Understanding Bleach Degradation Bernard Bubnis Nova Cem Laboratories

Understanding Bleach Degradation Bernard Bubnis Nova. Cem Laboratories, Inc. Oxford, OH USA www. novachemlabs. com Sponsored by: Powell Fabrication

Nova. Chem Laboratories, Inc. Testing bleach since 1989 1990’s - AWWA Bleach Decomposition Study Collaboration with Powell Fabrication & Manufacturing Decomposition Model Studies Chemistry Validation Measurement Protocols for ◦ Inorganic disinfection by-products ◦ Transition metal ions

Bleach Degradation Parameters That Influence ◦ Concentration ◦ Temperature ◦ Ionic Strength ◦ Transition Metal Ions Decomposition

What is the chemistry that controls decomposition and the formation of disinfection by-products ?
![Bleach Decomposition 2 nd Order Rate = k 2 [OCl-]2 Primary Pathway OCl- + Bleach Decomposition 2 nd Order Rate = k 2 [OCl-]2 Primary Pathway OCl- +](http://slidetodoc.com/presentation_image/52fa3ea011cccbec43346e345ef79800/image-5.jpg)
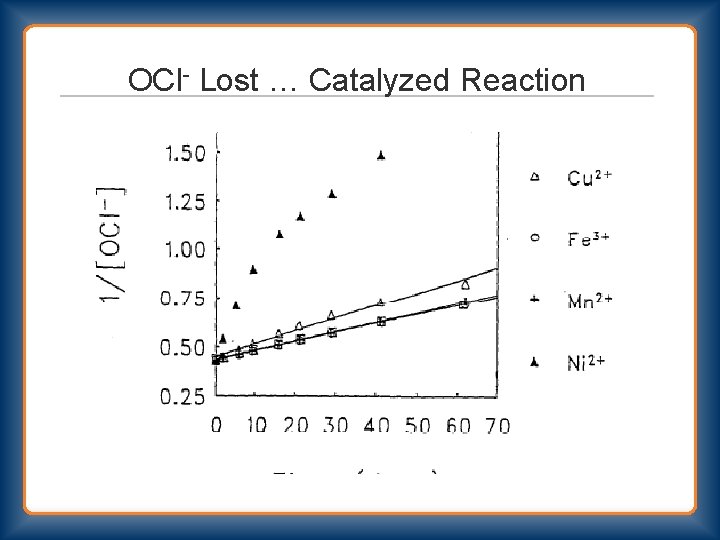
Bleach Decomposition 2 nd Order Rate = k 2 [OCl-]2 Primary Pathway OCl- + OCl- → Cl. O 2 - + Cl. OCl- + Cl. O 2 - → Cl. O 3 - + Cl- Stoichiometry 3 OCl- → Cl. O 3 - + 2 Cl- Secondary Pathway OCl- + OCl- → O 2 + 2 Cl“uncatalyzed” and “catalyzed”
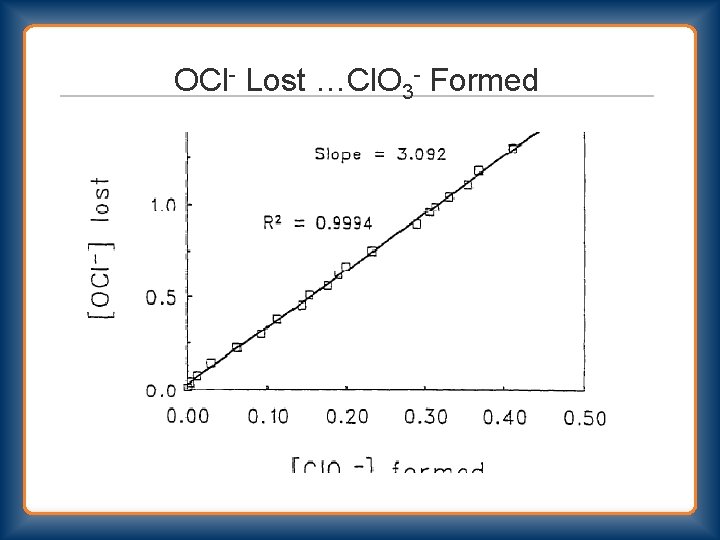
OCl- Lost …Cl. O 3 - Formed
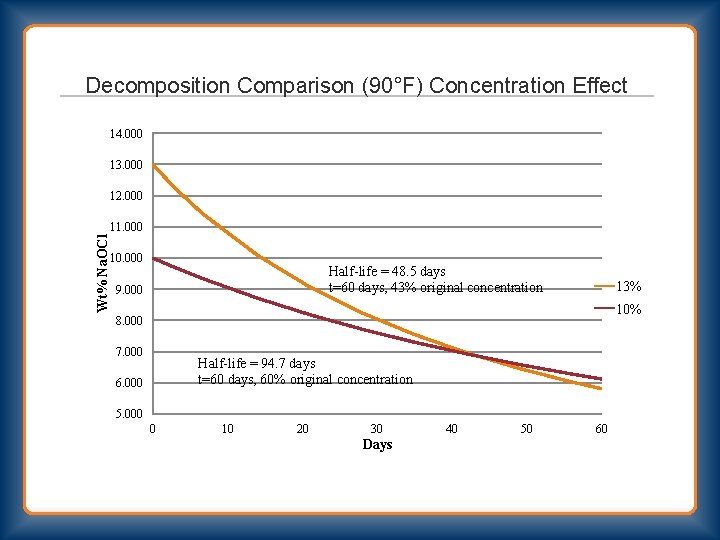
Decomposition Comparison (90°F) Concentration Effect 14. 000 13. 000 12. 000 Wt% Na. OCl 11. 000 10. 000 Half-life = 48. 5 days t=60 days, 43% original concentration 9. 