UltraLongActing Basal Insulin Analogues Seyed Adel Jahed MD


Ultra-Long-Acting Basal Insulin Analogues Seyed Adel Jahed, MD Islamic Azad University, Tehran Medical Branch 14 rd June 2012 RIES, Tehran, Iran

AGENDA §Why Search For Ultra-long-actings? §The Pipeline Of The Ultra-long-actings §Overview Of Insulin Degludec, its PK and PD §Clinical evidences in Degludec usage

§ Despite 90 years of insulin use and advances in therapy for patients with diabetes, treatment remains associated with notable limitations. § Attempts to achieve near normoglycemia with subcutaneous insulin continue to raise important questions, including: • The choice of preparation • Injection regimen • How to reduce incidence of hypoglycemia • How to prevent excess weight gain • How to avoid other effects of inappropriate hyperinsulinemia Tahrani AA, et al. Insulin degludec: a new ultra-long-acting insulin. The Lancet 2012; 379: 1465 -7.

Using Insulin: Importance of Ease of Use § Patients with DM spend >2 hr/day on self care, which includes glucose monitoring, sorting medications, deciding about insulin dosing, and dietary decisions. § A recent trend in many disorders, including DM 2, has been the introduction of long-acting drugs that could be administered less than daily to improve adherence while minimizing side effects. Russell LB. Time requirements for diabetes self-management: too much for many? J Fam Pract 2005; 54: 52 – 6

Which Properties of Currently Available Long-Acting Analogues Could Be Improved? § § § § Predictable flat or near-flat time–action profile Low or no variability Longer duration of action Soluble to allow mixing with rapid-acting analog Low costs Less or no weight gain No signal of increased mitogenicity Simon ACR, et al. The Future of Basal Insulin Supplementation. Diabetes Technology & Therapeutics 2011; 13, Suppl. 1, S 103 -8

The Pipeline Of The Ultra-Long-Actings § Sanofi: Sanofi The development of SAR 161271, an improved long-acting insulin, was discontinued according to business analysts. § Biodel: Biodel 2 formulations in the pipeline, both in the preclinical development phase : ü ü 37 BIOD-Adjustable Basal is a modified and possibly improved formulation of IGlar; with main advantage of its availability in long-, medium-, and short -acting forms, which could be mixed. BIOD-Smart Basal insulin explores a very different concept. Its formulation includes IGlar, glucose oxidase, and peroxidase at p. H*4. § Ce. Qur (Monreux, Switzerland) is developing a wearable insulin delivery device, a patch pump, that delivers basal insulin subcutaneously. § Eli Lilly is developing 2 new analogues, registered as LY 2605541 & LY 2963016. ü Only scarce data available on clinical trials so far. ü Both in phase 2 of development in 2011. ü LY 2605541 is a structurally novel basal insulin analog; with un-published molecular properties and its mode of protraction. ü LY 2963016 is a new IGlar product.

WHAT’S DEGLUDEC?

