Understanding Drug Labels Chapter 8 MAT 119 Drug

Understanding Drug Labels Chapter 8 MAT 119

Drug Names • Brand, trade, proprietary – Manufacturer’s name for a drug – Brand name is usually most prominent –® - means registered –™ - means Trade Mark • Generic – Established, nonproprietary name – Generic name must be on all drug labels 2

Brand Generic Names 3
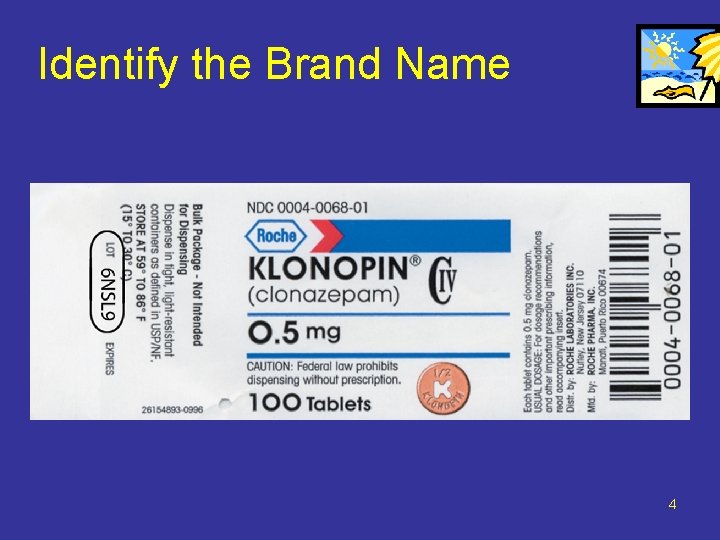
Identify the Brand Name 4

Identify the Generic Name 5

Identify Drug Name: Generic or Brand? Note: Generic Name only 6

Dosage Strength • Dosage weight or amount of drug provided in a specific unit of measurement 7

Form (more in Chapter 10) • Structure and composition of drug – Solid forms for oral use – Injectable – Suppositories – Creams – Patches 8

Identify Form Note apothecary & is incorrect Should be gr 1/150 9
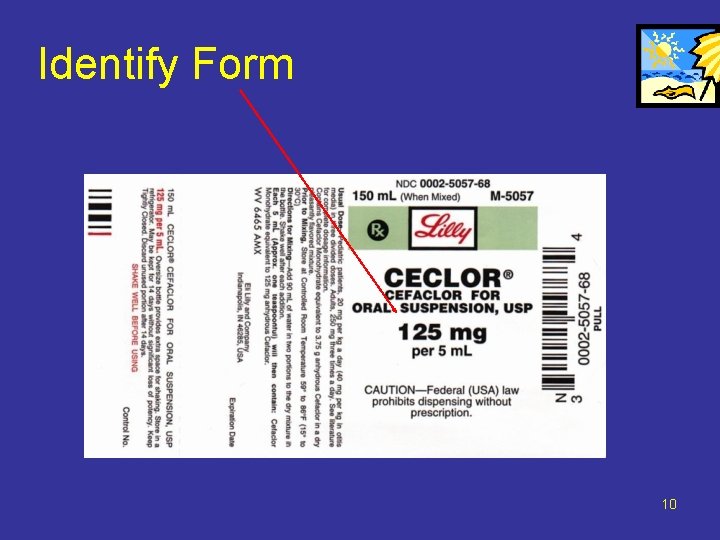
Identify Form 10

Supply Dosage • Both dosage and form – Dosage per tablet – Dosage per milliliter 11

Total Volume • Full quantity contained in bottle or vial – Total number of tablets or other solids – Total fluid volume for liquids 12

Administration Route • Site of body or method of drug delivery – Oral • Tablets, capsusles, caplets – Enteral (into the GI tract via tube) – Sublingual – Injection (IV, IM, subcut) – Topical 13

Identify the Administration Route 14

Directions for Reconstituting Some drugs are dispensed in powder form and must be reconstituted for use (more in chapter 12) 15

