Ultrasound Assisted Thrombolysis for Massive and Submassive Pulmonary

Ultrasound Assisted Thrombolysis for Massive and Submassive Pulmonary Embolism Scott M Lilly, MD Ph. D Interventional Cardiology Fellows School August 15 th, 2014

Acknowledgements § Peter Bittenbender, MD § Ray Magorien, MD § Michael Bray, EKOS Corporation

Outline § Background and Definitions § How to Determine Risk § Treatment of High Risk patients § Practical Points and Program Experience

Background and Definitions § 300 k-600 k per year § 1 -2 per 1000 people, or as high as 1 in 100 if > 80 § 10 -30% overall 30 day mortality § Sudden death is presenting symptom in ~ 25% § 2012: 166, 665 primary admissions for PE § In-hospital mortality ~ 3% § Most commonly from lower extremity DVT § Evidence of DVT in > 50% cdc. gov; Agency for Healthcare Research and Quality 4
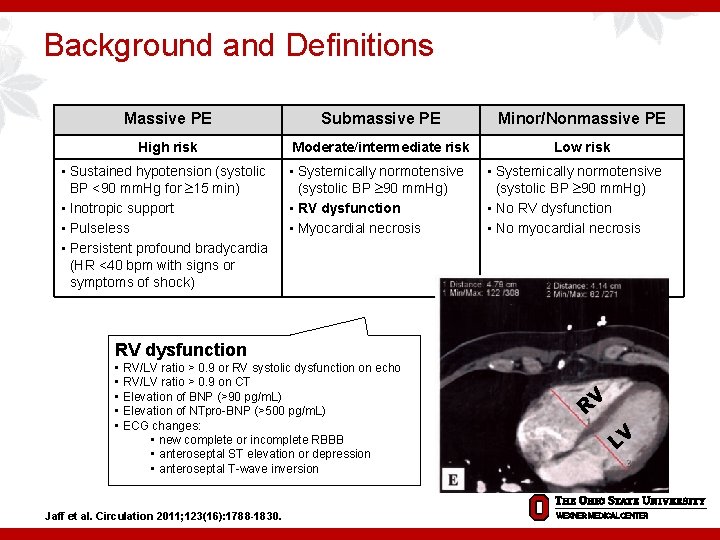
Background and Definitions Massive PE Submassive PE Minor/Nonmassive PE High risk Moderate/intermediate risk Low risk • Sustained hypotension (systolic BP <90 mm. Hg for 15 min) • Inotropic support • Pulseless • Persistent profound bradycardia (HR <40 bpm with signs or symptoms of shock) • Systemically normotensive (systolic BP 90 mm. Hg) • RV dysfunction • Myocardial necrosis • Systemically normotensive (systolic BP 90 mm. Hg) • No RV dysfunction • No myocardial necrosis RV dysfunction • RV/LV ratio > 0. 9 or RV systolic dysfunction on echo • RV/LV ratio > 0. 9 on CT • Elevation of BNP (>90 pg/m. L) • Elevation of NTpro-BNP (>500 pg/m. L) • ECG changes: • new complete or incomplete RBBB • anteroseptal ST elevation or depression • anteroseptal T-wave inversion Jaff et al. Circulation 2011; 123(16): 1788 -1830. RV LV Jaff et al. Circulation 2011; 123(16): 1788 -1830.
![Background and Definitions Massive PE [High risk] 5% PE population Minor PE [Low risk] Background and Definitions Massive PE [High risk] 5% PE population Minor PE [Low risk]](http://slidetodoc.com/presentation_image_h2/a00e0d07e369717466da28ecc6d88555/image-6.jpg)
Background and Definitions Massive PE [High risk] 5% PE population Minor PE [Low risk] 55% PE population Submassive PE [Moderate risk] 40% PE population