000 13% 10% 8. 000 7. 000 Half-life = 94. 7 days t=60 days, 60% original concentration 6. 000 5. 000 0 10 20 30 Days 40 50 60
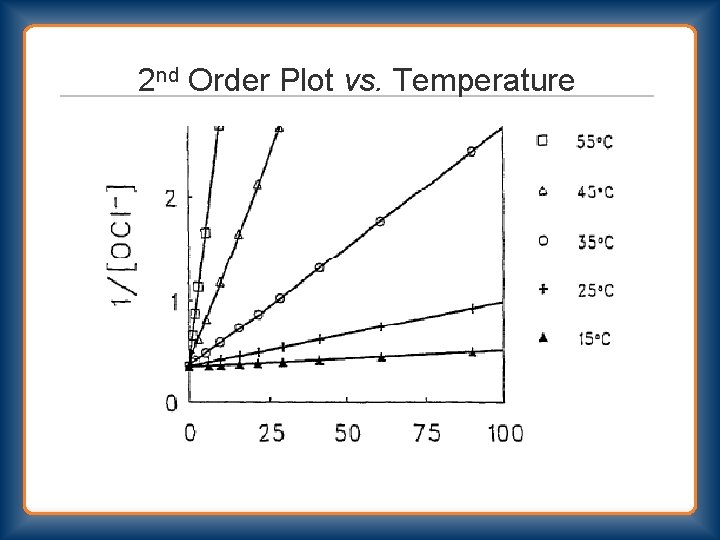
2 nd Order Plot vs. Temperature
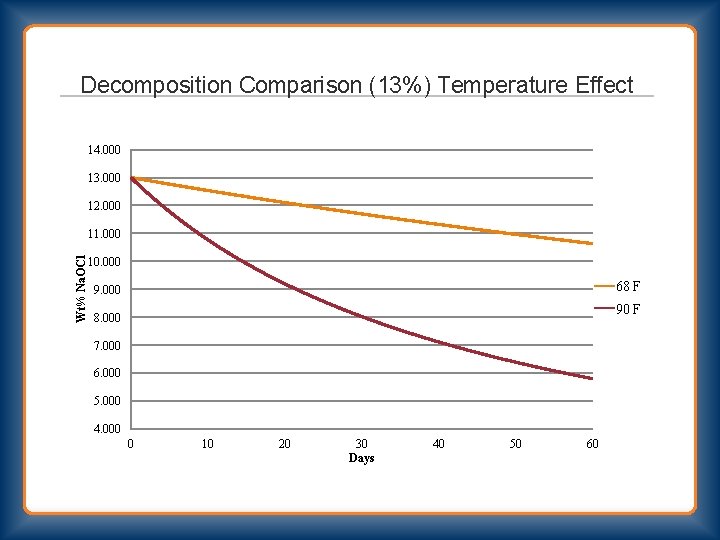
Decomposition Comparison (13%) Temperature Effect 14. 000 13. 000 12. 000 Wt% Na. OCl 11. 000 10. 000 68 F 9. 000 90 F 8. 000 7. 000 6. 000 5. 000 4. 000 0 10 20 30 Days 40 50 60
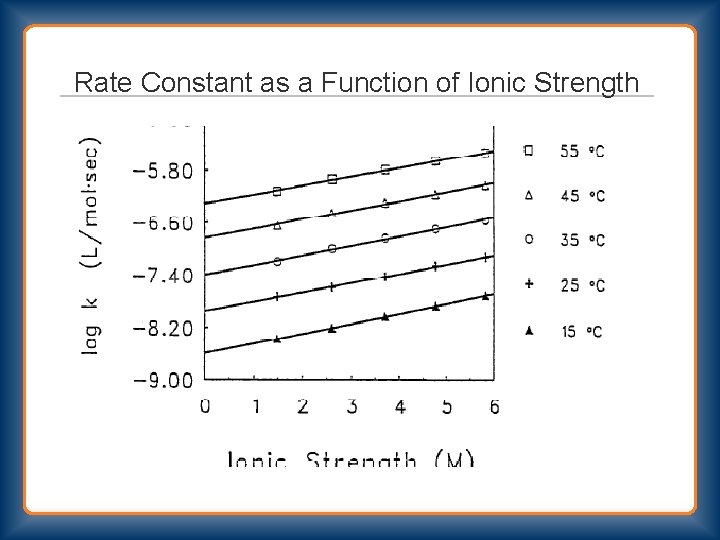
Rate Constant as a Function of Ionic Strength
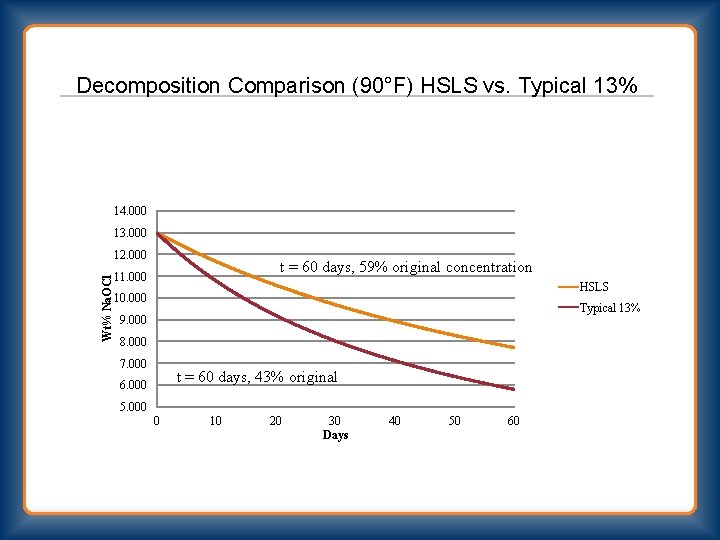
Decomposition Comparison (90°F) HSLS vs. Typical 13% 14. 000 13. 000 Wt% Na. OCl 12. 000 t = 60 days, 59% original concentration 11. 000 HSLS 10. 000 Typical 13% 9. 000 8. 000 7. 000 t = 60 days, 43% original 6. 000 5. 000 0 10 20 30 Days 40 50 60

