Ultrathin Bioresorbable Polymer SirolimusEluting Stents versus Thin Durable

Ultrathin Bioresorbable Polymer Sirolimus-Eluting Stents versus Thin Durable Polymer Everolimus-Eluting Stents in Patients Undergoing Coronary Revascularization Three-year Outcomes from the Randomized BIOFLOW V Trial David E. Kandzari, Jacques J. Koolen, Gheorghe Doros, Hector M. Garcia-Garcia, Johan Bennett, Ariel Roguin, Elie G. Gharib, Donald E. Cutlip, Ron Waksman, for the BIOFLOW V Investigators david. kandzari@piedmont. org @Kandzari

Perspective • Coronary drug-eluting stent development has included new metal alloys, changes in stent architecture and bioresorbable polymers • Whether these advancements improve long-term clinical safety and efficacy has been inconsistent in prior studies • BIOFLOW V Trial was a large, international RCT comparing an ultrathin strut (60 µm) bioresorbable polymer sirolimus-eluting stent (BP SES) and thin strut (81 µm) durable polymer everolimus eluting stent (DP EES) � Superior 1 - and 2 -year target lesion failure (TLF, primary endpoint) and target vessel myocardial infarction (MI) with BP SES versus DP EES; superior target lesion revascularization (TLR) at 2 years with BP SES versus D EES • Whether benefits favoring BP SES are maintained over longer term follow-up and might differences emerge regarding additional endpoints is uncertain Kandzari DE et al. Lancet 2017 Kandzari DE et al. JACC 2018

