Umbilical Cord Blood Transplantation in Pediatric Patients with

Umbilical Cord Blood Transplantation in Pediatric Patients with Malignant Diseases Results of a Prospective, Multi-Institutional NHLBI Sponsored Trial J Kurtzberg, SL Carter, JE Wagner, LA Baxter-Lowe, D Wall, N Kapoor, E Guinan, S Feig, E Wagner, NA Kernan on behalf of the COBLT Steering Committee

Overview of the COBLT Study • Prospective, multi-institutional study sponsored by the NHLBI. – 3 banks: • Duke • UCLA • CHOC/GT J Kurtzberg J Fraser M Cairo • Created inventory of approx 8, 000 units • 71 % of transplants used COBLT units • Transplantation study: Strata for adults, peds ID, metabolic ds & peds malignancies.

The Transplantation Study • Common protocol for preparative regimen, Gv. HD prophylaxis and supportive care. • Common criteria for evaluation of risk status at study entry, donor selection, engraftment, Gv. HD, infection and causes of death.

Study End Points Primary Endpoint • 180 day survival Secondary Endpoints • Disease-free survival • Incidence of neutrophil engraftment • Incidence of platelet engraftment • Incidence of acute and chronic GVHD • Incidence of relapse Analysis of high resolution HLA typing and effect on outcomes.

Definitions • Original HLA Matching – Low resolution DNA based typing for Class I, A and B, High resolution DNA typing for Class II, DRB 1 • Retrospective (final) HLA Matching – High resolution DNA typing for A, B, DRB 1 • Engraftment – ANC 500/u. L with >90% donor chimerism by day 42

Patient Eligibility • Pediatric Patients between 1 -18 years • Hematological malignancies – Exclude CML blast crisis – Exclude ALL relapse • No related donor • UCB donor delivering >10 e 7 cells/kg, matching at 4/6 HLA loci by low resolution typing at Class I, or 3/6 with high resolution matching, 1 allele at A, B, DRB 1.