HSLS Bleach “High Strength Low Salt” 30 Wt% Na. OCl ◦ 385 GPL Available Chlorine ◦ ≈1 Wt% Na. OH ◦ 8. 5 Wt% Na. Cl 13 Wt% Na. OCl ◦ 10. 4 Wt% Na. Cl (traditional)

HSLS Bleach “High Strength Low Salt” 13% Wt Na. OCl ◦ 10. 4% Wt Na. Cl 13% (traditional) Wt HSLS - 3. 7% Wt Na. Cl 6% Wt HSLS - 1. 7% Wt Na. Cl
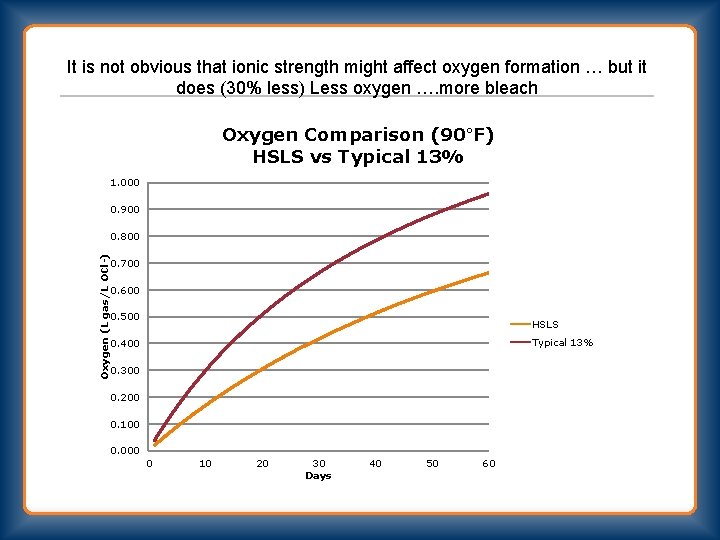
It is not obvious that ionic strength might affect oxygen formation … but it does (30% less) Less oxygen …. more bleach Oxygen Comparison (90°F) HSLS vs Typical 13% 1. 000 0. 900 Oxygen (L gas/L OCl-) 0. 800 0. 700 0. 600 0. 500 HSLS Typical 13% 0. 400 0. 300 0. 200 0. 100 0. 000 0 10 20 30 Days 40 50 60
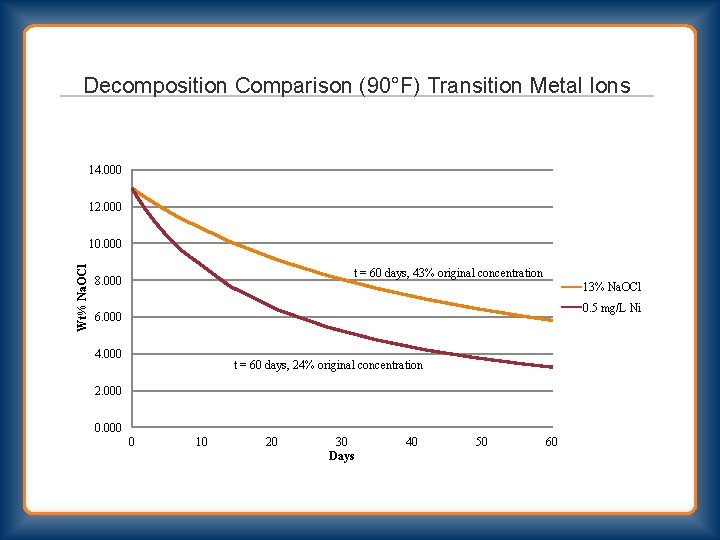
