an increase in air pressure? More molecules means more pressure. Higher temperature (meaning faster molecules!) means more pressure. A smaller container means more pressure.

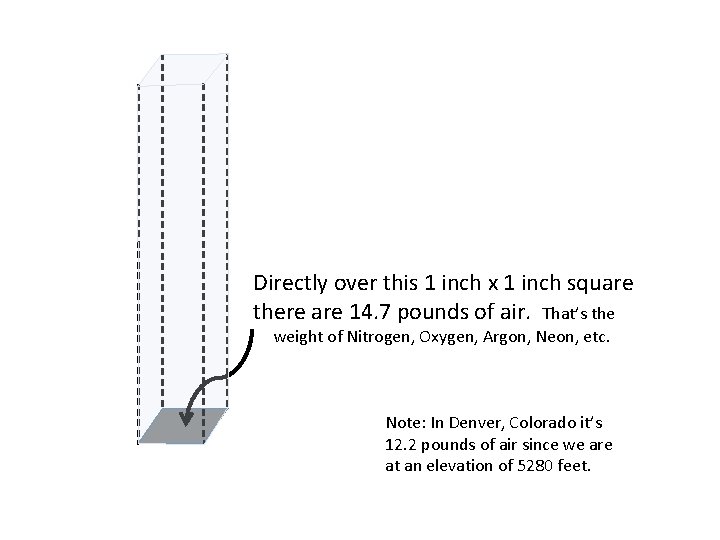
Directly over this 1 inch x 1 inch square there are 14. 7 pounds of air. That’s the weight of Nitrogen, Oxygen, Argon, Neon, etc. Note: In Denver, Colorado it’s 12. 2 pounds of air since we are at an elevation of 5280 feet.

Demonstrations: 1) Blowing a stream of air over the top of notebook paper 2) Drinking glass with water and index card

• Pressure is defined as a force applied per unit area • The weight of air is a force, equal to the mass m times the acceleration due to gravity g • Molecules bumping into an object also create a force on that object, or on one another • Air pressure results from the weight of the entire overlying column of air! Slide from: Scott Denning, 2007 Atmospheric Science Class

How do we measure pressure? Why does pressure decrease with altitude? Remember: Pressure = mass*gravity/unit area As you go higher, you have less mass above you. Slide from: Scott Denning, 2007 Atmospheric Science Class

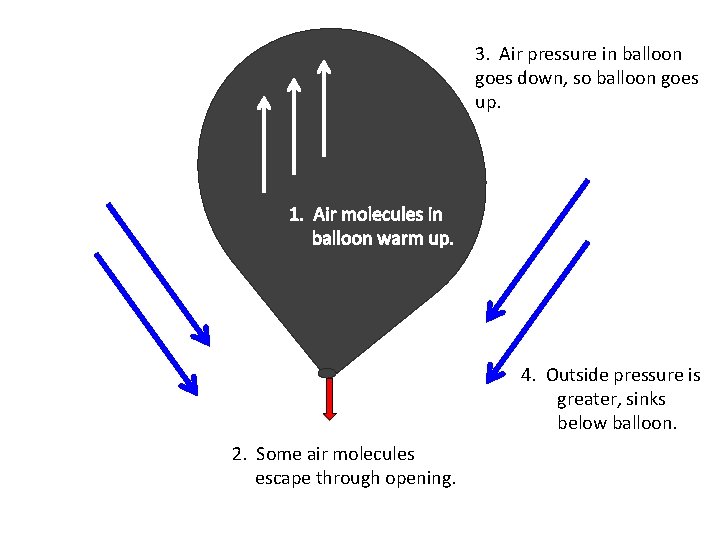
Back to the Hot Air Balloon… Balloon goes up What happens when the balloon goes up?

3. Air pressure in balloon goes down, so balloon goes up. 1. Air molecules in balloon warm up. 4. Outside pressure is greater, sinks below balloon. 2. Some air molecules escape through opening.

Can you explain what’s happening with a Weather Balloon, filled with helium with no opening at the bottom?

Atmospheric Measurements Helium-filled weather balloons are released from over 1000 locations around the world every 12 hours. Balloons carry Radiosondes high into the atmosphere. These instruments document temperature, pressure, humidity, and winds aloft as they ascend. Balloons are only partially filled with helium so as they ascend to about 25 miles above the earth they expand as they rise and get 24 - 32 feet across before they burst. Slide modified from: Scott Denning, 2007 Atmospheric Science Class

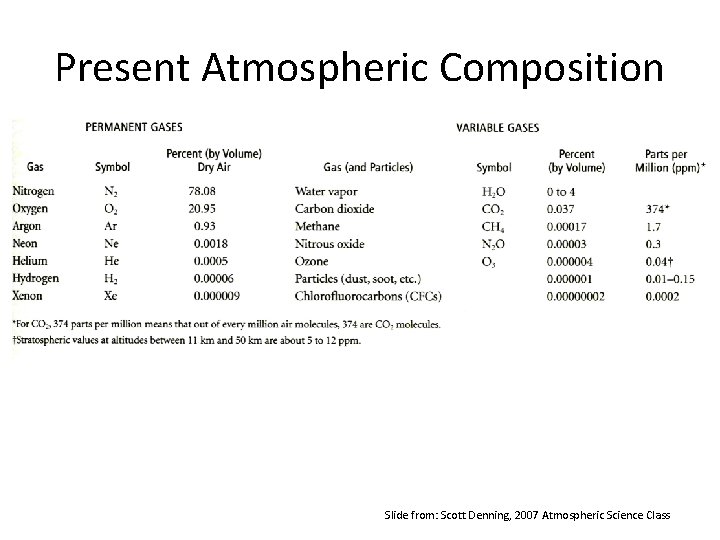
Present Atmospheric Composition Slide from: Scott Denning, 2007 Atmospheric Science Class

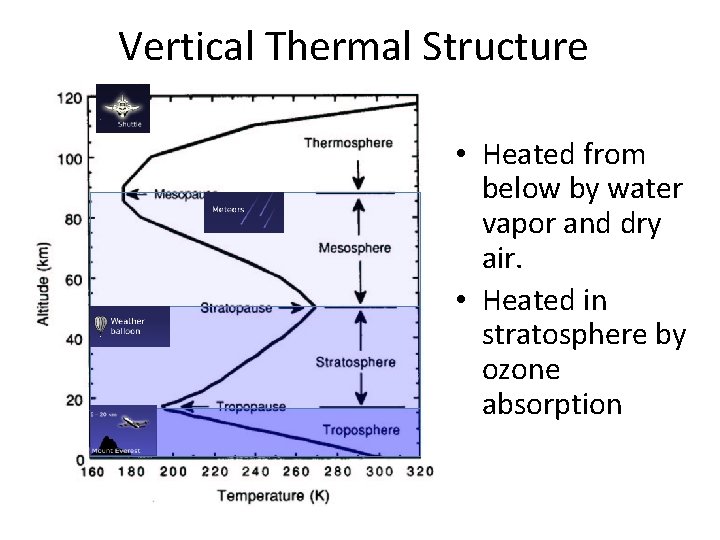
Vertical Thermal Structure • Heated from below by water vapor and dry air. • Heated in stratosphere by ozone absorption