BIOFLOW V Trial Design BIOFLOW-V N=1, 334 2: 1 Randomization 884 BP SES, 450 DP EES • 1, 334 patients randomized between May 2015 and March 2016 � 884 Orsiro and 450 Xience 1 & 6 -month follow-up • Patients enrolled in 13 countries in North America, Europe, Israel, Asia, and Australia and New Zealand • Primary endpoint TLF at 1 year with planned follow-up through 5 years 1 -year follow-up TLF Primary Endpoint 3 -year follow-up Pre-specified Analysis 4, 5 -year follow-up clinicaltrials. gov NCT 02389946 Doros et al. Am Heart J. 2017; 193: 35 -45
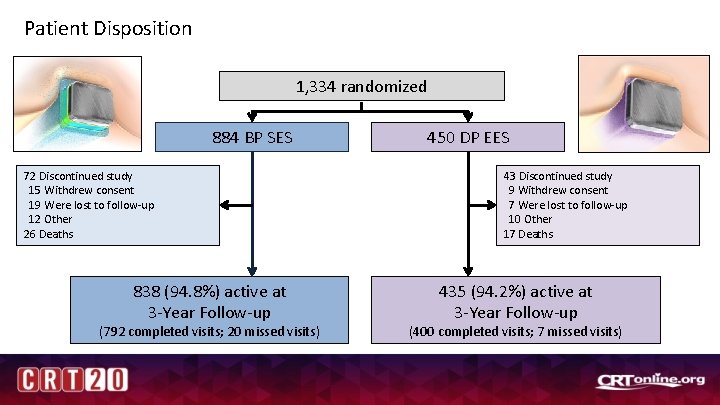
Patient Disposition 1, 334 randomized 884 BP SES 72 Discontinued study 15 Withdrew consent 19 Were lost to follow-up 12 Other 26 Deaths 838 (94. 8%) active at 3 -Year Follow-up (792 completed visits; 20 missed visits) 450 DP EES 43 Discontinued study 9 Withdrew consent 7 Were lost to follow-up 10 Other 17 Deaths 435 (94. 2%) active at 3 -Year Follow-up (400 completed visits; 7 missed visits)
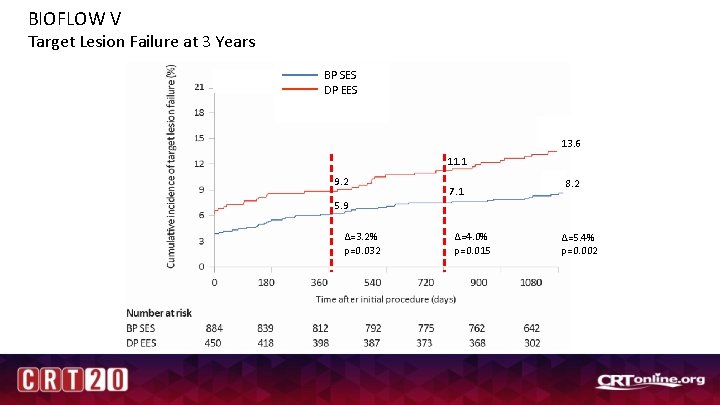
BIOFLOW V Target Lesion Failure at 3 Years BP SES DP EES 13. 6 11. 1 9. 2 5. 9 Δ=3. 2% p=0. 032 7. 1 Δ=4. 0% p=0. 015 8. 2 Δ=5. 4% p=0. 002
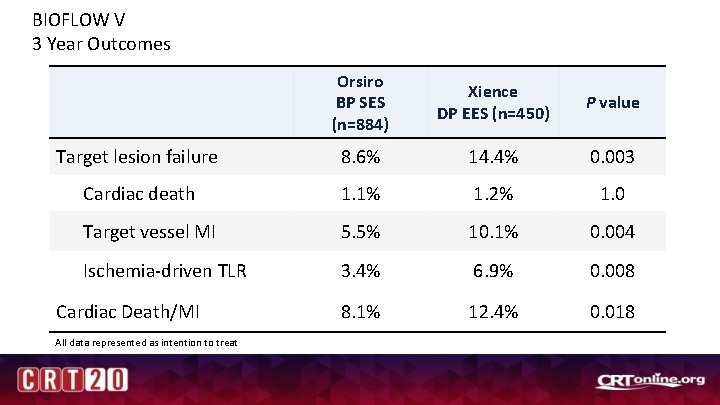
BIOFLOW V 3 Year Outcomes Orsiro BP SES (n=884) Xience DP EES (n=450) P value Target lesion failure 8. 6% 14. 4% 0. 003 Cardiac death 1. 1% 1. 2% 1. 0 Target vessel MI 5. 5% 10. 1% 0. 004 Ischemia-driven TLR 3. 4% 6. 9% 0. 008 8. 1% 12. 4% 0. 018 Cardiac Death/MI All data represented as intention to treat
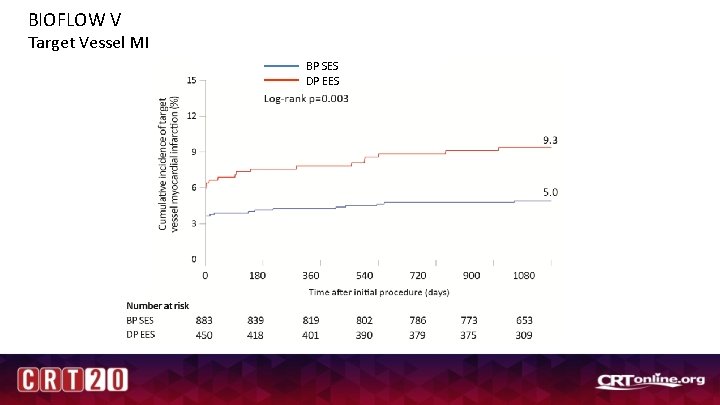
BIOFLOW V Target Vessel MI BP SES DP EES
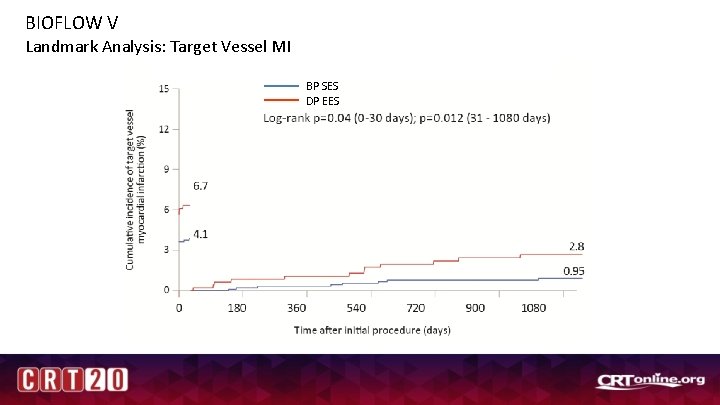