Decomposition Comparison (90°F) Transition Metal Ions 14. 000 12. 000 Wt% Na. OCl 10. 000 t = 60 days, 43% original concentration 8. 000 13% Na. OCl 0. 5 mg/L Ni 6. 000 4. 000 t = 60 days, 24% original concentration 2. 000 0 10 20 30 Days 40 50 60
OCl- Lost … Catalyzed Reaction
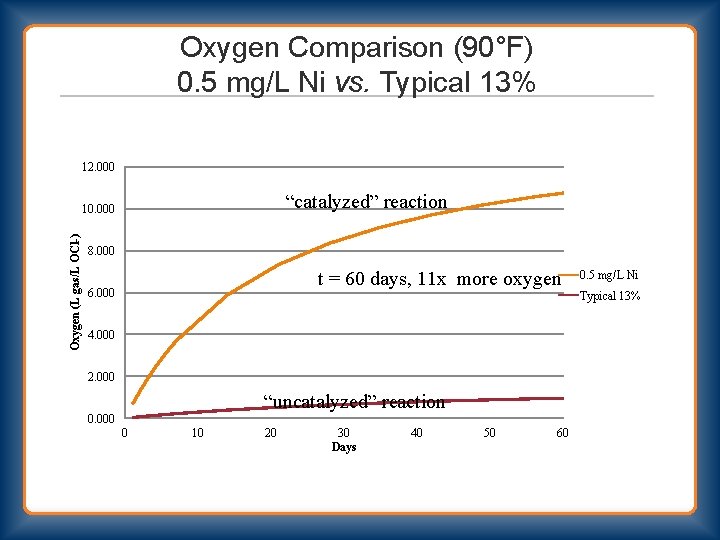
Oxygen Comparison (90°F) 0. 5 mg/L Ni vs. Typical 13% 12. 000 “catalyzed” reaction Oxygen (L gas/L OCl-) 10. 000 8. 000 t = 60 days, 11 x more oxygen 6. 000 0. 5 mg/L Ni Typical 13% 4. 000 2. 000 “uncatalyzed” reaction 0. 000 0 10 20 30 Days 40 50 60
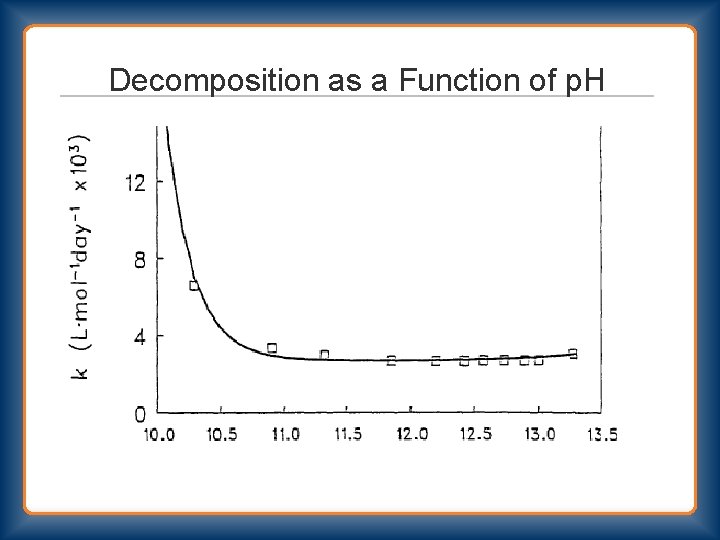
Decomposition as a Function of p. H
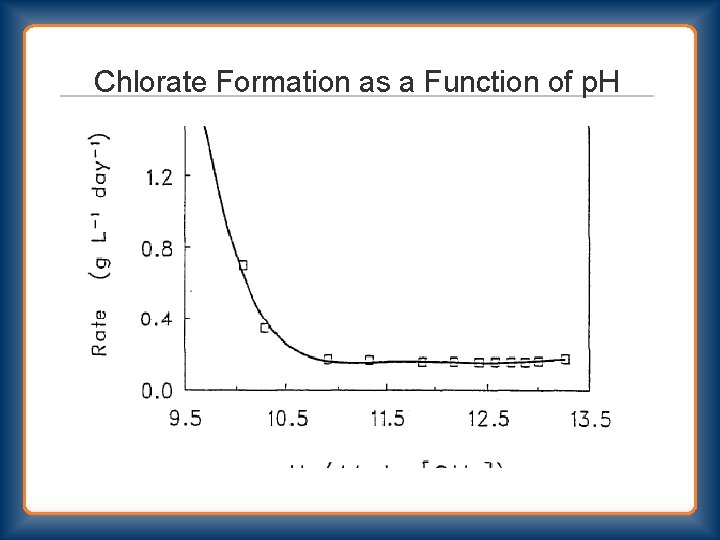
Chlorate Formation as a Function of p. H
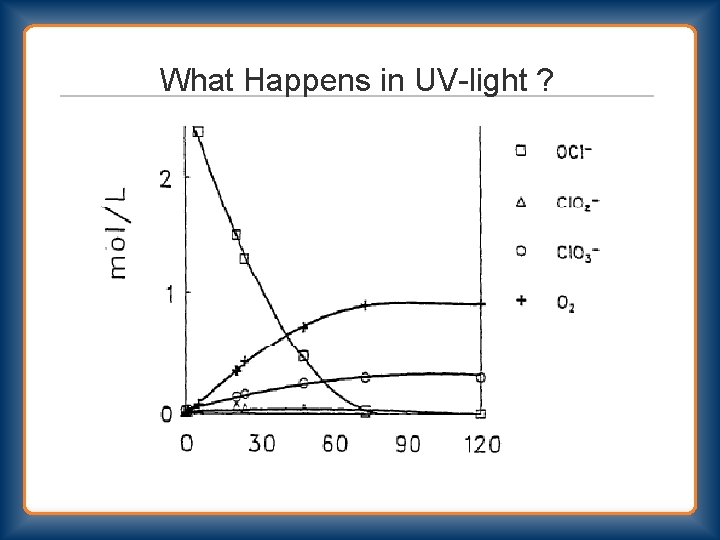
What Happens in UV-light ?