Treatment • Preparative Regimen – TBI 1350 – Cyclophosphamide 120 – Horse ATG 90 • Gv. HD Prophylaxis – Cyclosporine – Methyprednisolone • Supportive Care – – Anti viral, fungal and PCP prophylaxis Antigen/DNA monitoring for CMV G-CSF from day 0 IVIG prophylaxis
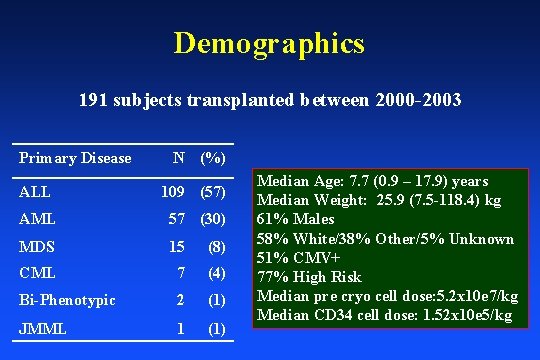
Demographics 191 subjects transplanted between 2000 -2003 Primary Disease N (%) ALL 109 (57) AML 57 (30) MDS 15 (8) CML 7 (4) Bi-Phenotypic 2 (1) JMML 1 (1) Median Age: 7. 7 (0. 9 – 17. 9) years Median Weight: 25. 9 (7. 5 -118. 4) kg 61% Males 58% White/38% Other/5% Unknown 51% CMV+ 77% High Risk Median pre cryo cell dose: 5. 2 x 10 e 7/kg Median CD 34 cell dose: 1. 52 x 10 e 5/kg
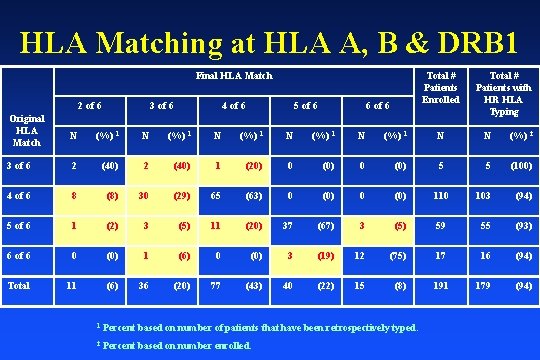
HLA Matching at HLA A, B & DRB 1 Final HLA Match 2 of 6 3 of 6 4 of 6 5 of 6 Total # Patients Enrolled 6 of 6 Total # Patients with HR HLA Typing Original HLA Match N (%) 1 N (%) 1 N N (%) 2 3 of 6 2 (40) 1 (20) 0 (0) 5 5 (100) 4 of 6 8 (8) 30 (29) 65 (63) 0 (0) 110 103 (94) 5 of 6 1 (2) 3 (5) 11 (20) 37 (67) 3 (5) 59 55 (93) 6 of 6 0 (0) 1 (6) 0 (0) 3 (19) 12 (75) 17 16 (94) Total 11 (6) 36 (20) 77 (43) 40 (22) 15 (8) 191 179 (94) 1 Percent based on number of patients that have been retrospectively typed. 2 Percent based on number enrolled.
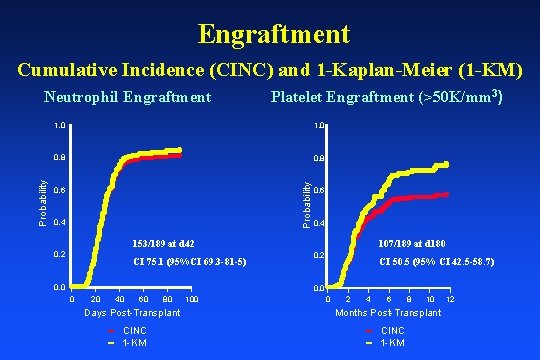
Engraftment Cumulative Incidence (CINC) and 1 -Kaplan-Meier (1 -KM) Platelet Engraftment (>50 K/mm 3) 1. 0 0. 8 Probability Neutrophil Engraftment 0. 6 0. 4 153/189 at d 42 0. 2 CI 75. 1 (95%CI 69. 3 -81 -5) 0. 0 107/189 at d 180 0. 2 CI 50. 5 (95% CI 42. 5 -58. 7) 0. 0 0 20 40 60 80 Days Post-Transplant CINC 1 -KM 100 0 2 4 6 8 10 Months Post-Transplant CINC 1 -KM 12
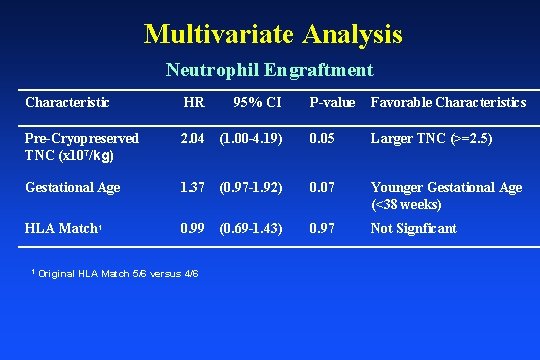
Multivariate Analysis Neutrophil Engraftment Characteristic HR 95% CI Pre-Cryopreserved TNC (x 107/kg) 2. 04 Gestational Age HLA Match 1 1 P-value Favorable Characteristics (1. 00 -4. 19) 0. 05 Larger TNC (>=2. 5) 1. 37 (0. 97 -1. 92) 0. 07 Younger Gestational Age (<38 weeks) 0. 99 (0. 69 -1. 43) 0. 97 Not Signficant Original HLA Match 5/6 versus 4/6
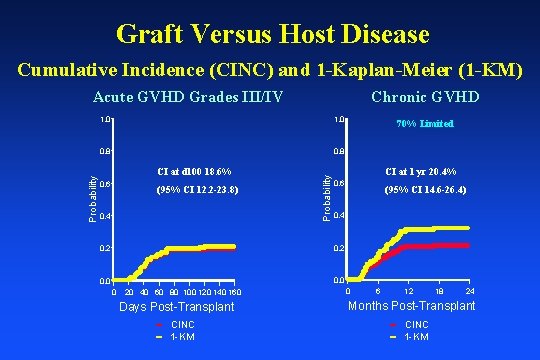
Graft Versus Host Disease Cumulative Incidence (CINC) and 1 -Kaplan-Meier (1 -KM) Chronic GVHD 1. 0 0. 8 CI at d 100 18. 6% 0. 6 (95% CI 12. 2 -23. 8) 0. 4 Probability Acute GVHD Grades III/IV 70% Limited CI at 1 yr 20. 4% 0. 6 (95% CI 14. 6 -26. 4) 0. 4 0. 2 0. 0 0 20 40 60 80 100 120 140 160 Days Post-Transplant CINC 1 -KM 0 6 12 18 24 Months Post-Transplant CINC 1 -KM