Outline § Background and Definitions § How to Determine Risk § Treatment of High Risk patients § Practical Points and Program Experience
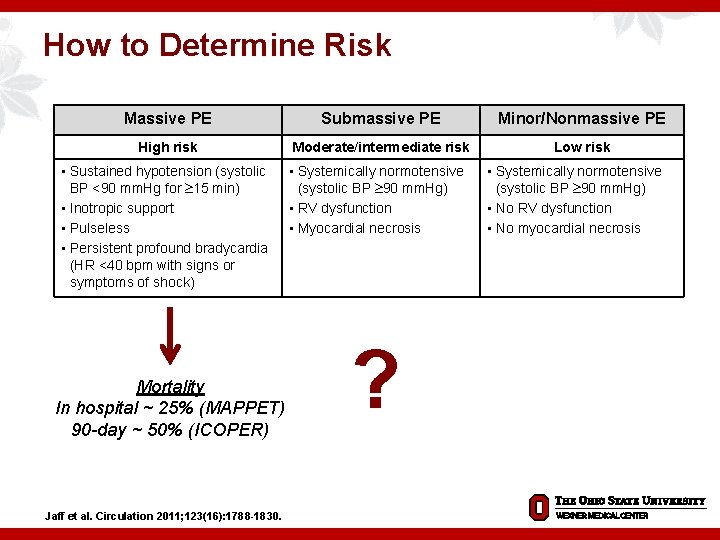
How to Determine Risk Massive PE Submassive PE Minor/Nonmassive PE High risk Moderate/intermediate risk Low risk • Sustained hypotension (systolic BP <90 mm. Hg for 15 min) • Inotropic support • Pulseless • Persistent profound bradycardia (HR <40 bpm with signs or symptoms of shock) Mortality In hospital ~ 25% (MAPPET) 90 -day ~ 50% (ICOPER) Jaff et al. Circulation 2011; 123(16): 1788 -1830. • Systemically normotensive (systolic BP 90 mm. Hg) • RV dysfunction • Myocardial necrosis ? • Systemically normotensive (systolic BP 90 mm. Hg) • No RV dysfunction • No myocardial necrosis
How to Determine Risk Massive PE Submassive PE Minor/Nonmassive PE High risk Moderate/intermediate risk Low risk • Sustained hypotension (systolic BP <90 mm. Hg for 15 min) • Inotropic support • Pulseless • Persistent profound bradycardia (HR <40 bpm with signs or symptoms of shock) Jaff et al. Circulation 2011; 123(16): 1788 -1830. • Systemically normotensive (systolic BP 90 mm. Hg) • RV dysfunction • Myocardial necrosis • Systemically normotensive (systolic BP 90 mm. Hg) • No RV dysfunction • No myocardial necrosis

How to Determine Risk − Registry of 1, 416 patients − Mortality rate: 1. 9% if RV/LV ratio < 0. 9 6. 6% if RV/LV ratio ≥ 0. 9 Fremont et al. CHEST 2008; 133: 358 -362 10

How to Determine Risk − Retrospective analysis of 120 patients with hemodynamically stable PE based on chest CT − PE-related mortality at 3 months: § 17% if RV/LV ≥ 1. 5 § 8% if 1. 0 ≤ RV/LV < 1. 5 § 0% if RV/LV < 1. 0 Van der Meer et al. Radiology 2005; 235: 798 -803. 11

How to Determine Risk − Retrospective analysis of 63 patients with chest CT − Adverse event rate at 30 days: § 80. 3% if RV/LV ratio > 0. 9 § 51. 3% if RV/LV ratio ≤ 0. 9 Quiroz et. al. Circulation. 2004; 109: 2401 -2404 12
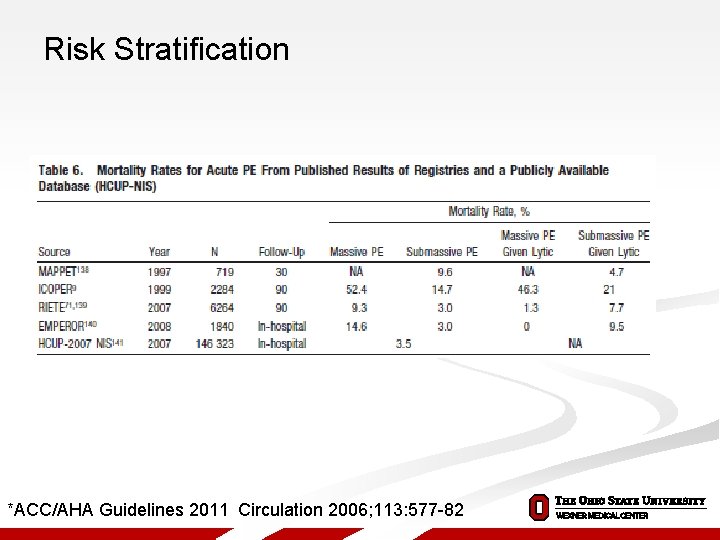
Risk Stratification *ACC/AHA Guidelines 2011 Circulation 2006; 113: 577 -82

Outline § Background and Definitions § How to Determine Risk § Treatment of High Risk patients § Practical Points and Program Experience

