UltravioletVisible Spectroscopy Introduction to UVVisible Absorption spectroscopy from

Ultraviolet-Visible Spectroscopy • Introduction to UV-Visible § Absorption spectroscopy from 160 nm to 780 nm § Measurement of transmittance à Conversion to absorbance * A=-log. T=ebc • • Measurement of transmittance and absorbance Beer’s law Noise Instrumentation 8 -1

Measurement • Scattering of light § Refraction at interfaces § Scatter in solution àLarge molecules àAir bubbles • Normalized by comparison to reference cell § Contains only solvent àMeasurement for transmittance is compared to results from reference cell 8 -2

Beer’s Law • Based on absorption of light by a sample § d. Px/Px=d. S/S àd. S/S=ratio of absorbance area to total area * Proportional to number of absorbing particles àd. S=adn * a is a constant, dn is number of particles § n is total number of particles within a sample 8 -3

Beer’s Law • Area S can be described by volume and length § S=V/b (cm 2) § Substitute for S § n/V = concentration § Substitute concentration and collect constant into single term e • Beer’s law can be applied to mixtures § Atot=SAx 8 -4
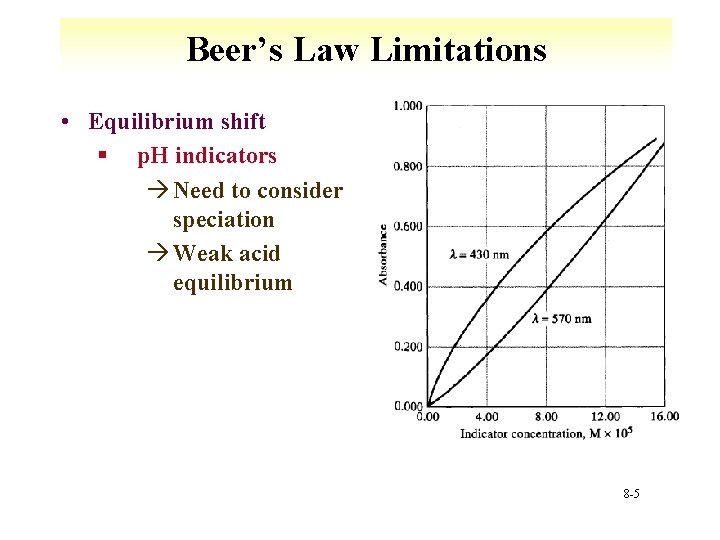
Beer’s Law Limitations • Equilibrium shift § p. H indicators à Need to consider speciation à Weak acid equilibrium 8 -5
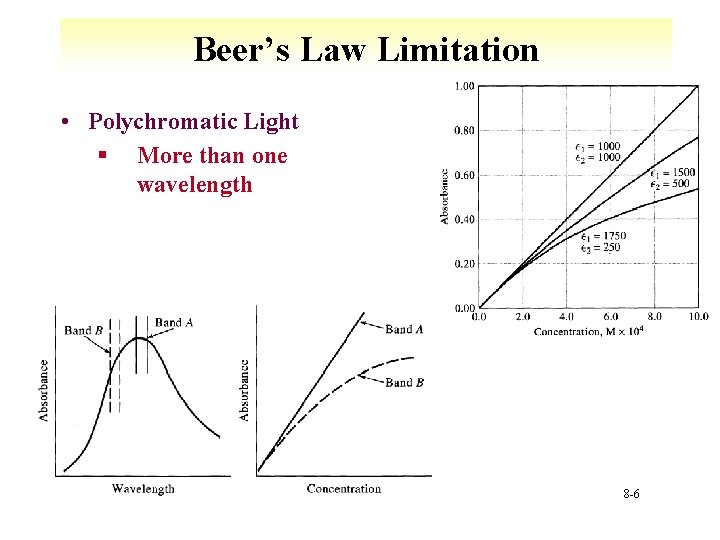
Beer’s Law Limitation • Polychromatic Light § More than one wavelength 8 -6

Noise • • Limited readout resolution Dark current and electronic noise Photon detector shot noise Cell position uncertainty § Changing samples • Flicker 8 -7