Multivariate Analysis Acute GVHD Grades III/IV Characteristic HR 95% CI HLA Match 1 0. 35 (0. 16 -0. 75) 1 Original HLA Match 5/6 or 6/6 versus 3/6 or 4/6 P-value Favorable Characteristics 0. 007 5/6 or 6/6 HLA Match
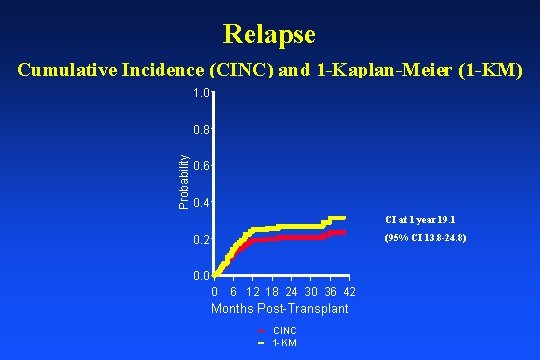
Relapse Cumulative Incidence (CINC) and 1 -Kaplan-Meier (1 -KM) 1. 0 Probability 0. 8 0. 6 0. 4 CI at 1 year 19. 1 (95% CI 13. 8 -24. 8) 0. 2 0. 0 0 6 12 18 24 30 36 42 Months Post-Transplant CINC 1 -KM
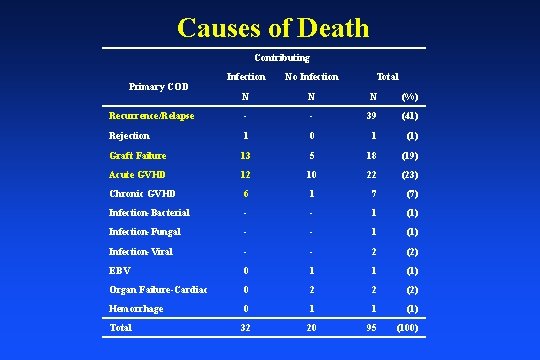
Causes of Death Contributing Infection No Infection N N N (%) Recurrence/Relapse - - 39 (41) Rejection 1 0 1 (1) Graft Failure 13 5 18 (19) Acute GVHD 12 10 22 (23) Chronic GVHD 6 1 7 (7) Infection-Bacterial - - 1 (1) Infection-Fungal - - 1 (1) Infection-Viral - - 2 (2) EBV 0 1 1 (1) Organ Failure-Cardiac 0 2 2 (2) Hemorrhage 0 1 1 (1) Total 32 20 95 (100) Primary COD Total
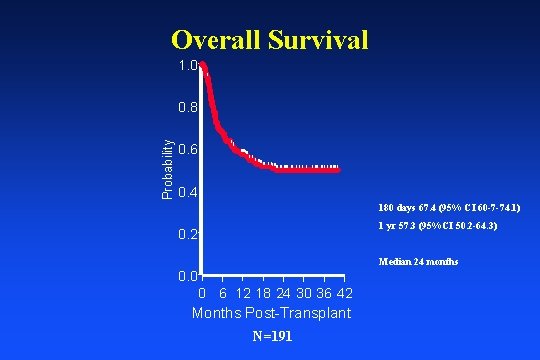
Overall Survival 1. 0 | Probability 0. 8 0. 6 | || | ||| || | | |||| | | ||| | | || | 0. 4 | | | | 180 days 67. 4 (95% CI 60 -7 -74. 1) 1 yr 57. 3 (95%CI 50. 2 -64. 3) 0. 2 Median 24 months 0. 0 0 6 12 18 24 30 36 42 Months Post-Transplant N=191
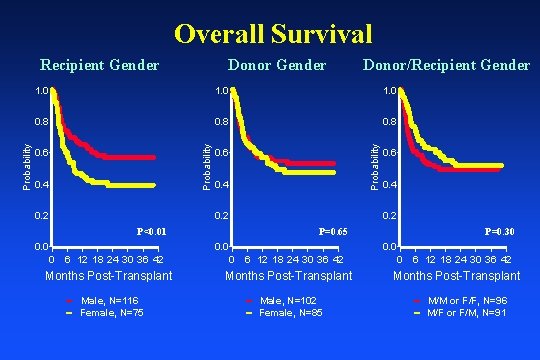