Treatment of High Risk patients Degree of PE Treatment* Bleeding Risk Non-Massive Heparin (I) Less Sub-Massive Lytics (IIb) Massive Lytics (IIa) More 20% risk of major bleeding 3% risk of intracranial hemorrhage *ACC/AHA Guidelines 2011 Circulation 2006; 113: 577 -82
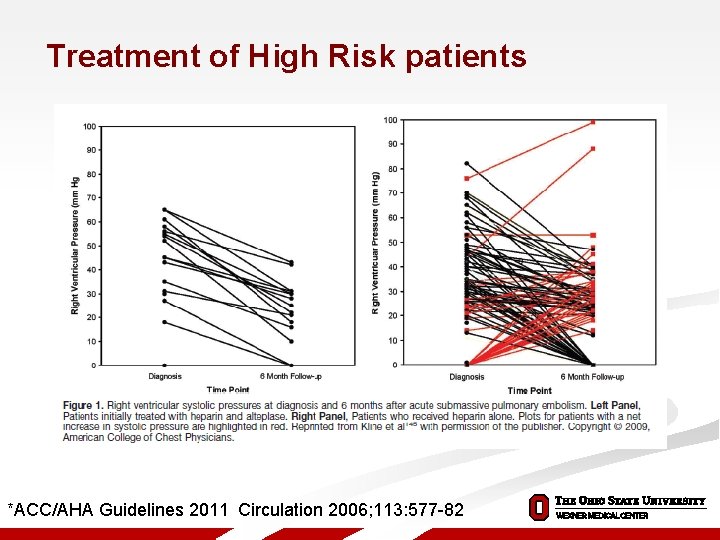
Treatment of High Risk patients *ACC/AHA Guidelines 2011 Circulation 2006; 113: 577 -82
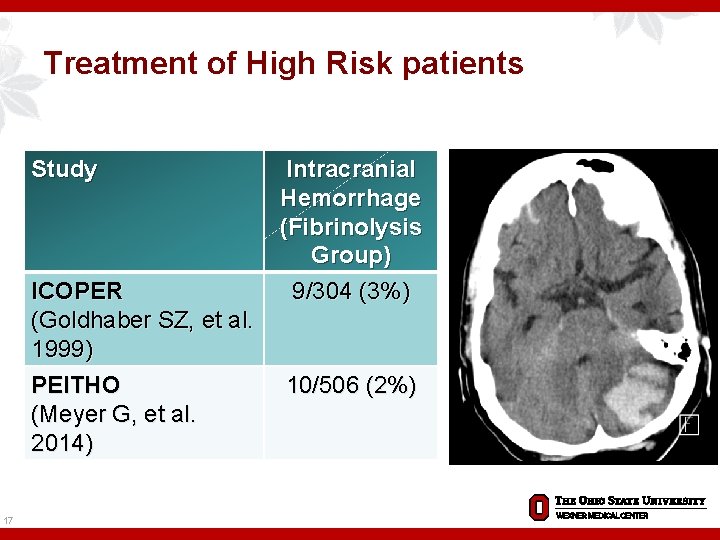
Treatment of High Risk patients Study ICOPER (Goldhaber SZ, et al. 1999) PEITHO (Meyer G, et al. 2014) 17 Intracranial Hemorrhage (Fibrinolysis Group) 9/304 (3%) 10/506 (2%)
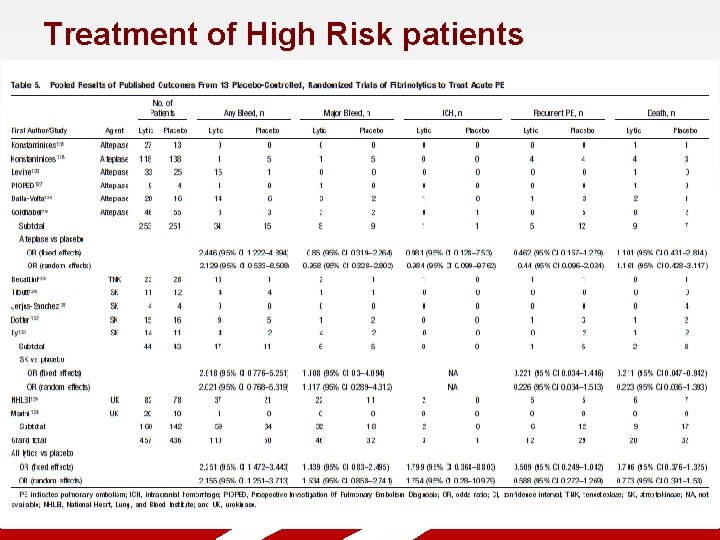
Treatment of High Risk patients Status of Trials

Treatment of High Risk patients The ULTIMA Trial A Prospective, Randomized, Controlled Study of Ultrasound Accelerated Thrombolysis for the Treatment of Acute Pulmonary Embolism Annual Meeting of the American College of Cardiology, March 9, 2013

