Understanding FA Revenue Accounts Receivable Fiscal Closing UCLA

Understanding F&A, Revenue Accounts Receivable, Fiscal Closing UCLA Department of Medicine Office of Research Administration

Facilities & Administrative Costs (F&A) Basics UCLA Department of Medicine Office of Research Administration

What is F&A? • Direct Cost: Any cost that can be specifically identified with a particular project, program, or activity or that can be directly assigned to such activities relatively easily and with a high degree of accuracy. • F&A Costs: Costs for common or joint objectives that cannot be readily identified with an individual project, program, or organizational activity. UCLA Department of Medicine Office of Research Administration

F&A Costs are otherwise known as… • Overhead Costs • Indirect Costs • Sub 9 H – at UCLA only UCLA Department of Medicine Office of Research Administration

Types of F&A Bases UCLA Department of Medicine Office of Research Administration

Modified Total Direct Cost (MTDC) Base • MTDC component is the base against which the federally negotiated rates are applied, and is derived by excluding certain costs from the Direct Cost total. • MTDC exclusions include: • • • Equipment Patient Care Alterations/Renovations Space Rental Tuition Remission/Fees For Non-UC subawards, any amount beyond the first $25, 000 TC of each subaward. • The total cost (TC) of subawards to other UC campuses UCLA Department of Medicine Office of Research Administration

Total Direct Cost (TDC) Base • F&A calculated on the total of all direct costs of a project. There are no exclusions. • Most non-federal F&A rates are based on Total Direct Cost (TDC), not MTDC. • e. g. Pharm sponsored Clinical Trials (26% TDC), Non-profit Foundations, etc. UCLA Department of Medicine Office of Research Administration

What is the “Current F&A Base Code”? • How do I find it? • Summary-By-Sub/Overhead Rate field • COP/Approp & Financial tab UCLA Department of Medicine Office of Research Administration
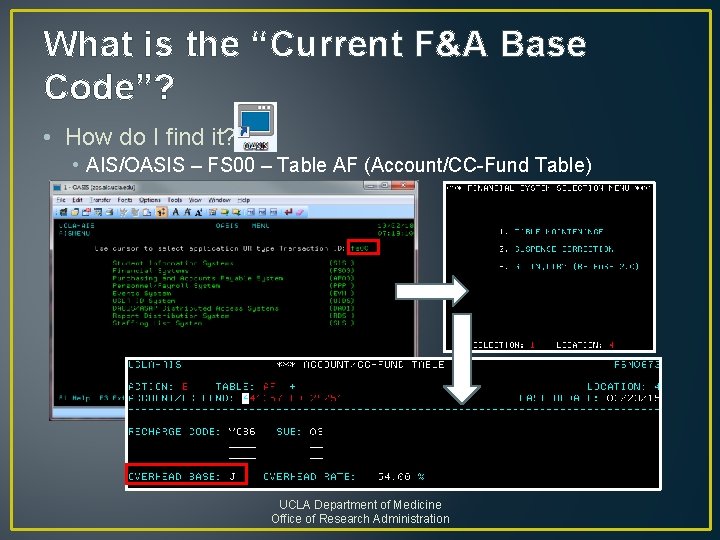
What is the “Current F&A Base Code”? • How do I find it? • AIS/OASIS – FS 00 – Table AF (Account/CC-Fund Table) UCLA Department of Medicine Office of Research Administration

What is the “Current F&A Base Code”? • What do the letters mean (i. e. J, B, C)? • Base & Object code Table for Indirect costs • Used for F&A cost inclusion and exclusion in calculations of indirect costs for contract and grant projects UCLA Department of Medicine Office of Research Administration