Insulin Degludec: a new generation, ultra-long acting basal insulin De glu dec Insulin backbone q Deletion of Thr. B 30 and the addition of a 16 -carbon fatty acid attached to Lys. B 29 via a glutamic acid spacer. Side chain: fatty acid + linker Allows formation of multi-hexamers Confers albumin binding
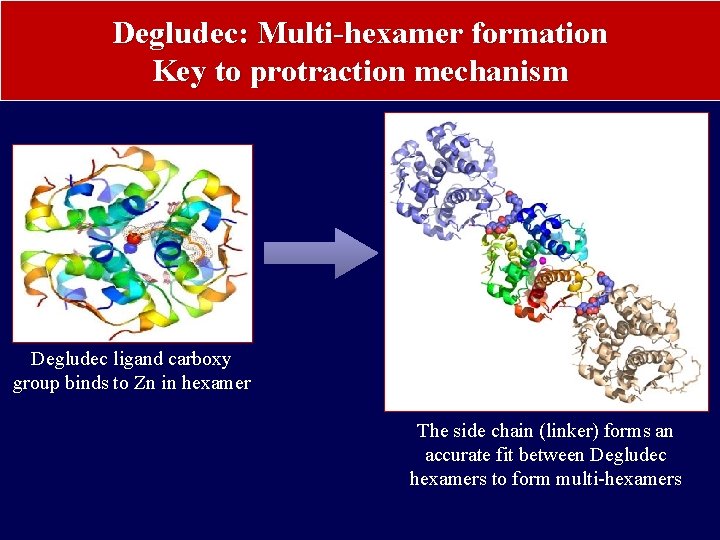
Degludec: Multi-hexamer formation Key to protraction mechanism Degludec ligand carboxy group binds to Zn in hexamer The side chain (linker) forms an accurate fit between Degludec hexamers to form multi-hexamers
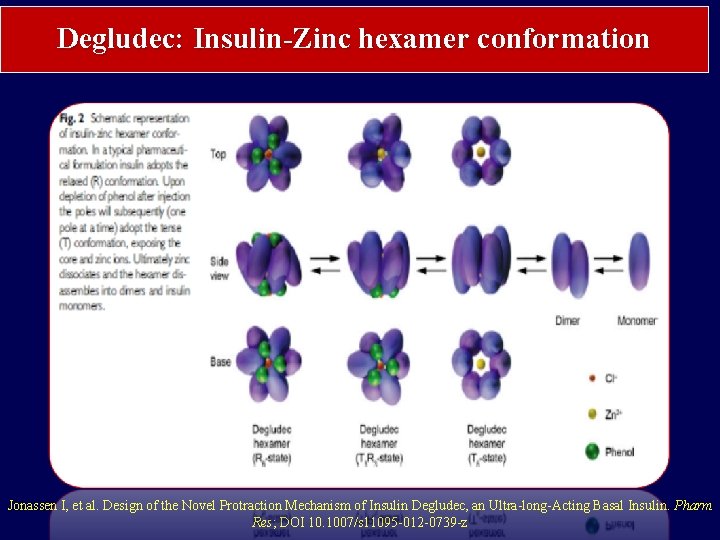
Degludec: Insulin-Zinc hexamer conformation Jonassen I, et al. Design of the Novel Protraction Mechanism of Insulin Degludec, an Ultra-long-Acting Basal Insulin. Pharm Res; DOI 10. 1007/s 11095 -012 -0739 -z
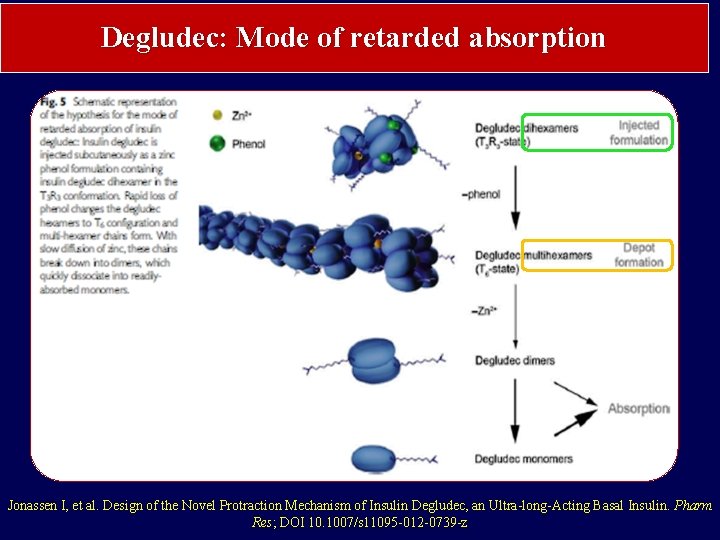
Degludec: Mode of retarded absorption Jonassen I, et al. Design of the Novel Protraction Mechanism of Insulin Degludec, an Ultra-long-Acting Basal Insulin. Pharm Res; DOI 10. 1007/s 11095 -012 -0739 -z
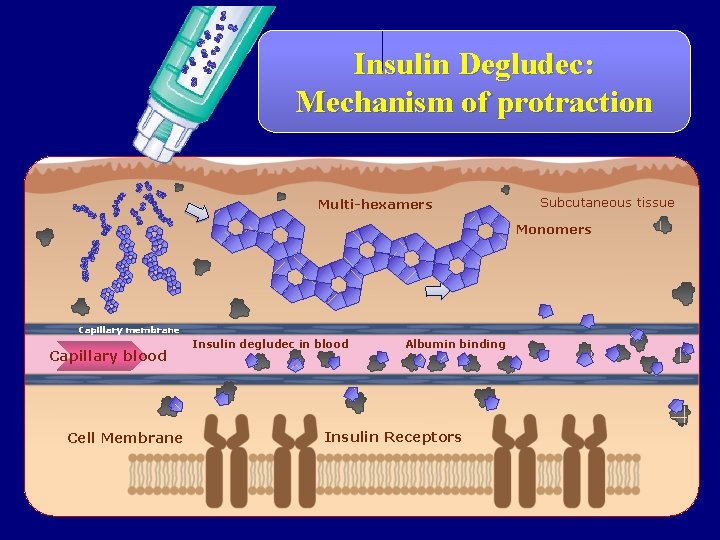
Insulin Degludec: Mechanism of protraction Multi-hexamers Subcutaneous tissue Monomers Capillary membrane Capillary blood Cell Membrane Insulin degludec in blood Albumin binding Insulin Receptors
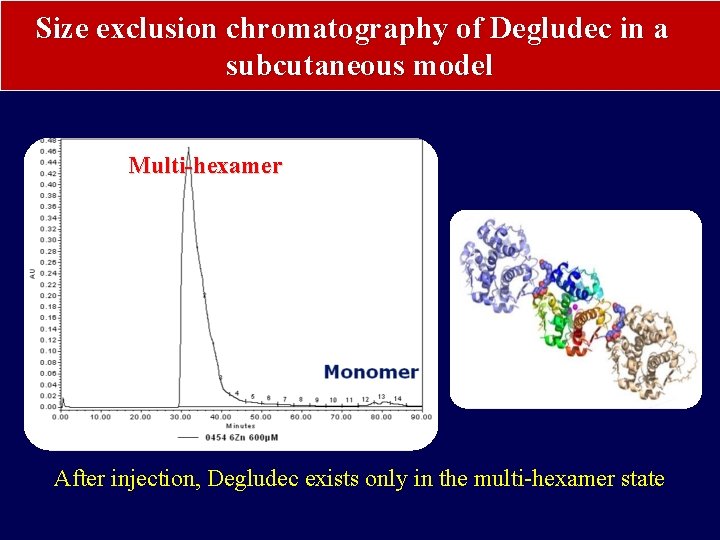
Size exclusion chromatography of Degludec in a subcutaneous model Multi-hexamer After injection, Degludec exists only in the multi-hexamer state

Degludec Half-life l A randomized, double-blind, two-period, crossover comparison of IDeg and IGlar in 66 patients with DM 1 reported: § A half-life of 25. 4 hrs with IDeg compared with 12. 5 hrs with IGlar. § Similar serum exposure of IDeg between the 1 st and 2 nd 12 -hr period postdose. § 60% of the serum exposure to IGlar occurred over the 1 st 12 -hrs postdose. l These results highlight that IDeg is an ultra–long-acting insulin preparation with improved pharmacodynamic stability Heise T, et al. IDeg has a two-fold longer half-life and a more consistent pharmacokinetic profile than IGlar. Paper presented at: ADA 71 st Scientific Sessions; June 24 -28, 2011; San Diego, CA.