Treatment of High Risk patients The ULTIMA Trial ULTrasound Accelerated Thrombolys. Is of Pul. Mon. Ary Embolism Hypothesis: Ultrasound-assisted, catheter-directed thrombolysis is superior to treatment with heparin alone for reversing RV enlargement within 24 hours 22
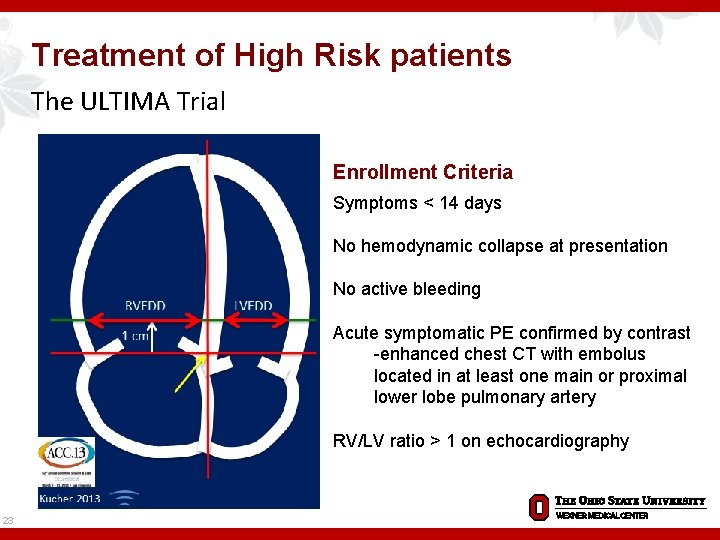
Treatment of High Risk patients The ULTIMA Trial Enrollment Criteria Symptoms < 14 days No hemodynamic collapse at presentation No active bleeding Acute symptomatic PE confirmed by contrast -enhanced chest CT with embolus located in at least one main or proximal lower lobe pulmonary artery RV/LV ratio > 1 on echocardiography 23
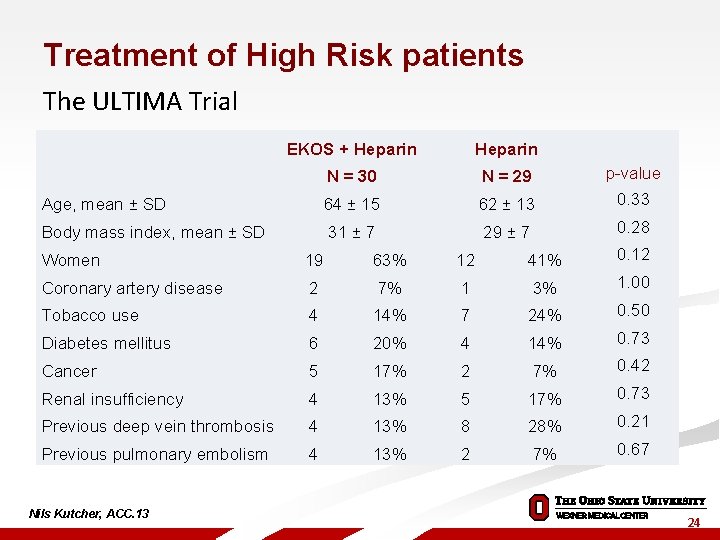
Treatment of High Risk patients The ULTIMA Trial EKOS + Heparin N = 30 N = 29 p-value Age, mean ± SD 64 ± 15 62 ± 13 0. 33 Body mass index, mean ± SD 31 ± 7 29 ± 7 0. 28 Women 19 63% 12 41% 0. 12 Coronary artery disease 2 7% 1 3% 1. 00 Tobacco use 4 14% 7 24% 0. 50 Diabetes mellitus 6 20% 4 14% 0. 73 Cancer 5 17% 2 7% 0. 42 Renal insufficiency 4 13% 5 17% 0. 73 Previous deep vein thrombosis 4 13% 8 28% 0. 21 Previous pulmonary embolism 4 13% 2 7% 0. 67 Nils Kutcher, ACC. 13 24

Treatment of High Risk patients The ULTIMA Trial Troponin test positive, n (%) Pulmonary occlusion score (CT)1, mean ± SD Pulmonary occlusion score (CT)1, median (min-max) 1 1 1 20 5 1 1 1 2 2 1 1 10 10 1 1 Qanadli Nils Kutcher, ACC. 13 Heparin N = 30 N = 29 p-value 16/20 (80%) 17/22 (77%) 1. 00 26 ± 7 24 ± 8 0. 24 26 (9 -36) 22 (13 -38) 1 3 3 EKOS + Heparin 5 1 Pulmonary occlusion score 1 • Multiply score points for non-occlusive embolus by one • Multiply score points for occlusive embolus by two • Maximum score is 40. 1 1 1 Am J Roentgenology 2001; 176: 1415 -20 25