Overall Survival Recipient Gender 1. 0 Donor Gender 1. 0 | | | 0. 8 Donor/Recipient Gender | | | 0. 8 | | | 0. 6 | || || || | | || | || | | 0. 4 || || | | | || || | 0. 2 0. 6 | | ||| | | | | || | || || | | | || | 0. 4 0. 2 0. 0 | | | 0. 6 | | || | || || | | || | | | | | || | | | | 0. 4 0. 2 | P<0. 01 Probability || ||| P=0. 65 0. 0 P=0. 30 0. 0 0 6 12 18 24 30 36 42 Months Post-Transplant Male, N=116 Female, N=75 Male, N=102 Female, N=85 M/M or F/F, N=96 M/F or F/M, N=91
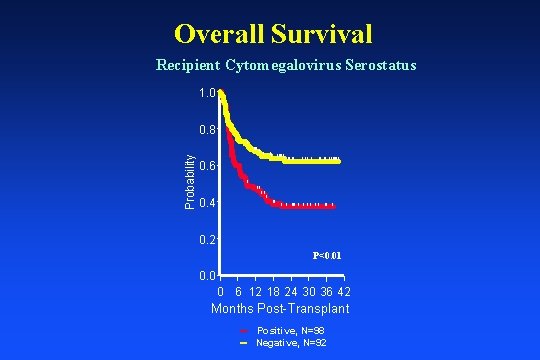
Overall Survival Recipient Cytomegalovirus Serostatus 1. 0 | 0. 8 | Probability ||| | ||| || | 0. 6 || | | ||| | | 0. 4 | | || || | | || | 0. 2 P<0. 01 0. 0 0 6 12 18 24 30 36 42 Months Post-Transplant Positive, N=98 Negative, N=92
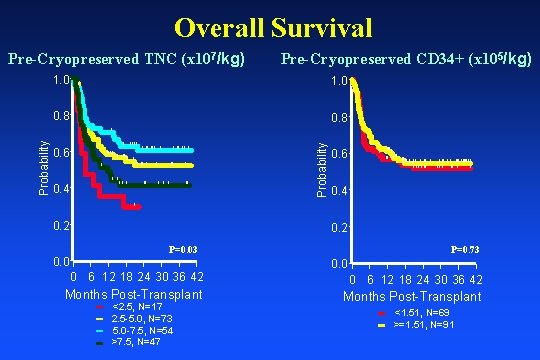
Overall Survival Pre-Cryopreserved TNC (x 107/kg) 1. 0 Pre-Cryopreserved CD 34+ (x 105/kg) 1. 0 | | 0. 8 | | 0. 6 | || | | || || | | | | || | | 0. 4 | | || | || 0. 6 | | || || | | | | | || || | | |||| | | | | 0. 4 | 0. 2 0. 0 Probability | 0. 2 P=0. 03 0 6 12 18 24 30 36 42 Months Post-Transplant <2. 5, N=17 2. 5 -5. 0, N=73 5. 0 -7. 5, N=54 >7. 5, N=47 P=0. 73 0. 0 0 6 12 18 24 30 36 42 Months Post-Transplant <1. 51, N=69 >=1. 51, N=91
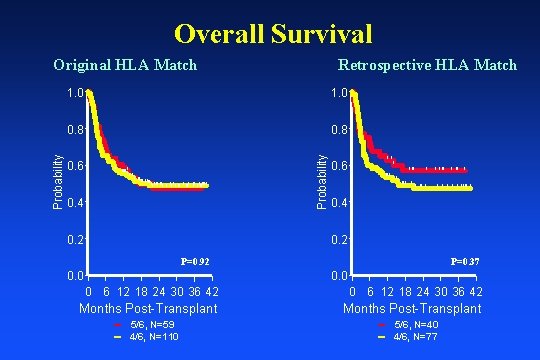
Overall Survival Original HLA Match 1. 0 Retrospective HLA Match 1. 0 | 0. 6 0. 8 | || | | || || | | ||| | | | || | || | || 0. 4 0. 2 Probability 0. 8 | 0. 6 | | ||| | || | || | | | | || | 0. 4 0. 2 P=0. 92 P=0. 37 0. 0 0 6 12 18 24 30 36 42 Months Post-Transplant 5/6, N=59 4/6, N=110 5/6, N=40 4/6, N=77