Instrumentation • Light source § Deuterium and hydrogen lamps § W filament lamp § Xe arc lamps • Sample containers § Cuvettes à Plastic à Glass à Quartz 8 -8
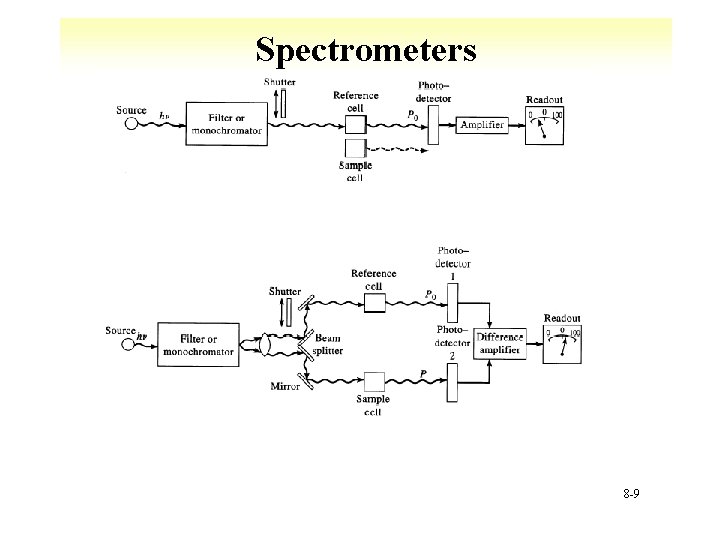
Spectrometers 8 -9
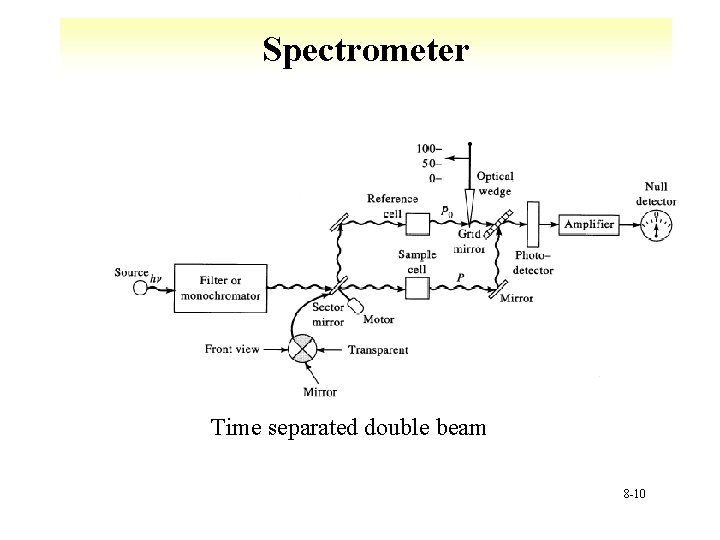
Spectrometer Time separated double beam 8 -10
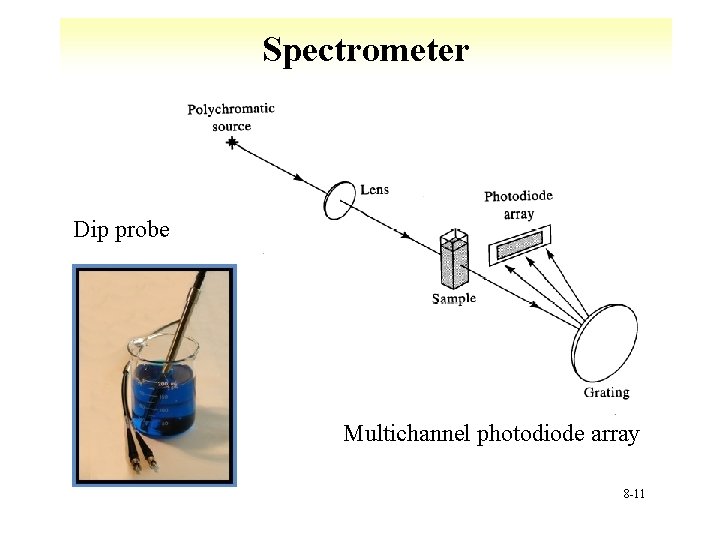
Spectrometer Dip probe Multichannel photodiode array 8 -11