Treatment of High Risk patients The ULTIMA Trial EKOS + Heparin N = 30 Heparin N = 29 p-value 30939 ± 7907 34277 ± 5632 0. 08 100% - Device placement time, minutes, median (min-max) 42 (15 – 102) - Dual device procedure 25 (83%) - Total rt-PA dose two devices, mg 20. 7 ± 2. 5 - 5 (17%) - Total rt-PA dose single device, mg 12. 2 ± 3. 8 - Length of stay in ICU/IMC, days 2. 6 ± 1. 3 1. 7 ± 1. 3 0. 01 Length of hospital stay, days 8. 8 ± 3. 5 8. 7 ± 3. 9 0. 88 Total heparin dose from randomization to 24 hours, IU Technical success device placement Single device procedure Nils Kutcher, ACC. 13 26
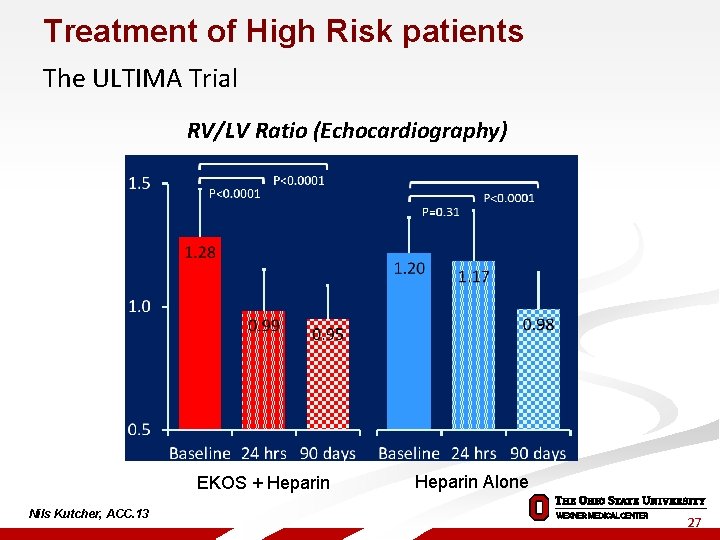
Treatment of High Risk patients The ULTIMA Trial RV/LV Ratio (Echocardiography) EKOS + Heparin Nils Kutcher, ACC. 13 Heparin Alone 27
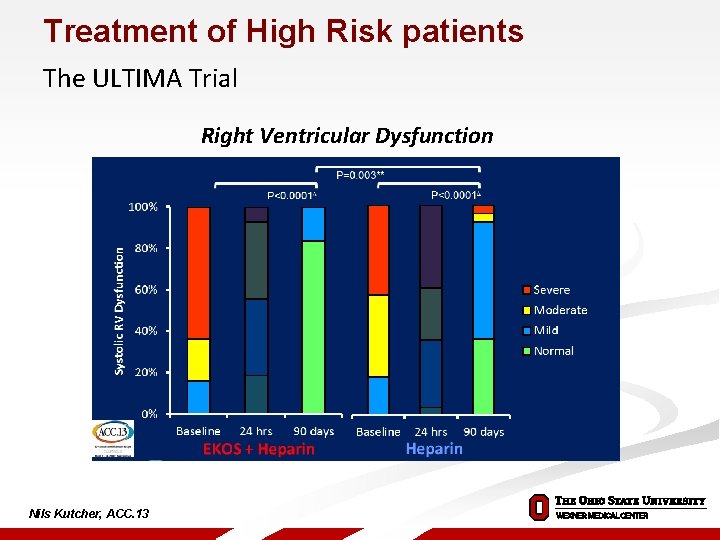
Treatment of High Risk patients The ULTIMA Trial Right Ventricular Dysfunction Nils Kutcher, ACC. 13

Treatment of High Risk patients The ULTIMA Trial EKOS + Heparin N = 30 N = 29 Clinical outcomes at 90 days p-value Death 0 0% 1* 3% 0. 49 Recurrent venous thromboembolism 0 0% 1. 00 Major bleeding 0 0% 1. 00 Minor bleeding 3** 10% 1§ 3% 0. 61 • rehospitalization and death from advanced pancreatic cancer ** two patients with transient mild hemoptysis without medical intervention, one patient with groin hematoma requiring manual compression § one patient with transient anal bleeding following endoscopic removal of colon polyp 29
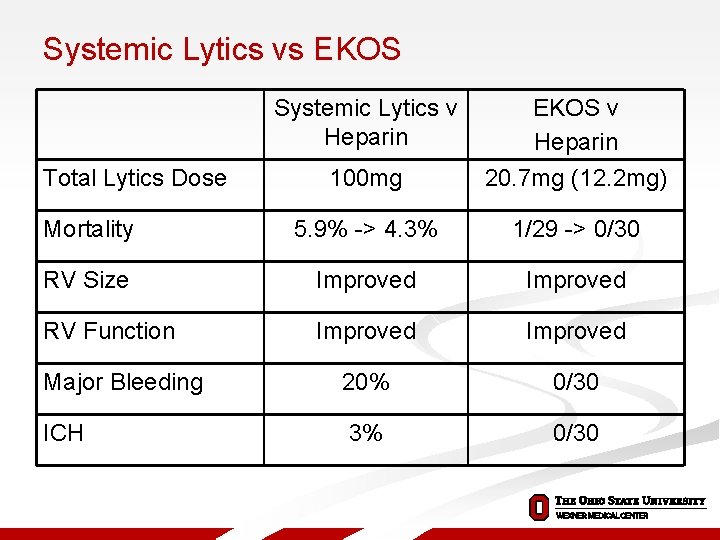
Systemic Lytics vs EKOS Systemic Lytics v Heparin 100 mg EKOS v Heparin 20. 7 mg (12. 2 mg) Mortality 5. 9% -> 4. 3% 1/29 -> 0/30 RV Size Improved RV Function Improved Major Bleeding 20% 0/30 ICH 3% 0/30 Total Lytics Dose