Unwanted By-Products What are they ? ◦ Bromate Br. O 3◦ Chlorate Cl. O 3◦ Perchlorate Cl. O 4 How do they get into the bleach? Raw materials and decomposition

Bromate Ion, Br. O 3 Bromide ion in salt used to make ◦ Forms Br 2 ◦ Reacts with caustic to form Br. O 3 Impurity in caustic Cl 2

Perchlorate Ion, Cl. O 4 Source: Water Research Foundation, 2009, An Assessment of the Factors that Influence the Formation of Perchlorate and Other Contaminants Reaction OCl- + Cl. O 3 - → Cl. O 4 - + Cl- Strongly dependent on Temperature and Ionic Strength Rate Law d[Cl. O 4 -]/dt = k. Cl. O 4 - [OCl-] [Cl. O 3 -] Rate Constant log (k. Cl. O 4 - ) = 0. 0788(I) + log (2. 084 x 1016 x T x e -1. 01 x 10^5/RT x e -10^6/R)
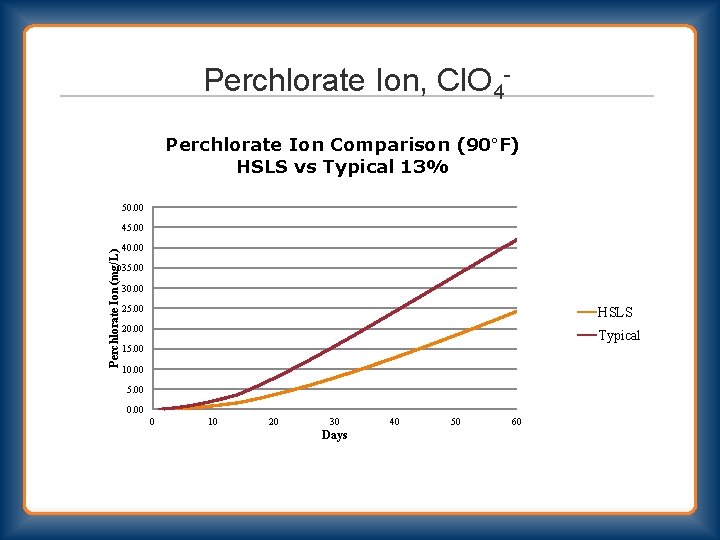
Perchlorate Ion, Cl. O 4 Perchlorate Ion Comparison (90°F) HSLS vs Typical 13% 50. 00 Perchlorate Ion (mg/L) 45. 00 40. 00 35. 00 30. 00 25. 00 HSLS 20. 00 Typical 15. 00 10. 00 5. 00 0 10 20 30 Days 40 50 60

Basis of a Chemical Model Chlorate and Oxygen Formation (3 k. Cl + 2 kox)t = 1/[OCl-]t – 1/[OCl-]0 Perchlorate Ion Formation d[Cl. O 4 -]/dt = k. Cl. O 4 - [OCl-] [Cl. O 3 -] log (k. Cl. O 4 - ) = 0. 0788(I) + log (2. 084 x 1016 x T x e -1. 01 x 10^5/RT x e -10^6/R)