Application of UV-Visible Spectroscopy • Identification of inorganic and organic species • Widely used method • Magnitude of molar absorptivities • Absorbing species • methods 8 -12

Molar Absorptivties • Range from 0 to 1 E 5 § e=8. 7 E 19 PA à P=transition probability à A=target cross section (cm 2) * Allowed transitions 0. 1>P>1 e range 1 E 4 to 1 E 5 * Forbidden transition 0. 01 • Absorbing species § M+g->M* à M* has a short lifetime (nanoseconds) à Relaxation processes * Heat * Photo emission Fluorescence or phosphorescence 8 -13
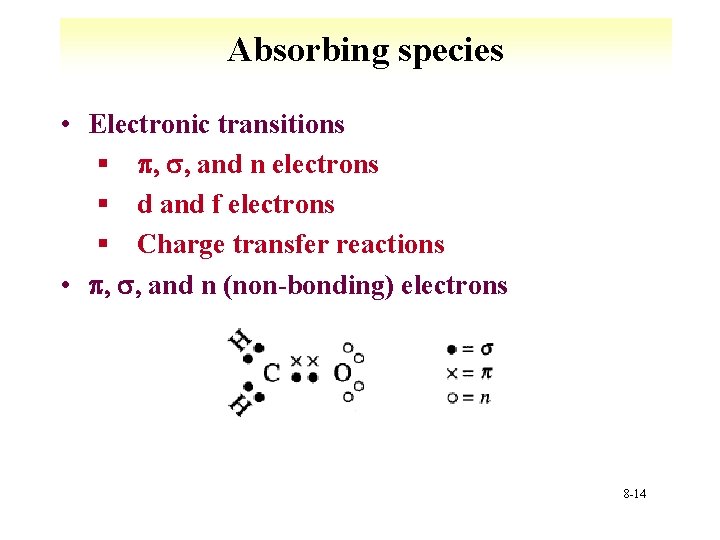
Absorbing species • Electronic transitions § p, s, and n electrons § d and f electrons § Charge transfer reactions • p, s, and n (non-bonding) electrons 8 -14
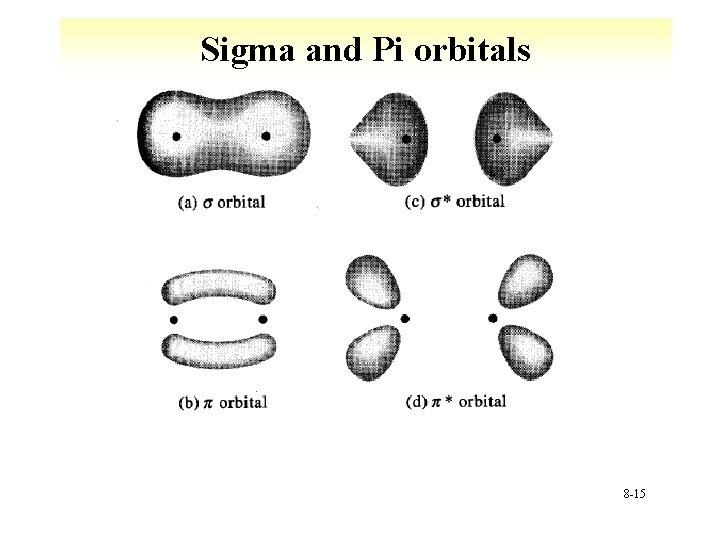
Sigma and Pi orbitals 8 -15
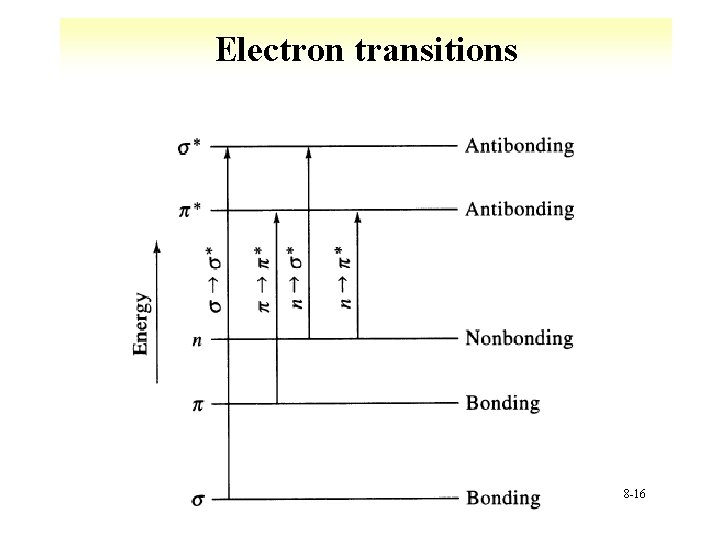
Electron transitions 8 -16