Degludec Mitogenicity §Insulin receptor binding studies and in vitro studies indicated that IDeg has a low affinity for the human IGF-1 receptor, comparable with that of human insulin. §It has also a low mitogenic/metabolic potency ratio Nishimura E, et al. Insulin degludec: a new ultra-long, basal insulin designed to maintain full metabolic effect while minimizing mitogenic potential [abstract]. Diabetologia 2010; 53(Suppl 1): S 388–S 9.
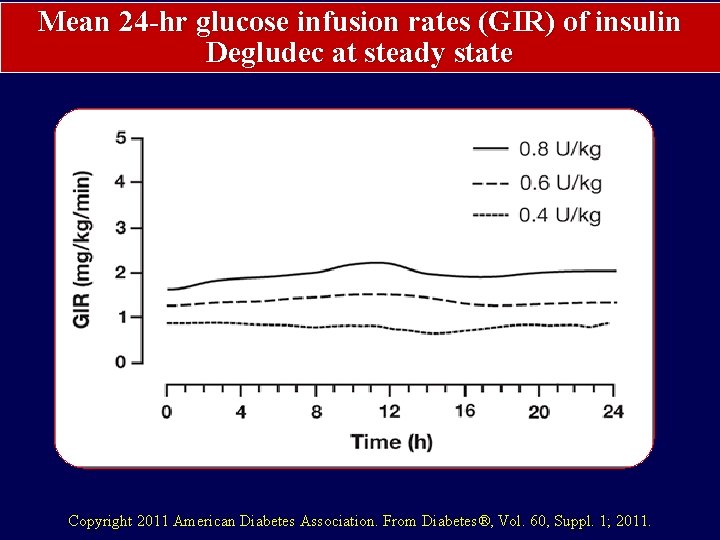
Mean 24 -hr glucose infusion rates (GIR) of insulin Degludec at steady state Copyright 2011 American Diabetes Association. From Diabetes®, Vol. 60, Suppl. 1; 2011.
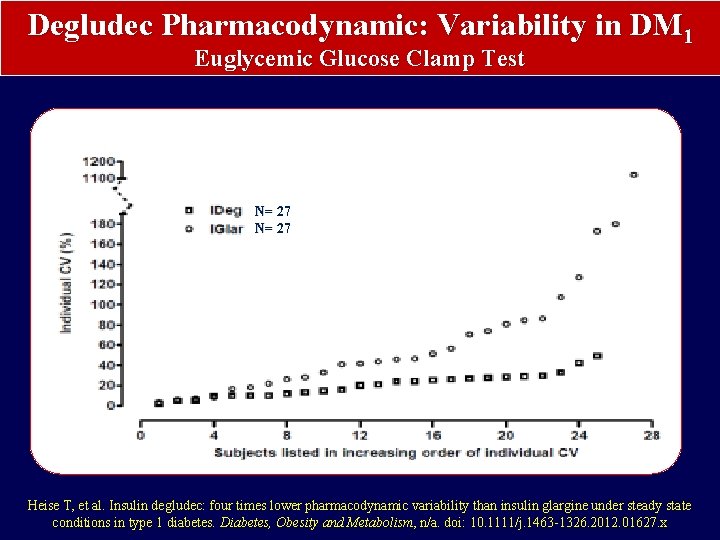
Degludec Pharmacodynamic: Variability in DM 1 Euglycemic Glucose Clamp Test N= 27 Heise T, et al. Insulin degludec: four times lower pharmacodynamic variability than insulin glargine under steady state conditions in type 1 diabetes. Diabetes, Obesity and Metabolism, n/a. doi: 10. 1111/j. 1463 -1326. 2012. 01627. x
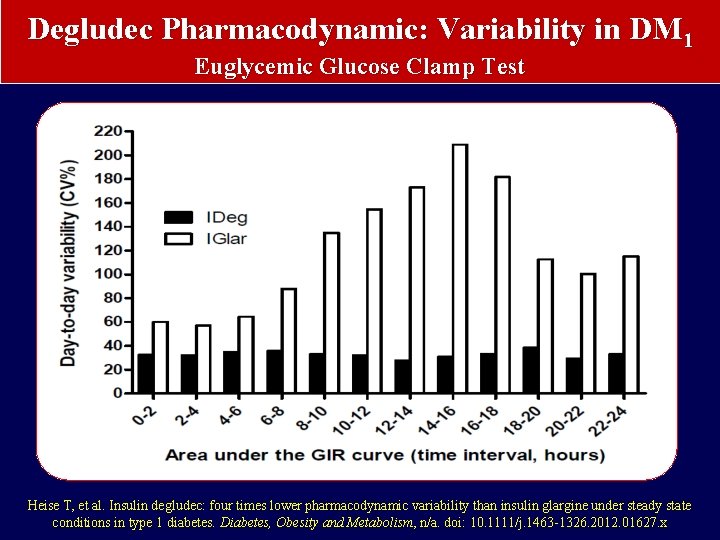
Degludec Pharmacodynamic: Variability in DM 1 Euglycemic Glucose Clamp Test Heise T, et al. Insulin degludec: four times lower pharmacodynamic variability than insulin glargine under steady state conditions in type 1 diabetes. Diabetes, Obesity and Metabolism, n/a. doi: 10. 1111/j. 1463 -1326. 2012. 01627. x
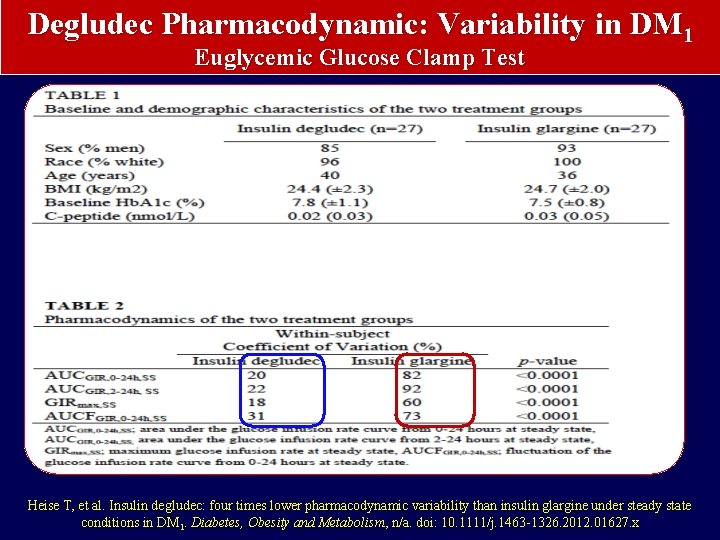
Degludec Pharmacodynamic: Variability in DM 1 Euglycemic Glucose Clamp Test Heise T, et al. Insulin degludec: four times lower pharmacodynamic variability than insulin glargine under steady state conditions in DM 1. Diabetes, Obesity and Metabolism, n/a. doi: 10. 1111/j. 1463 -1326. 2012. 01627. x
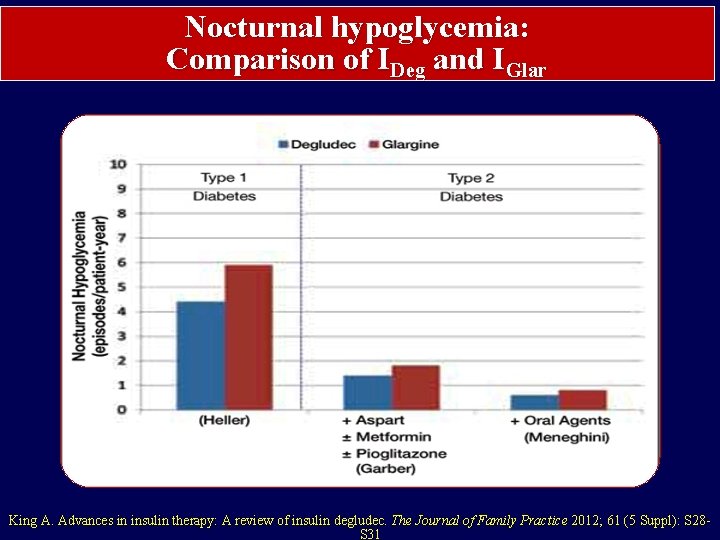
Nocturnal hypoglycemia: Comparison of IDeg and IGlar King A. Advances in insulin therapy: A review of insulin degludec. The Journal of Family Practice 2012; 61 (5 Suppl): S 28 S 31