BIOFLOW V Landmark Analysis: Target Vessel MI BP SES DP EES
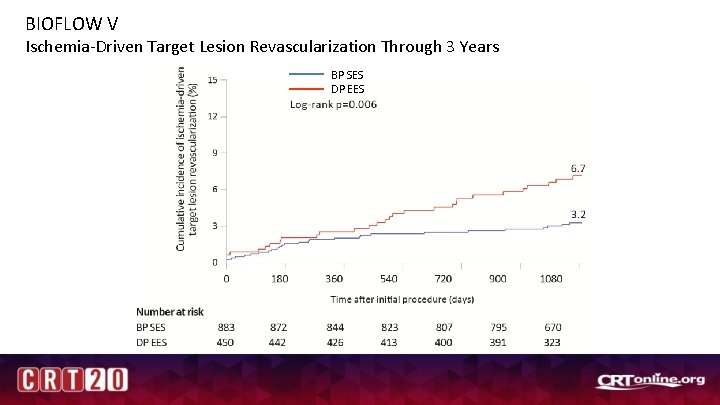
BIOFLOW V Ischemia-Driven Target Lesion Revascularization Through 3 Years BP SES DP EES Figure to be corrected
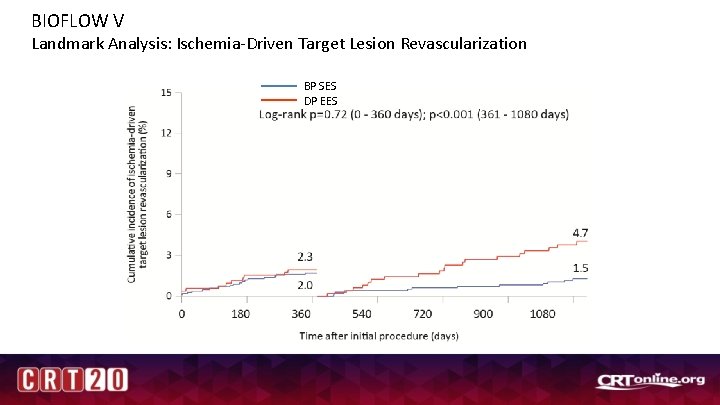
BIOFLOW V Landmark Analysis: Ischemia-Driven Target Lesion Revascularization BP SES DP EES
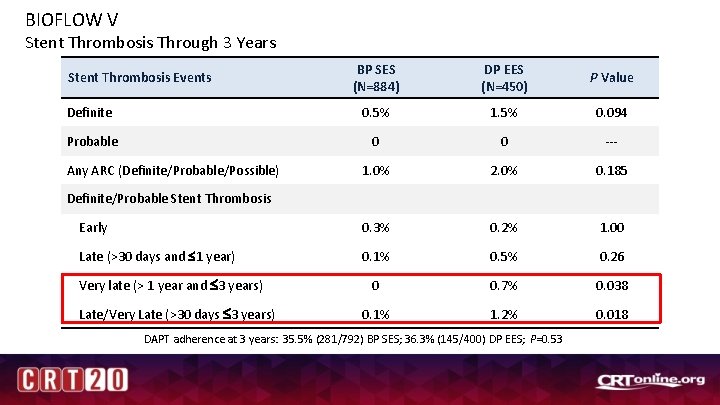
BIOFLOW V Stent Thrombosis Through 3 Years BP SES (N=884) DP EES (N=450) P Value Definite 0. 5% 1. 5% 0. 094 Probable 0 0 --- 1. 0% 2. 0% 0. 185 Early 0. 3% 0. 2% 1. 00 Late (>30 days and 1 year) 0. 1% 0. 5% 0. 26 0 0. 7% 0. 038 0. 1% 1. 2% 0. 018 Stent Thrombosis Events Any ARC (Definite/Probable/Possible) Definite/Probable Stent Thrombosis Very late (> 1 year and 3 years) Late/Very Late (>30 days 3 years) DAPT adherence at 3 years: 35. 5% (281/792) BP SES; 36. 3% (145/400) DP EES; P=0. 53

BIOFLOW V Conclusions • Through 3 years in the randomized BIOFLOW V trial, significant differences in TLF, target vessel MI, TLR and ST were maintained and continue to diverge in favor an ultrathin strut BP SES over a contemporary generation thin strut DP EES � Lower TLF (8. 6% vs 14. 4%, P=0. 003) � Lower target vessel related MI (5. 5% vs 10. 1%, P=0. 004), both early (≤ 30 days) and late (31 days to 3 years) � Lower ischemia-driven TLR, driven by differences in late (>1 year) TLR (3. 4% vs 6. 9%, P=0. 008) � Lower late/very definite/probable ST (0. 1% vs 1. 2%, P=0. 018) and emergence of very late ST (0 vs 0. 7%, P=0. 