Transitions • s->s* § UV photon required, high energy à Methane at 125 nm à Ethane at 135 nm • n-> s* § Saturated compounds with unshared eà Absorption between 150 nm to 250 nm à e between 100 and 3000 L cm-1 mol-1 à Shifts to shorter wavelengths with polar solvents * Minimum accessibility § Halogens, N, O, S 8 -17

Transitions • n->p*, p->p* § Organic compounds, wavelengths 200 to 700 nm § Requires unsaturated groups àn->p* low e (10 to 100) * Shorter wavelengths àp->p* higher e (1000 to 10000) 8 -18
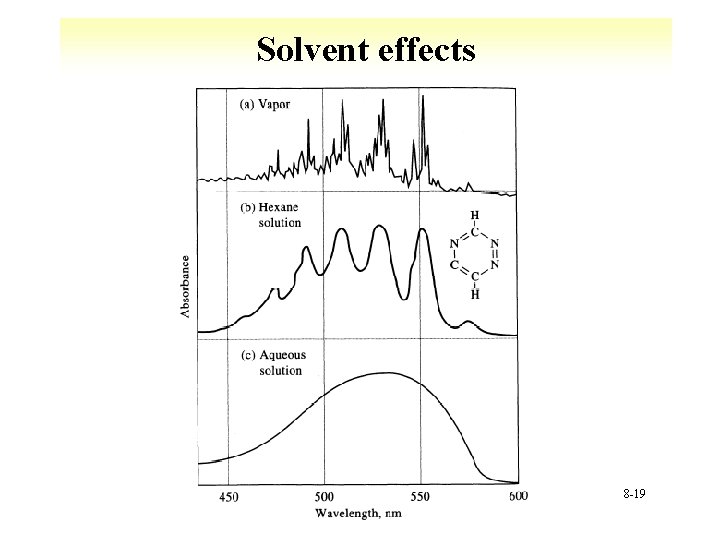
Solvent effects 8 -19