DEGLUDEC CLINICAL EVIDENCES

§OBJECTIVE To assess the efficacy and safety of IDeg formulations administered once daily in combination with mealtime IAsp in people with DM 1 §AIM o. Not to determine superiority or noninferiority of IDeg o. Rather to estimate a treatment difference (in A 1 C) with a sufficient precision §No formal statistical testing, no P values Birkeland K, et al. Insulin Degludec in Type 1 Diabetes Care 2011; 34: 661 -5

IDeg in DM 1: A RCT of a new-generation ultra-long-acting insulin compared with IGlar RESEARCH DESIGN AND METHODS § 16 -week, randomized, open-label trial §Participants 178 cases 28 centers, 5 countries: Australia, Germany, Norway, Sweden, and the USA Mean age: 45. 8 yrs A 1 C: 8. 4% FPG: 178 mg/d. L BMI: 26. 9 kg/m 2 §Treatment groups: once daily in the evening of either IDeg(A) (600 μmol/L; n = 59) IDeg(B) (900 μmol/L; n = 60) Glargine (IGlar; n = 59) plus Insulin aspart at mealtimes Birkeland K, et al. Insulin Degludec in Type 1 Diabetes Care 2011; 34: 661 -5
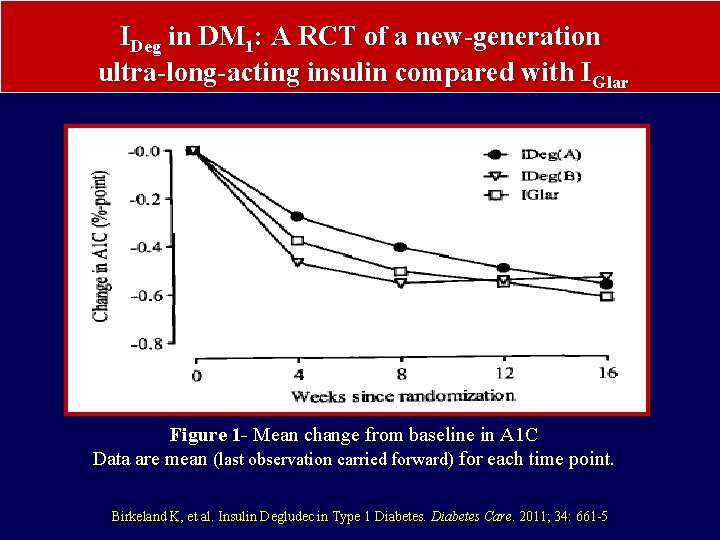
IDeg in DM 1: A RCT of a new-generation ultra-long-acting insulin compared with IGlar Figure 1 - Mean change from baseline in A 1 C Data are mean (last observation carried forward) for each time point. Birkeland K, et al. Insulin Degludec in Type 1 Diabetes Care. 2011; 34: 661 -5
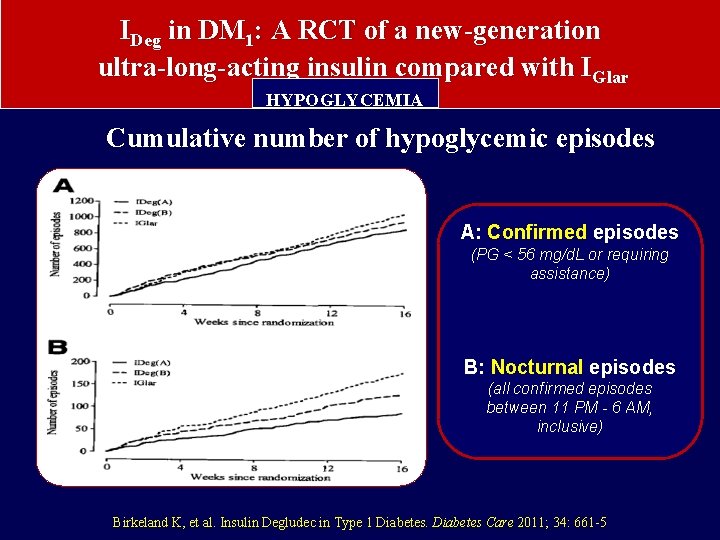
IDeg in DM 1: A RCT of a new-generation ultra-long-acting insulin compared with IGlar HYPOGLYCEMIA Cumulative number of hypoglycemic episodes A: Confirmed episodes (PG < 56 mg/d. L or requiring assistance) B: Nocturnal episodes (all confirmed episodes between 11 PM - 6 AM, inclusive) Birkeland K, et al. Insulin Degludec in Type 1 Diabetes Care 2011; 34: 661 -5

IDeg in DM 1: A RCT of a new-generation ultra-long-acting insulin compared with IGlar CONCLUSIONS In this clinical exploratory phase 2 trial in people with DM 1, IDeg (used in combination with mealtime IAsp) is safe and well tolerated and provides comparable glycemic control to IGlar at similar doses, with reduced rates of hypoglycemia. Birkeland K, et al. Insulin Degludec in Type 1 Diabetes Care 2011; 34: 661 -5