038) • These results both advance a new standard of comparison for DES but also direct attention to strut thickness and polymer composition as key features for iterative DES development Kandzari et al. JACC Cardiovasc Interv 2020 In Press Kandzari et al. JACC Cardiovasc Interv 2020


Orsiro Ultrathin Strut (BP SES) Stent System Stent material L-605 Cobalt-Chromium Strut thickness 60 µm* Polymer material Poly-L-lactic acid (PLLA) Polymer type Bioresorbable, asymmetric circumferential thickness Passive coating Amorphous silicon carbide Antiproliferative drug Sirolimus (1. 4 µg/mm 2), >80% eluted in first 90 days *For 2. 25 mm to 3. 0 mm diameter stents, 80 µm for >3. 0 mm diameter stents BIOTRONIK AG, Bülach, Switzerland

Key Enrollment Criteria Inclusion Criteria • • Age ≥ 18 years IHD, stable or unstable angina, or silent ischemia ≤ 3 de novo target lesions in ≤ 2 native target vessels (TV) RVD ≥ 2. 25 mm and ≤ 4. 0 mm LL ≤ 36 mm TIMI flow > 1 Eligible for DAPT therapy (aspirin + P 2 Y 12) Provided informed consent Doros et al. Am Heart J. 2017; 193: 35 -45 Exclusion Criteria • • • Recent (< 72 hours prior to procedure) STEMI or hemodynamically unstable NSTEMI/ ACS patients Chronic total occlusions, bypass grafts Bifurcations with side branch > 2. 0 mm In-stent restenosis or active stent thrombosis LVEF < 30% Prior PCI within 30 days (non-TV) or within 9 months (TV) Planned staged PCI post-procedure Renal impairment > 2. 5 mg/d. L or 221 µmol/L or dialysis dependent Excessively tortuous/ angulated or severely calcified (operator visual assessment)