Treatment of High Risk patients The SEATTLE II Trial A Prospective, Single-Arm, Multicenter Trial of Ultrasound-Facilitated, Low-Dose Fibrinolysis for Acute Massive and Submassive Pulmonary Embolism (SEATTLE II)

Treatment of High Risk patients The SEATTLE II Trial A prospective, single-arm, multicenter trial to: Evaluate the efficacy of ultrasound-facilitated, catheter-directed low-dose fibrinolysis to reverse RV dysfunction as measured by CT-determined RV/LV diameter ratio in patients with acute massive and submassive PE Assess the safety of ultrasound-facilitated, catheter -directed low-dose fibrinolysis in patients with acute massive and submassive PE

Treatment of High Risk patients The SEATTLE II Trial Main Inclusion Criteria: Main Exclusion Criteria: § Proximal PE on CT (filling defect in ≥ 1 main, lobar, or segmental pulmonary artery) AND § Age ≥ 18 years AND § Stroke/TIA, head trauma, or intracranial or intraspinal disease within 1 year § Active or recent (within 1 month) bleeding from a major organ § Major surgery within 7 days § PE symptom duration ≤ 14 days AND § Massive PE (syncope, systemic arterial hypotension, cardiogenic shock, or resuscitated cardiac arrest) OR § Submassive PE (RV/LV diameter ≥ 0. 9 on contrast-enhanced chest CT) 33 § Hematocrit < 30%, platelets < 100 k/μL, INR > 3, a. PTT > 50 seconds on no anticoagulation § Serum creatinine > 2 mg/d. L § Clinician-determined high-risk for catastrophic bleeding § Hemodynamic instability despite medical therapy § Pregnancy
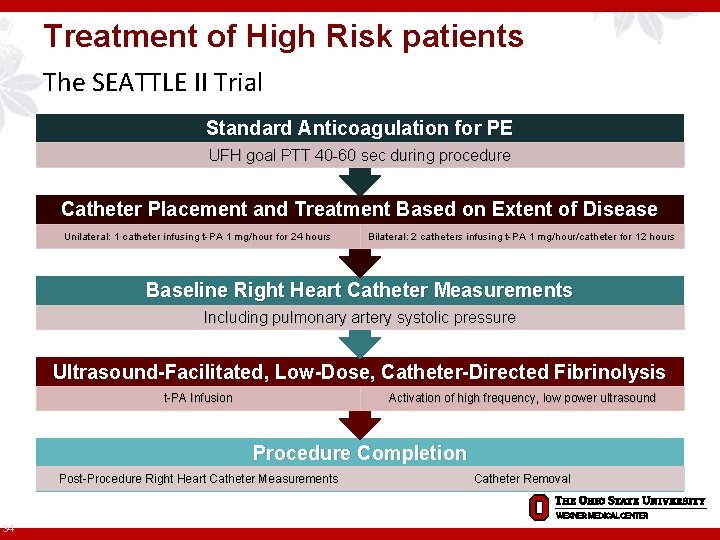
Treatment of High Risk patients The SEATTLE II Trial Standard Anticoagulation for PE UFH goal PTT 40 -60 sec during procedure Catheter Placement and Treatment Based on Extent of Disease Unilateral: 1 catheter infusing t-PA 1 mg/hour for 24 hours Bilateral: 2 catheters infusing t-PA 1 mg/hour/catheter for 12 hours Baseline Right Heart Catheter Measurements Including pulmonary artery systolic pressure Ultrasound-Facilitated, Low-Dose, Catheter-Directed Fibrinolysis t-PA Infusion Activation of high frequency, low power ultrasound Procedure Completion Post-Procedure Right Heart Catheter Measurements 34 Catheter Removal