OBJECTIVE l IDeg. Asp (Degludec Plus) 30% l is a soluble coformulation of IDeg 70% and IAsp We compared the safety and efficacy of IDeg. Asp , an alternative formulation (AF) (55% IDeg and 45% IAsp), and IGlar in insulin-naïve subjects with DM 2 inadequately controlled with OHAs. Heise T, et al. Diabetes Care 2011; 34: 669 -74.

IDeg With a Bolus Boost Compared With IGlar in Insulin-Naïve People With DM 2, An RCT RESEARCH DESIGN AND METHODS §Phase 2, open-label, randomized, controlled 16 -week trial. §Participants 178 insulin-naïve subjects with DM 2 inadequately controlled with OADs 22 sites in 5 European countries (France, Germany, Norway, Romania, and Spain) Mean age: 59. 1 years A 1 C 8. 5% BMI 30. 3 kg/m 2 §Statistical analyses ITT; for missing values: last observation carried forward. §Treatment groups: Insulin before the evening meal, titrated to a FPG target of 72– 108 mg/d. L: IDeg. Asp (n = 59) AF (n = 59) IGlar (n = 60) all in combination with metformin Heise T, et al. Diabetes Care. 2011; 34: 669 -74.
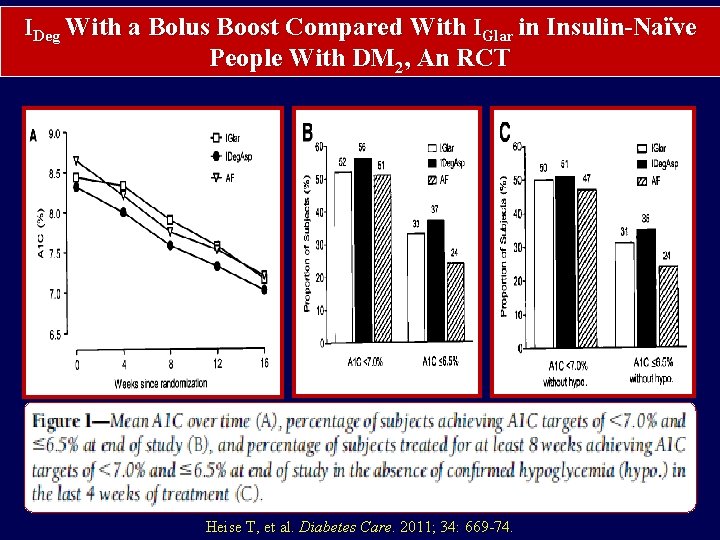
IDeg With a Bolus Boost Compared With IGlar in Insulin-Naïve People With DM 2, An RCT Heise T, et al. Diabetes Care. 2011; 34: 669 -74.
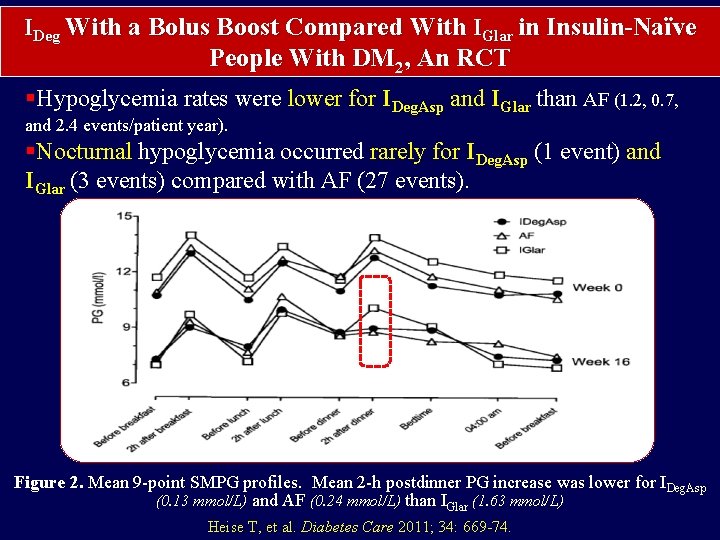
IDeg With a Bolus Boost Compared With IGlar in Insulin-Naïve People With DM 2, An RCT §Hypoglycemia rates were lower for IDeg. Asp and IGlar than AF (1. 2, 0. 7, and 2. 4 events/patient year). §Nocturnal hypoglycemia occurred rarely for IDeg. Asp (1 event) and IGlar (3 events) compared with AF (27 events). Figure 2. Mean 9 -point SMPG profiles. Mean 2 -h postdinner PG increase was lower for IDeg. Asp (0. 13 mmol/L) and AF (0. 24 mmol/L) than IGlar (1. 63 mmol/L) Heise T, et al. Diabetes Care 2011; 34: 669 -74.