Continued Directions for reconstitution 16

Continued 17

Continued 18

Label Alerts • Warnings or special alerts – Examples • Refrigerate at all times • Keep in a dry place Alert 19

Name of Manufacturer 20

Expiration Date • The medication should be used, discarded or returned to the pharmacy by the expiration date. Exp in powder form Exp when reconstituted 21

Lot or Control Numbers • Important if drug is recalled – required by Federal Law • Quickly identifies a particular group of medication packages Control No. 22

National Drug Code (NDC) • Every drugs has an NDC – similar to SS# 23

Controlled Substance Schedule • Classifies drugs according to potential for use and abuse – Schedule I – highest potential for abuse – Schedule V – lowest potential for abuse Has the letter C with Roman Numerals 24

Bar Code Symbols • Used in retail sales; document drug dosing, even at the patient’s bedside. Bar Code 25

United States Pharmacopeia (USP) National Formulary (NF) • The USP and NF are the two official national lists of approved drugs. • These initials are placed after the generic drug name – be careful not to confuse a drug form like SR – sustained release 26

Unit or Single-Dose Labels • Most oral and some IV medications given in the hospital setting are available in single (unit) doses. • The total volume is usually omitted on these containers because they only hold a single dose. 27
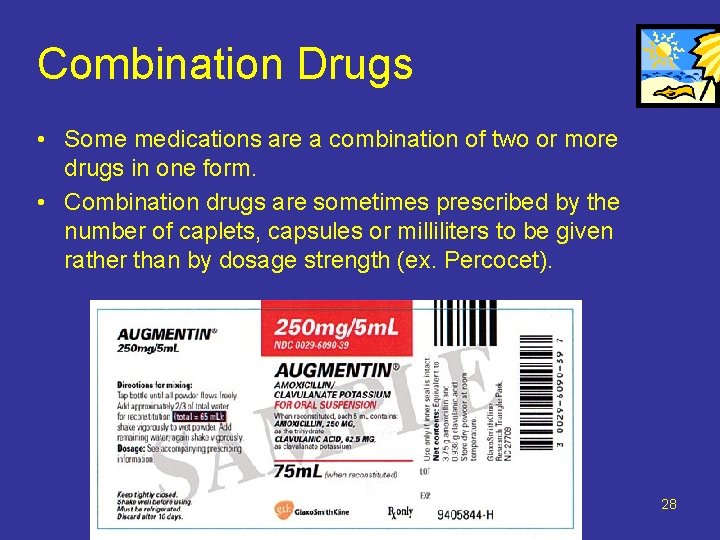
Combination Drugs • Some medications are a combination of two or more drugs in one form. • Combination drugs are sometimes prescribed by the number of caplets, capsules or milliliters to be given rather than by dosage strength (ex. Percocet). 28

Supply Dosage expressed as a Percent • Percentage (%) solutions express the number of grams of the drug per 100 milliliters of solution. • Betoptic Opthalmic Solution 0. 5% – contains 0. 5 g of drug per 100 m. L 0. 5 g : 100 m. L = 500 mg : 100 m. L = 5 mg/m. L 29

Supply Dosage expressed as a Ratio • Ratio solutions express the number of grams of the drug per total milliliters of solution. • Epinephrine 1: 10, 000 – contains 1 g of the drug per 10, 000 m. L solution 1 g: 10, 000 m. L = 1, 000 mg: 10, 000 m. L = 1 mg/10 m. L 30

Check Labels Before administering a medication to a patient, check the drug label three times: 1. On first contact - against the medication order or MAR 2. When preparing the medication 3. Before administering it. 31

Six Rights of Medication Administration – Right patient – Right drug – Right amount – Right route – Right time – Right documentation • The right patient must receive the right drug in the right amount by the right route at the right time, followed by the right documentation. 32

Identify parts of the drug label üBrand name? üRoute? üLabel alerts? üManufacturer? 33

Identify parts of the drug label • NDC #? • Generic name? • Route? 34

Identify parts of the drug label o. Supply dosage? o. Volume? o. Brand name? 35
- Slides: 35