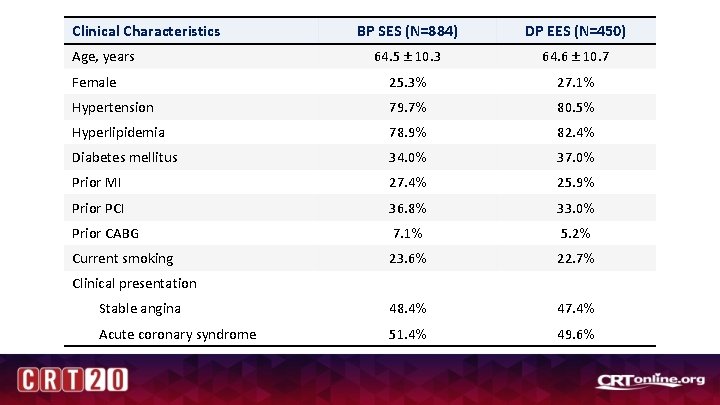
Clinical Characteristics BP SES (N=884) DP EES (N=450) 64. 5 ± 10. 3 64. 6 ± 10. 7 Female 25. 3% 27. 1% Hypertension 79. 7% 80. 5% Hyperlipidemia 78. 9% 82. 4% Diabetes mellitus 34. 0% 37. 0% Prior MI 27. 4% 25. 9% Prior PCI 36. 8% 33. 0% Prior CABG 7. 1% 5. 2% Current smoking 23. 6% 22. 7% Stable angina 48. 4% 47. 4% Acute coronary syndrome 51. 4% 49. 6% Age, years Clinical presentation
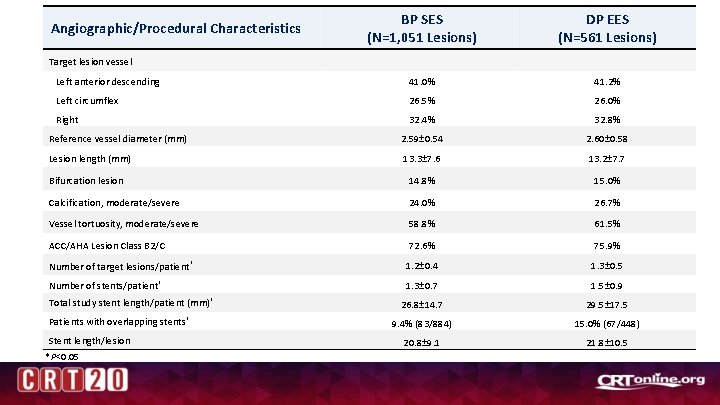
BP SES (N=1, 051 Lesions) DP EES (N=561 Lesions) Left anterior descending 41. 0% 41. 2% Left circumflex 26. 5% 26. 0% Right 32. 4% 32. 8% Reference vessel diameter (mm) 2. 59± 0. 54 2. 60± 0. 58 Lesion length (mm) 13. 3± 7. 6 13. 2± 7. 7 Bifurcation lesion 14. 8% 15. 0% Calcification, moderate/severe 24. 0% 26. 7% Vessel tortuosity, moderate/severe 58. 8% 61. 5% ACC/AHA Lesion Class B 2/C 72. 6% 75. 9% Number of target lesions/patient* 1. 2± 0. 4 1. 3± 0. 5 Number of stents/patient* 1. 3± 0. 7 1. 5± 0. 9 26. 8± 14. 7 29. 5± 17. 5 9. 4% (83/884) 15. 0% (67/448) 20. 8± 9. 1 21. 8± 10. 5 Angiographic/Procedural Characteristics Target lesion vessel Total study stent length/patient (mm)* Patients with overlapping stents* Stent length/lesion *P<0. 05
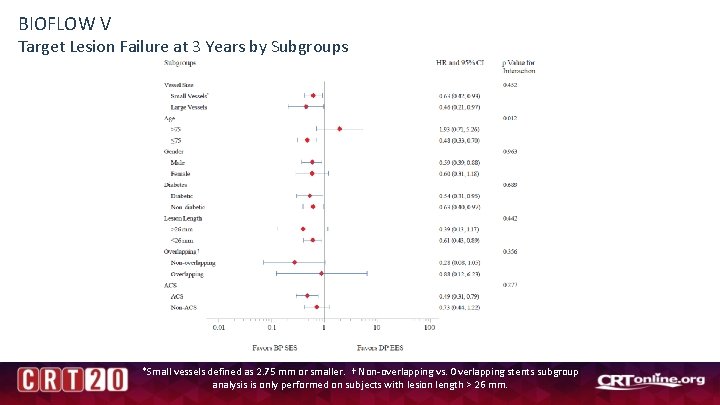
BIOFLOW V Target Lesion Failure at 3 Years by Subgroups *Small vessels defined as 2. 75 mm or smaller. † Non-overlapping vs. Overlapping stents subgroup analysis is only performed on subjects with lesion length > 26 mm.
- Slides: 18