IDeg With a Bolus Boost Compared With IGlar in Insulin-Naïve People With DM 2, An RCT CONCLUSIONS l In this proof-of-concept trial, once-daily IDeg. Asp was safe, well tolerated, and provided comparable overall glycemic control to IGlar at similar low rates of hypoglycemia, hypoglycemia but better post-dinner plasma glucose control. Heise T, et al. Diabetes Care. 2011; 34: 669 -74

www. thelancet. com Vol 377 March 12, 2011 Aim: to assess efficacy and safety of IDeg injected once a day or 3 times a week compared with IGlar once daily in insulin-naive people with DM 2, who were inadequately controlled with OADs. Zinman B, et al. Lancet 2011; 377: 924– 31

IDeg once a day or 3 times a week vs. IGlar once a day in patients with DM 2: a 16 -week, randomised, open-label, phase 2 trial RESEARCH DESIGN AND METHODS § 16 -week, randomised, open-label, parallel-group phase 2 trial §Participants 245 insulin-naïve subjects with DM 2 inadequately controlled with OADs 28 sites in 4 countries (Canada, India, South Africa, and USA) aged 18– 75 years Hb. A 1 C: 7– 11% BMI: 23– 42 kg/m 2 §Treatment group: Insulin was dose-titrated to a FPG target of 72– 108 mg/d. L. IDeg(n=62) 3 times a week, starting dose 20 U [1 U=9 nmol] IDeg(n=60) once a day, starting dose 10 U [1 U=6 nmol]; group A IDeg(n=61) once a day, starting dose 10 U [1 U=9 nmol]; group B IGlar(n=62) once a day, starting dose 10 U [1 U=6 nmol] once a day all in combination with metformin Zinman B, et al. Lancet 2011; 377: 924– 31.
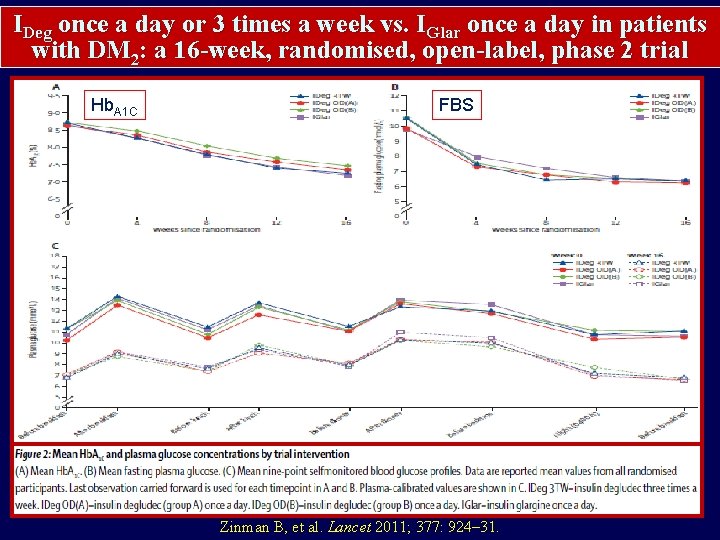
IDeg once a day or 3 times a week vs. IGlar once a day in patients with DM 2: a 16 -week, randomised, open-label, phase 2 trial Hb. A 1 C FBS Zinman B, Fulcher G, Rao P, et al. Lancet 2011; 377: 924– 31. Zinman B, et al. Lancet 2011; 377: 924– 31.

IDeg once a day or 3 times a week vs. IGlar once a day in patients with DM 2: a 16 -week, randomised, open-label, phase 2 trial HYPOGLYCEMIA l Overall reported rates of hypoglycemia were low in all treatment groups § 77– 92% of participants did not have a hypoglycemic episode. § Rates of hypoglycemia did not differ between groups. § The proportion of participants who had hypoglycemia in IDeg group A was lower than was the proportion in the IGlar group and the IDeg 3 times a week group (odds ratio 0. 26 and 0. 31, 0. 31 respectively). § The rate of confirmed nocturnal hypoglycemia was low in all treatment Zinman B, et al. Lancet 2011; 377: 924– 31.