Transitions • d-d § 3 d and 4 d 1 st and 2 nd transitions series § Broad transitions àImpacted by solution 8 -20
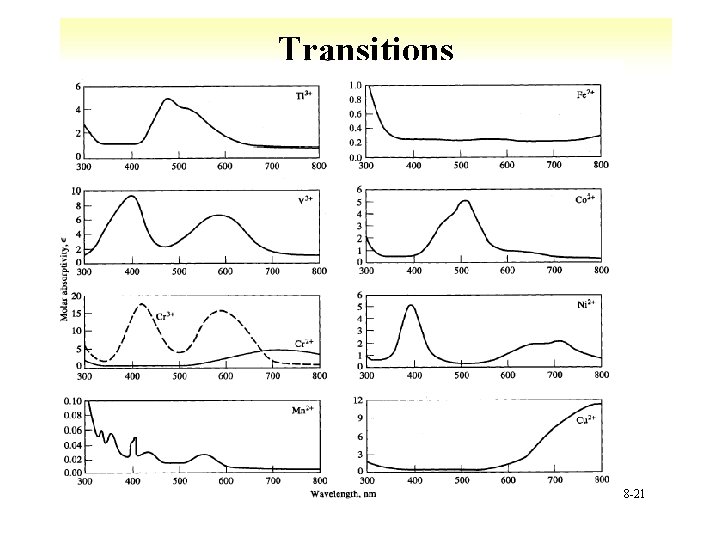
Transitions 8 -21
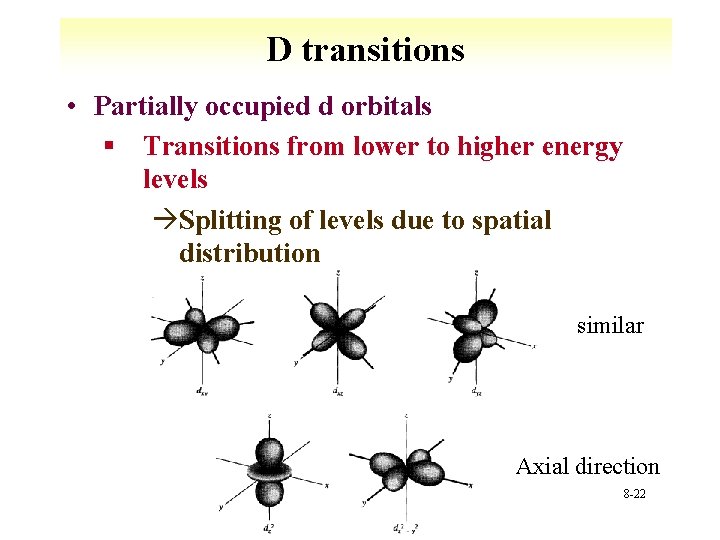
D transitions • Partially occupied d orbitals § Transitions from lower to higher energy levels àSplitting of levels due to spatial distribution similar Axial direction 8 -22
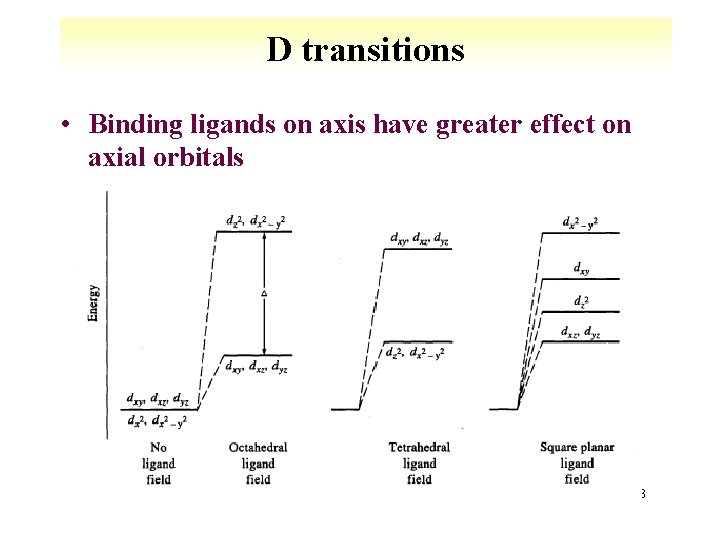
D transitions • Binding ligands on axis have greater effect on axial orbitals 8 -23

D transitions • D value dependent upon ligand field strength § <Br-<Cl-<F-<OH-<C 2 O 42 -~H 2 O<SCN<NH 3<en<NO 2 -<CN§ D increases with increasing field strength • f-f § 4 f and 5 f (lanthanides and actinides) § Sharper transitions 8 -24

Actinide transitions Figure 2: UV-vis spectra of organic phases for 13 M HNO 3 system 8 -25