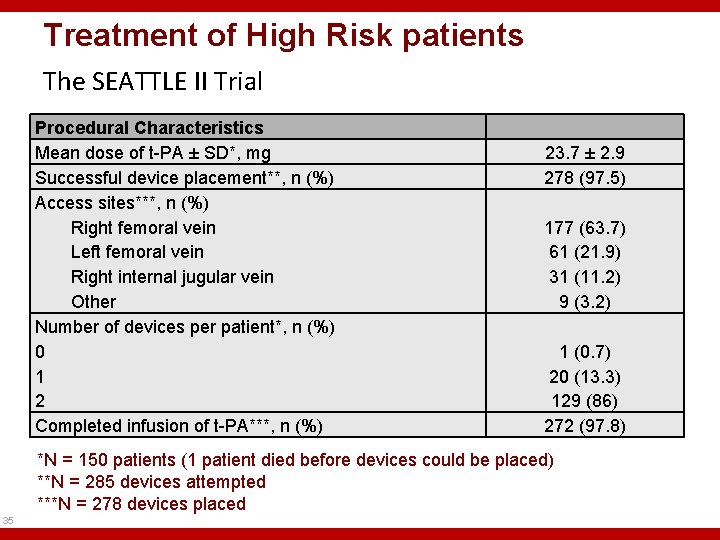
Treatment of High Risk patients The SEATTLE II Trial Procedural Characteristics Mean dose of t-PA ± SD*, mg Successful device placement**, n (%) Access sites***, n (%) Right femoral vein Left femoral vein Right internal jugular vein Other Number of devices per patient*, n (%) 0 1 2 Completed infusion of t-PA***, n (%) 23. 7 ± 2. 9 278 (97. 5) 177 (63. 7) 61 (21. 9) 31 (11. 2) 9 (3. 2) 1 (0. 7) 20 (13. 3) 129 (86) 272 (97. 8) *N = 150 patients (1 patient died before devices could be placed) **N = 285 devices attempted ***N = 278 devices placed 35
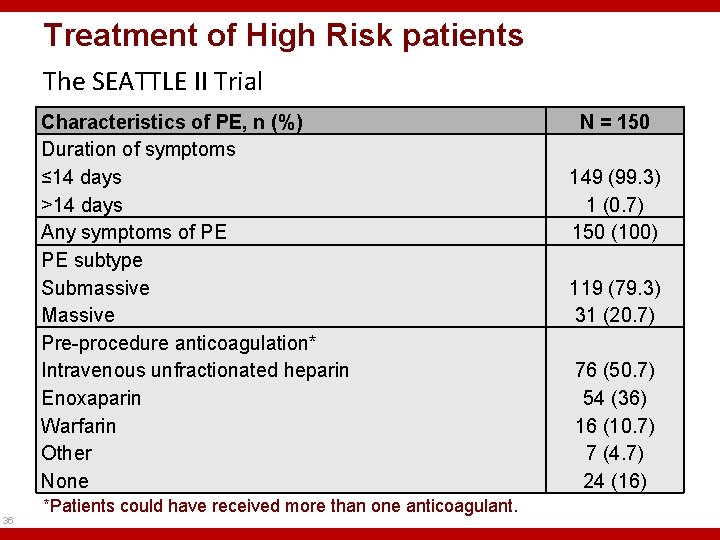
Treatment of High Risk patients The SEATTLE II Trial Characteristics of PE, n (%) Duration of symptoms ≤ 14 days >14 days Any symptoms of PE PE subtype Submassive Massive Pre-procedure anticoagulation* Intravenous unfractionated heparin Enoxaparin Warfarin Other None 36 *Patients could have received more than one anticoagulant. N = 150 149 (99. 3) 1 (0. 7) 150 (100) 119 (79. 3) 31 (20. 7) 76 (50. 7) 54 (36) 16 (10. 7) 7 (4. 7) 24 (16)
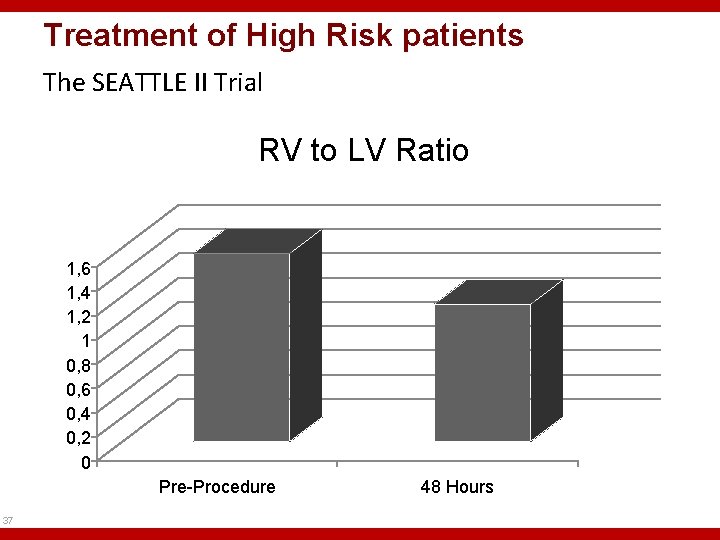
Treatment of High Risk patients The SEATTLE II Trial RV to LV Ratio 1, 6 1, 4 1, 2 1 0, 8 0, 6 0, 4 0, 2 0 Pre-Procedure 37 48 Hours
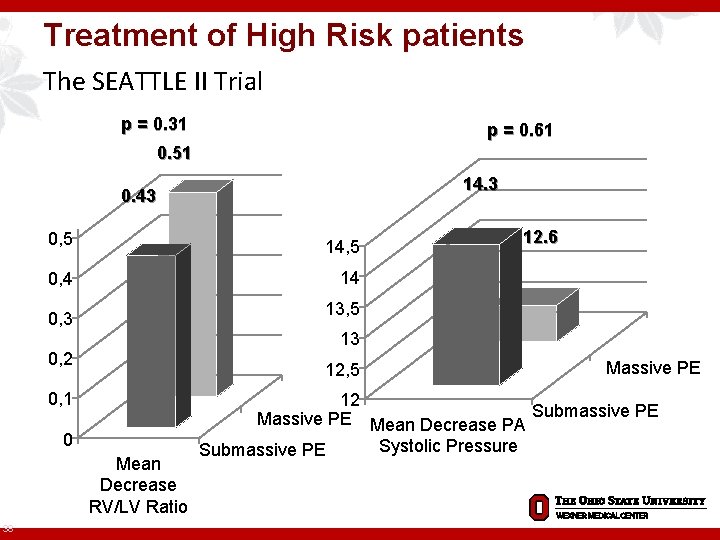
Treatment of High Risk patients The SEATTLE II Trial p = 0. 31 p = 0. 61 0. 51 14. 3 0. 43 0, 5 14, 5 0, 4 14 13, 5 0, 3 13 0, 2 12, 5 0, 1 0 Mean Decrease RV/LV Ratio 38 12. 6 Massive PE 12 Massive PE Mean Decrease PA Submassive PE Systolic Pressure Submassive PE
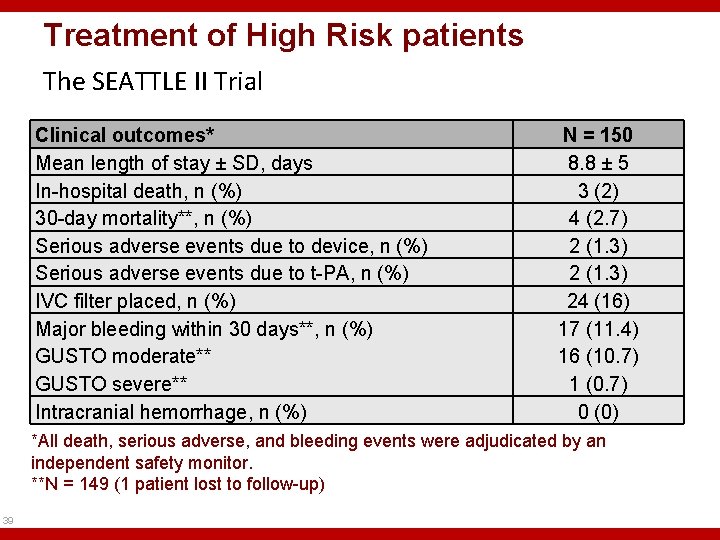
Treatment of High Risk patients The SEATTLE II Trial Clinical outcomes* Mean length of stay ± SD, days In-hospital death, n (%) 30 -day mortality**, n (%) Serious adverse events due to device, n (%) Serious adverse events due to t-PA, n (%) IVC filter placed, n (%) Major bleeding within 30 days**, n (%) GUSTO moderate** GUSTO severe** Intracranial hemorrhage, n (%) N = 150 8. 8 ± 5 3 (2) 4 (2. 7) 2 (1. 3) 24 (16) 17 (11. 4) 16 (10. 7) 1 (0. 7) 0 (0) *All death, serious adverse, and bleeding events were adjudicated by an independent safety monitor. **N = 149 (1 patient lost to follow-up) 39