IDeg once a day or 3 times a week vs. IGlar once a day in patients with DM 2: a 16 -week, randomised, open-label, phase 2 trial This proof-of-concept trial has several limitations inherent to phase 2 regulatory studies. Mean Hb. A 1 C lowering with the addition of basal insulin alone to OADs of 1. 5% is greater than we commonly observe in routine clinical practice, and is probably related to the Hawthorne effect of increased self-awareness, increased surveillance, and the frequent visits demanded by trials. The open-label design that was used to accommodate the different insulin-injection systems could have affected efforts to attain blood glucose control and reporting of hypoglycemia and adverse events. Zinman B, et al. Lancet 2011; 377: 924– 31.

IDeg once a day or 3 times a week vs. IGlar once a day in patients with DM 2: a 16 -week, randomised, open-label, phase 2 trial CONCLUSION § IDeg once daily or 3 times weekly as add-on to Metformin, provides comparable glycaemic control to IGlar without additional adverse events and might reduce dosing frequency due to its ultra-long action profile. § The safety, efficacy, and optimum use of treatment regimens for IDeg need to be established. Zinman B, et al. Lancet 2011; 377: 924– 31.

www. thelancet. com Vol 379 April 21, 2012 Aim: to assess the efficacy and safety of IDeg compared with IGlar in a basal-bolus regimen in patients with longstanding DM 2. Garber AJ, et al. Lancet 2012; 379: 1498– 507
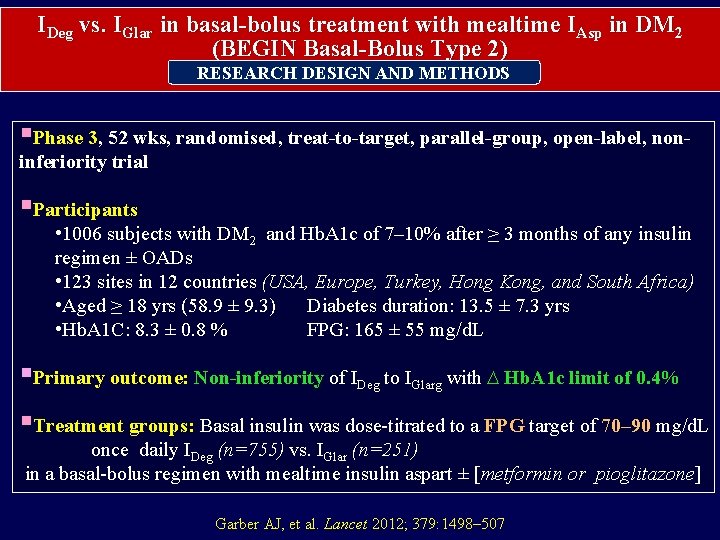
IDeg vs. IGlar in basal-bolus treatment with mealtime IAsp in DM 2 (BEGIN Basal-Bolus Type 2) RESEARCH DESIGN AND METHODS §Phase 3, 52 wks, randomised, treat-to-target, parallel-group, open-label, noninferiority trial §Participants • 1006 subjects with DM 2 and Hb. A 1 c of 7– 10% after ≥ 3 months of any insulin regimen ± OADs • 123 sites in 12 countries (USA, Europe, Turkey, Hong Kong, and South Africa) • Aged ≥ 18 yrs (58. 9 ± 9. 3) Diabetes duration: 13. 5 ± 7. 3 yrs • Hb. A 1 C: 8. 3 ± 0. 8 % FPG: 165 ± 55 mg/d. L §Primary outcome: Non-inferiority of IDeg to IGlarg with ∆ Hb. A 1 c limit of 0. 4% §Treatment groups: Basal insulin was dose-titrated to a FPG target of 70– 90 mg/d. L once daily IDeg (n=755) vs. IGlar (n=251) in a basal-bolus regimen with mealtime insulin aspart ± [metformin or pioglitazone] Garber AJ, et al. Lancet 2012; 379: 1498– 507
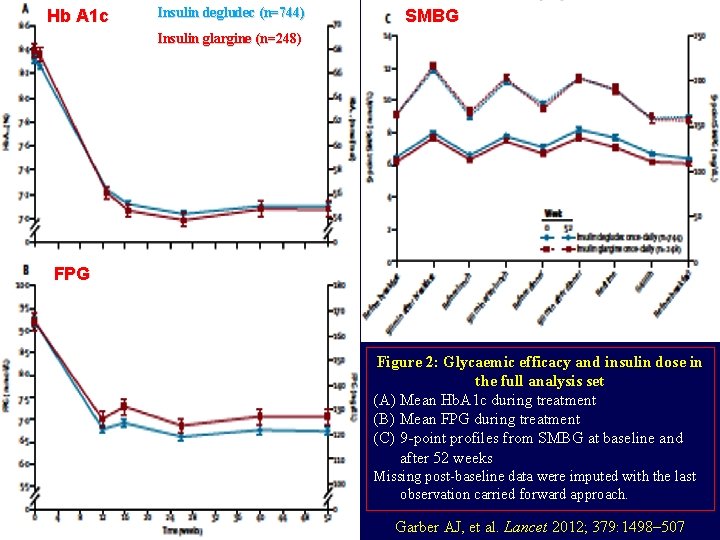
Hb A 1 c Insulin degludec (n=744) SMBG Insulin glargine (n=248) FPG Figure 2: Glycaemic efficacy and insulin dose in the full analysis set (A) Mean Hb. A 1 c during treatment (B) Mean FPG during treatment (C) 9 -point profiles from SMBG at baseline and after 52 weeks Missing post-baseline data were imputed with the last observation carried forward approach. Garber AJ, et al. Lancet 2012; 379: 1498– 507
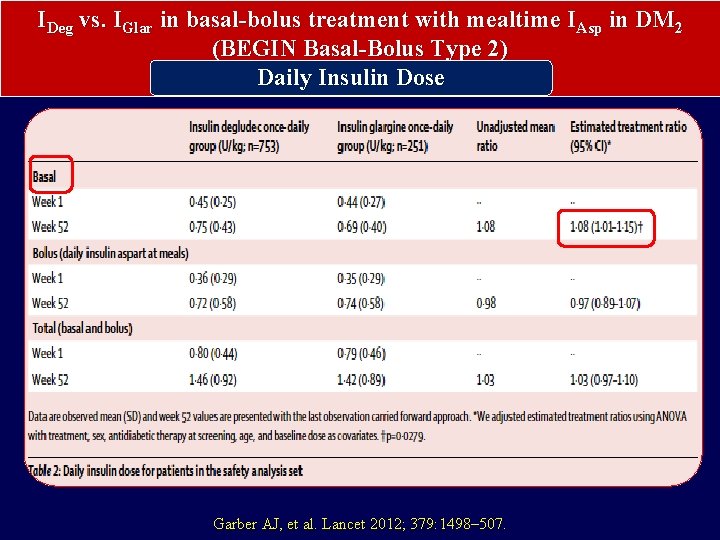
IDeg vs. IGlar in basal-bolus treatment with mealtime IAsp in DM 2 (BEGIN Basal-Bolus Type 2) Daily Insulin Dose Garber AJ, et al. Lancet 2012; 379: 1498– 507.
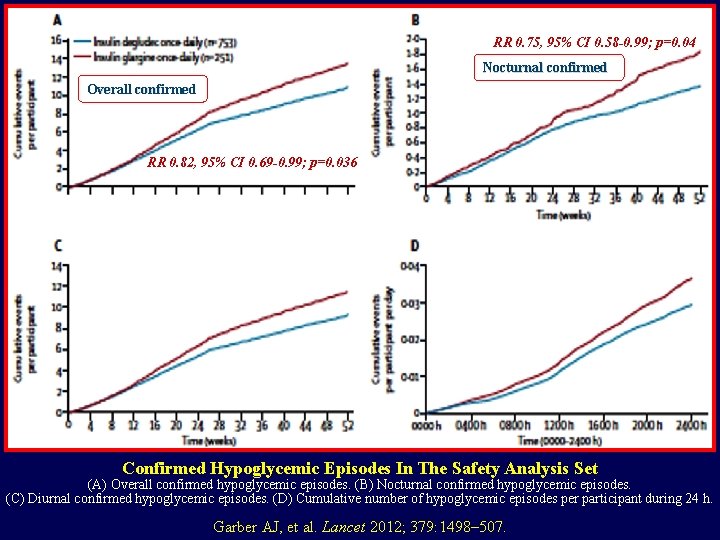
RR 0. 75, 95% CI 0. 58 -0. 99; p=0. 04 Nocturnal confirmed Overall confirmed RR 0. 82, 95% CI 0. 69 -0. 99; p=0. 036 Confirmed Hypoglycemic Episodes In The Safety Analysis Set (A) Overall confirmed hypoglycemic episodes. (B) Nocturnal confirmed hypoglycemic episodes. (C) Diurnal confirmed hypoglycemic episodes. (D) Cumulative number of hypoglycemic episodes per participant during 24 h. Garber AJ, et al. Lancet 2012; 379: 1498– 507.