Decomposition Software Modeling What can you model ? ◦ Bleach Loss ◦ Chlorate Ion Formation ◦ Oxygen Formation ◦ Perchlorate Ion Formation What factors can you manipulate? ◦ Concentration ◦ Temperature ◦ Caustic strength ◦ Ionic strength ◦ Metal ion concentration ◦ Time parameters
Powell Software
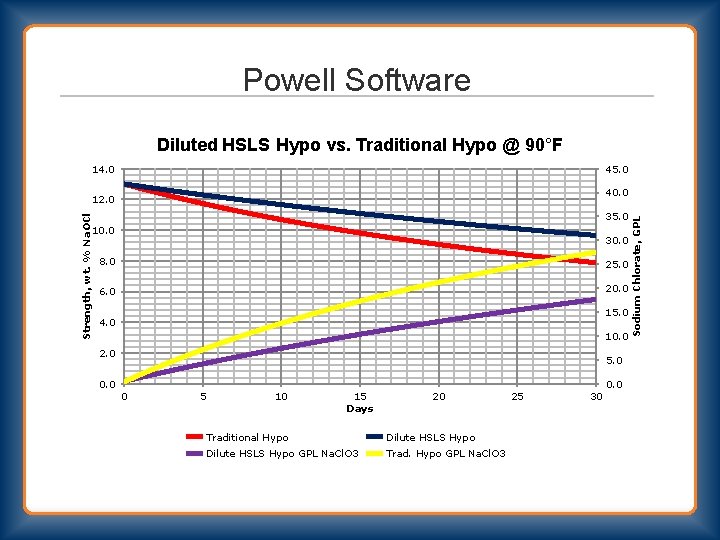
Powell Software Diluted HSLS Hypo vs. Traditional Hypo @ 90°F 14. 0 45. 0 Strength, wt. % Na. OCl 35. 0 10. 0 30. 0 8. 0 25. 0 6. 0 20. 0 15. 0 4. 0 10. 0 2. 0 5. 0 0 5 10 15 Days 20 Traditional Hypo Dilute HSLS Hypo GPL Na. Cl. O 3 Trad. Hypo GPL Na. Cl. O 3 25 30 Sodium Chlorate, GPL 40. 0 12. 0
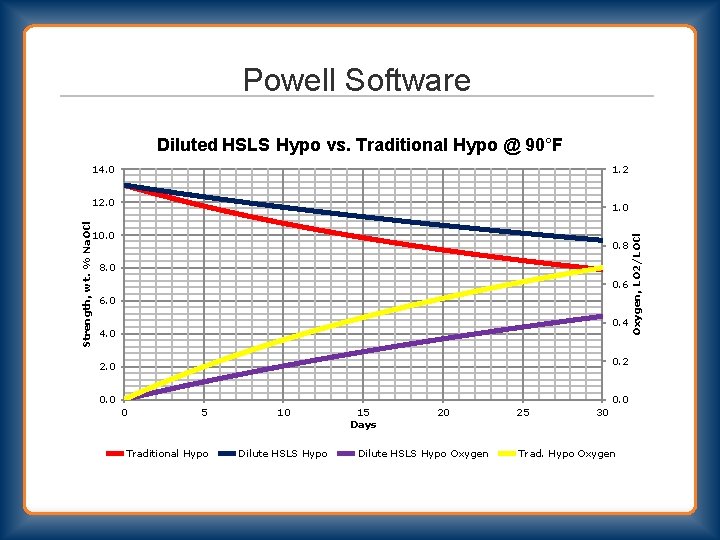
Powell Software Diluted HSLS Hypo vs. Traditional Hypo @ 90°F 14. 0 1. 2 1. 0 10. 0 0. 8 8. 0 0. 6 6. 0 0. 4 4. 0 0. 2 2. 0 0 5 Traditional Hypo 10 Dilute HSLS Hypo 15 Days 20 Dilute HSLS Hypo Oxygen 25 30 Trad. Hypo Oxygen, LO 2/LOCl Strength, wt. % Na. OCl 12. 0
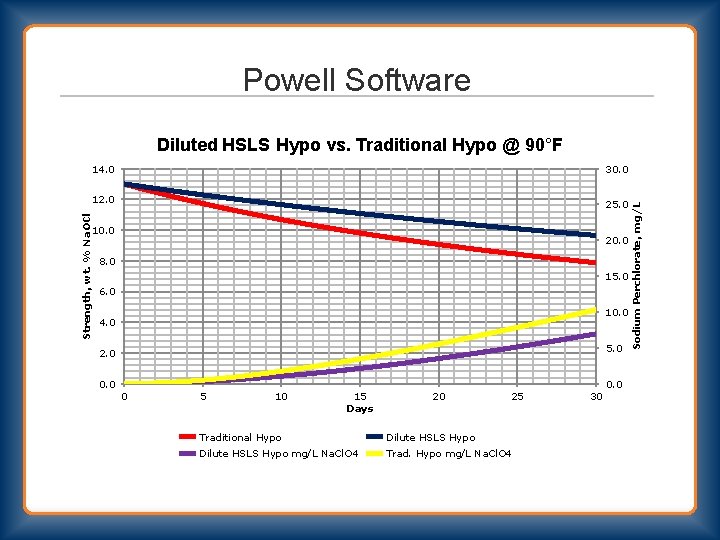
Powell Software Diluted HSLS Hypo vs. Traditional Hypo @ 90°F 30. 0 Strength, wt. % Na. OCl 12. 0 25. 0 10. 0 20. 0 8. 0 15. 0 6. 0 10. 0 4. 0 5. 0 2. 0 0 5 10 15 Days 20 25 Traditional Hypo Dilute HSLS Hypo mg/L Na. Cl. O 4 Trad. Hypo mg/L Na. Cl. O 4 30 Sodium Perchlorate, mg/L 14. 0