Treatment of High Risk patients The SEATTLE II Trial Study 40 Intracranial Hemorrhage (Fibrinolysis Group) ICOPER (Goldhaber SZ, et al. 1999) 9/304 (3%) PEITHO (Meyer G, et al. 2014) 10/506 (2%) SEATTLE II (Piazza G, et al. 2014) 0/150 (0%)

Treatment of High Risk patients Status of Trials § Lysis vs Placebo § § § 13 placebo controlled, randomized trials of lysis vs placebo Minority for massive PE, total 480 patients. Variable drugs, dosing, timing and adjunctive therapies No independent mortality effect Meta-analyses reduction in death/recurrent PE Improvement in RV size/function, m. PA pressures § EKOS v Heparin § No study large enough to evaluate death/recurrent PE § Improved RV size/function at 24 hrs, catch up at 90 days § Improved RV function at 90 days

Outline § Background and Definitions § How to Determine Risk § Treatment of High Risk patients § Practical Points, Program Experience

Who is an EKOS Candidate? § Large, Central PE – symptoms <14 days § § SBP<90 (not responsive to fluids) Need for inotrope HR<40 w/ s/s Shock PEA (after return of circulation) § RV: LV ratio > 1. 0 (CTPE) § RV: LV ratio > 1. 0 (TTE) § Signs of RV dysfxn (TTE) MASSIVE SUBMASSIVE Activate a PE Alert by dialing the Transfer Center Hotline phone number: 366 -8111

The PE Guideline is Located on One Source Listed under “Venous Thromboembolism” 44
- Slides: 44