Current F&A Rates & Cost Returns UCLA Department of Medicine Office of Research Administration
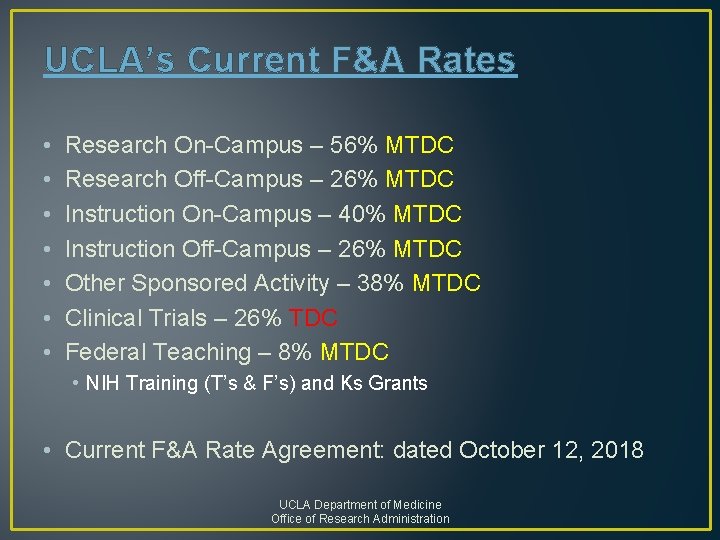
UCLA’s Current F&A Rates • • Research On-Campus – 56% MTDC Research Off-Campus – 26% MTDC Instruction On-Campus – 40% MTDC Instruction Off-Campus – 26% MTDC Other Sponsored Activity – 38% MTDC Clinical Trials – 26% TDC Federal Teaching – 8% MTDC • NIH Training (T’s & F’s) and Ks Grants • Current F&A Rate Agreement: dated October 12, 2018 UCLA Department of Medicine Office of Research Administration

Annual Indirect Cost Return* • What is it? • What is it based on? Actual expenses from previous fiscal year! • Usually receive ~Jan-Mar • New DOM Fund 07427 & 75014. Handled by MSO. • *Based on DOM. Every School may handle differently. UCLA Department of Medicine Office of Research Administration

F&A Reconciliation UCLA Department of Medicine Office of Research Administration

Why Does F&A Need to be Reconciled? • Unbudgeted changes can occur: • PI spent the $ on an item that is excluded from F&A, but was not originally budgeted. EXAMPLE: Unbudgeted equipment. See Scenario 1 • PI did NOT spend the $ allocated to an excluded item, but rather, spent it on an item NOT excluded from F&A. Example: Budgeted equipment. See Scenario 2 • F&A rate changed during the project period, but may not have been changed in the UCLA financial system. Use F&A Reconciliation Tool • PI moved from on-campus to off-campus space, or vice-versa. Use F&A Reconciliation Tool • Item was not correctly Object Coded in the UCLA financial system. Use F&A Reconciliation Tool UCLA Department of Medicine Office of Research Administration

Two F&A Tools Available • 1) FPM/QDB F&A Reconciliation Tool • This report is also run automatically in the Smart Closeout Tool/PAMS • 2) 9 H Reconciliation Adjustment note located at the bottom of the FPM/QDB Summary by Sub Report • Run both reports frequently throughout the life of the award. • Do NOT assume the. UCLA reports correct. Do some Departmentare of Medicine Office of Research Administration research to determine WHY before making any

Two Type of F&A Reconciliation: Appropriations & Expenditures • Appropriations • Which Tool Do I Use? • Summary-By-Sub report* for the 9 H reconciliation note located at the bottom of the report. • Why does this happen? • Actual expenditures differ from budget. • Be sure to research and understand what happened prior to making adjustment • How does it get adjusted/fixed? • FM can prepare a TOF *Be sure to always enter the full FAU into QDB. Do not enter an “*” in the Account of Cost Center field. This will return incorrect results. • Expenditures • Which Tool Do I Use? • F&A Reconciliation report in QDB • Why does this happen? • Incorrect F&A Rate was charged to expenditures • How does it get adjusted/fixed? • EFM must prepare a journal entry adjustment. FM can request. UCLA Department of Medicine Office of Research Administration