Charge-transfer Transitions • Electron donor and acceptor characteristics § Absorption involves e- transitions from donor to acceptor àSCN to Fe(III) * Fe(II) and neutral SCN § Metal is acceptor àReduced metals can be exception 8 -26
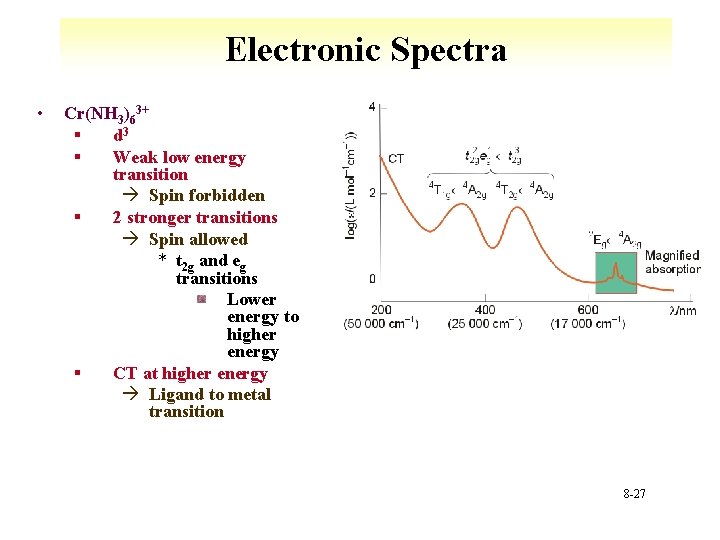
Electronic Spectra • Cr(NH 3)63+ § d 3 § Weak low energy transition à Spin forbidden § 2 stronger transitions à Spin allowed * t 2 g and eg transitions Lower energy to higher energy § CT at higher energy à Ligand to metal transition 8 -27
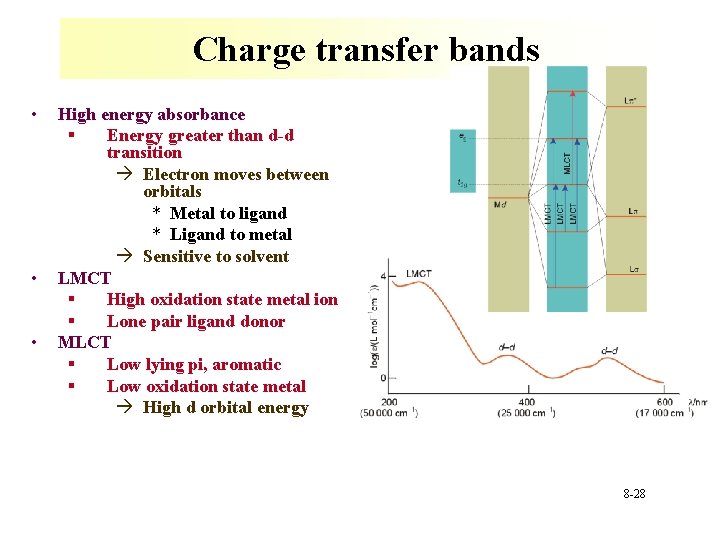
Charge transfer bands • • • High energy absorbance § Energy greater than d-d transition à Electron moves between orbitals * Metal to ligand * Ligand to metal à Sensitive to solvent LMCT § High oxidation state metal ion § Lone pair ligand donor MLCT § Low lying pi, aromatic § Low oxidation state metal à High d orbital energy 8 -28
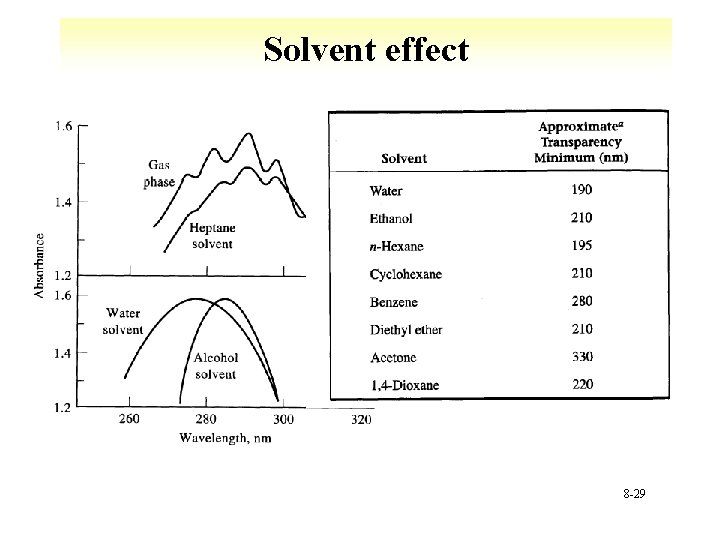
Solvent effect 8 -29

Methods • Titration § Change of absorbance with solution variation àp. H, ligand, metal • Photoacoustic effect § Emission of sound 8 -30
- Slides: 30