Bleach Testing Essential Wt% Na. OCl Wt% Na. OH Optional mg/L bromate Optional Filter test Analyses: (Tier 1) g/L Available Cl 2 Wt% Na 2 CO 3 Specific gravity mg/L chlorate Analyses I: (Tier 2) mg/L perchlorate mg/L Fe, Cu, Ni Analysis II: (Tier 3) Dissolved Solids Suspended Solids
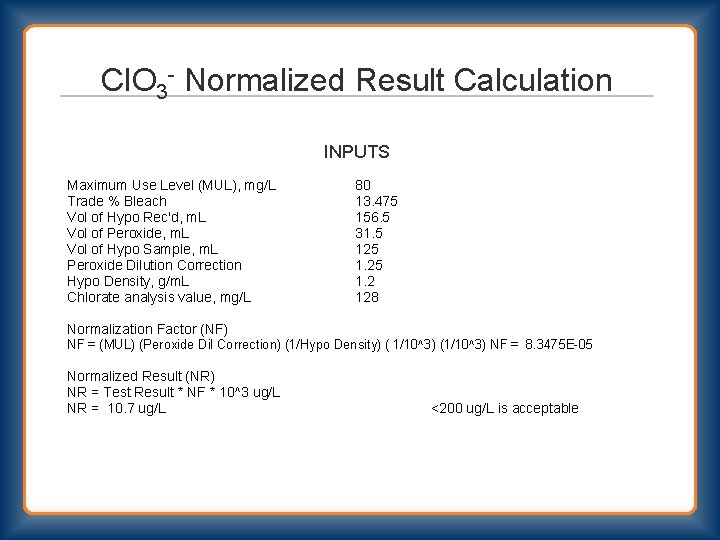
Cl. O 3 - Normalized Result Calculation INPUTS Maximum Use Level (MUL), mg/L Trade % Bleach Vol of Hypo Rec'd, m. L Vol of Peroxide, m. L Vol of Hypo Sample, m. L Peroxide Dilution Correction Hypo Density, g/m. L Chlorate analysis value, mg/L 80 13. 475 156. 5 31. 5 125 1. 2 128 Normalization Factor (NF) NF = (MUL) (Peroxide Dil Correction) (1/Hypo Density) ( 1/10^3) (1/10^3) NF = 8. 3475 E-05 Normalized Result (NR) NR = Test Result * NF * 10^3 ug/L NR = 10. 7 ug/L <200 ug/L is acceptable

Filter Test Operations Tool Particulate matter causes problems In-field testing determined the parameters At 20 inches of Hg, 1 liter of bleach should filter through 0. 8 micron filter paper in < 3 minutes

Suggested Bleach QA Manage the Decomposition ◦ Concentration Temperature Transition Metal ◦ Ionic Strength Manage Ions the Storage and Delivery Model the Bleach and Verify Make Measurements Tier I analysis (minimum)
- Slides: 34