• OCGA’s F&A Website • http: //www. research. ucla. edu/ocga/sr 2/idc info. htm • Base & Object Code Table for F&A Links from Today’s Class • https: //www. finance. ucla. edu/corporateaccounting/base-object-code-table-forindirect-costs • FPM/QDB F&A Reconciliation Tool • http: //www. deans. medsch. ucla. edu/overh ead 2015/ • Past F&A Rates • http: //ora. med. ucla. edu/workfiles/referenc ematerials/F-and-A-Cost-Rates. pdf UCLA Department of Medicine Office of Research Administration

Understanding F&A, Revenue Accounts Receivable, Fiscal Closing UCLA Department of Medicine Office of Research Administration

Revenue Account & Accounts Receivable • It is the Fund Manager’s responsibility to ensure that the total amount of the actual payments received from the sponsor match the amount budgeted and appropriated into the expense account. UCLA Department of Medicine Office of Research Administration

Payment Methods UCLA Department of Medicine Office of Research Administration

Understanding the Sponsor’s Payment Methods • A few of the payment methods (check the Award Snapshot): • Milestone Payment: Sponsor provides payments to UCLA based upon the completion of a milestone, as defined in the award document. • Letter of Credit: Sponsor provides payment electronically via the US Treasury (e. g. NIH). UCLA is required to submit drawdown requests and periodic financial reports. • UCLA Standard Invoice: Sponsor provides payments to UCLA based upon expenditures incurred. Sponsor does not have an invoice format specified, therefore, EFM uses the standard UCLA invoice template. • Sponsor Invoice Template: Sponsor provides payment based upon a request from UCLA on the Sponsor’s form. Formats vary and are identified in the Sponsor’s terms. UCLA Department of Medicine Office of Research Administration

Clinical Trial Funds UCLA Department of Medicine Office of Research Administration

Clinical Trial Funds • It is the Fund Manager’s responsibility to: • Ensure that checks from the sponsor have been received an processed for all outstanding invoices/income earned. • Ensure all revenue received has been transferred to the PI’s linked expense account (e. g. 4413 xx). • Ensure the income received actually belongs to the clinical trial in question. • Clinical Trial CRU FAU: 116019 -59995 UCLA Department of Medicine Office of Research Administration

Tracking Payments UCLA Department of Medicine Office of Research Administration

Tracking Payments • Review the CRU funds • Private: 116019 -59995 • State/Local Gov’t: 116026 -20998 • Federal: 116013 -28997 • If you still cannot find the check, do the following: • Request a copy of the front & back of the paid check from the Sponsor. • If paid electronically, request a screen shot from the sponsor’s payment system. • Contact Robert Scott at EFM, and provide him with the following information: • Sponsor name • Check amount • Check date UCLA Department of Medicine Office of Research Administration

Live Demonstration – Revenue Account & Accounts Receivable • Revenue Account & Accounts Receivable Chapter • Revenue Account List • Payment Methods UCLA Department of Medicine Office of Research Administration

Payments in Foreign Funds • Discussion Points: • Contract language • Conversion rates • Payment scheduling UCLA Department of Medicine Office of Research Administration

Understanding F&A, Revenue Accounts Receivable, Fiscal Closing UCLA Department of Medicine Office of Research Administration

Fiscal Closing • Ledger clean-up • Zero out subs to close funds • Submit Closing Packets to EFM to close contract & grants • NPEARs • Unrestricted funds are limited to current fiscal year • Contract & Grant funds are limited to current fiscal year, plus 2 previous fiscal years • State funds • Carryforward requests • Indirect Cost Return balance should be < 10% of annual appropriation UCLA Department of Medicine Office of Research Administration

• Fiscal Close • https: //www. finance. ucla. edu/corporateaccounting/closing-schedule-for-fiscalyear-end Links from Today’s Class UCLA Department of Medicine Office of Research Administration

Survey Link http: //goo. gl/forms/C 3 gdjs. L 5 y 1 We appreciate if you would take a few moments to complete a short 5 question survey to help us improve your training experience. Thank you! UCLA Department of Medicine Office of Research Administration
- Slides: 32