Multivariate Analysis For Survival Characteristic HR 95% CI ANC Engraftment 1 3. 03 Recipient Cytomegalovirus Serostatus Recipient Gender 1 Time Dependent Covariate P-value Favorable Characteristics (1. 87 -4. 91) <0. 001 Early Engrafter 0. 50 (0. 33 -0. 77) 0. 002 Negative 1. 64 (1. 09 -2. 48) 0. 01 Male

Summary • The COBLT Study represents the first prospective, multi-institutional study of cord blood transplantation – Standard treatment protocol – Standard supportive care – Standard outcome definitions and assessments – Analysis of retrospective high resolution HLA typing on outcomes (with 34% of patients having greater disparity than original typing)
Conclusions • Incidence of engraftment is 75% which correlated with nucleated cell dose and donor gestational age. • Incidence of acute grade III/IV acute GVHD was 19%; chronic GVHD was 20% (70% limited) and GVHD was cause of death in 30% of fatalities. • Incidence of relapse was 25% (77% of pts were high risk).

Conclusions • Overall survival was 55% with early engraftment, recipient CMV seronegative status and male gender being favorable risk factors. • Notably, level of HLA match (original or high resolution) and risk status at diagnoses did not impact survival.

Acknowledgements • Cardinal Glennon Children's Hospital - Dr. Michael Kelly • Hackensack University Medical Center - Drs. Joel Brochstein & Stuart Goldberg • Case Western Reserve University Hospital - Dr. Mary • Indiana University Cancer Center - Dr. Paul Haut Laughlin • North Texas Hospital for Children - Dr. Joel Weinthal • Children's Hospital – Los Angeles - Dr. Neena Kapoor • Roswell Park Cancer Institute - Drs. Philip Mc. Carthy & • Children's Hospital – New Orleans - Dr. Lolie Yu Barbara Bambach • Children's Hospital – Orange County - Dr. Steven Neudorf • Schneider Children's Hospital - Dr. Indira Sahdev • Children's Hospital – Philadelphia - Dr. Nancy Bunin • Texas Transplant Institute - Drs. Carlos Bachier & Donna • Children's Hospital – Pittsburgh - Dr. Rakesh Goyal Wall • Children's Medical Center of Dallas - Drs. Victor Aquino & • University of California, Los Angeles - Dr. Stephen Feig Donna Wall • University of California, San Francisco - Drs. Morton Cowan & Biljana Horn • Children's Mercy Hospital - Dr. Andrew Gilman • • Children's National Medical Center - Dr. Naynesh Kamani University of Florida - Dr. Steven Hunger • University of Minnesota - Dr. John Wagner • City of Hope National Medical Center - Dr. Joseph Rosenthal • University of Rochester - Drs. John Horan & Jane Liesveld • Dana Farber Cancer Institute - Drs. Eva Guinan & Leslie • Vanderbilt University - Dr. Haydar Frangoul Lehmann • The EMMES Corporation – Dr. Shelly Carter & Mr. • De. Vos Children's Hospital - Dr. Daniel Pietryga Adam Mendizabal • Fred Hutchinson Cancer Research Center - Dr. Colleen • National Heart, Lung and Blood Institute – Dr. Luiz Delaney Barbosa, Ms. Liz Wagner & Dr. Lee. Ann Jensen • Dr. Nancy A. Kernan, • Steering Committee Chair
- Slides: 25