IDeg vs. IGlar in basal-bolus treatment with mealtime IAsp in DM 2 (BEGIN Basal-Bolus Type 2) Interpretation l. A policy of suboptimum diabetes control to reduce the risk of hypoglycemia and its consequences in advanced DM 2 might be unwarranted with newer basal insulins such as IDeg , which are associated with lower risks of hypoglycemia than IGlar. l The significantly lower rate of nocturnal hypoglycemia with IDeg while achieving similar glycaemic control, might offer a safer option in patients with longstanding DM 2 who need basalbolus insulin therapy. Garber AJ, et al. Lancet 2012; 379: 1498– 507.

www. thelancet. com Vol 379 April 21, 2012 Aim: to compare the efficacy and safety of once-daily usage of IDeg with IGlar in a basal-bolus regimen in patients with longstanding DM 1 Heller S, et al. Lancet 2012; 379: 1489– 97

IDeg vs. IGlar in basal-bolus treatment with mealtime IAsp in DM 1 (BEGIN Basal-Bolus Type 1) RESEARCH DESIGN AND METHODS §Phase 3, 52 wks, randomised, controlled, treat-to-target, parallel design, openlabel, non-inferiority trial §Participants • 629 subjects with DM 1 > 1 year duration, BMI ≤ 35, and Hb. A 1 c of ≤ 10% who had received any basal-bolus insulin therapy for at least 1 year before screening. • 79 sites in 6 countries (USA, UK, France, Germany, , Russia, and South Africa) • Aged ≥ 18 yrs (42. 5 ± 13. 6) Diabetes duration: 18. 8 yrs • Hb. A 1 C: 7. 7 ± 0. 9 % FPG: 165 mg/d. L §Primary outcome: Non-inferiority of IDeg to IGlarg with ∆ Hb. A 1 c limit of 0. 4% §Treatment groups: Basal insulin was dose-titrated to a FPG target of 70– 90 mg/d. L once daily IDeg (n=472) vs. IGlar (n=157) in a basal-bolus regimen with mealtime insulin aspart Heller S, et al. Lancet 2012; 379: 1489– 97
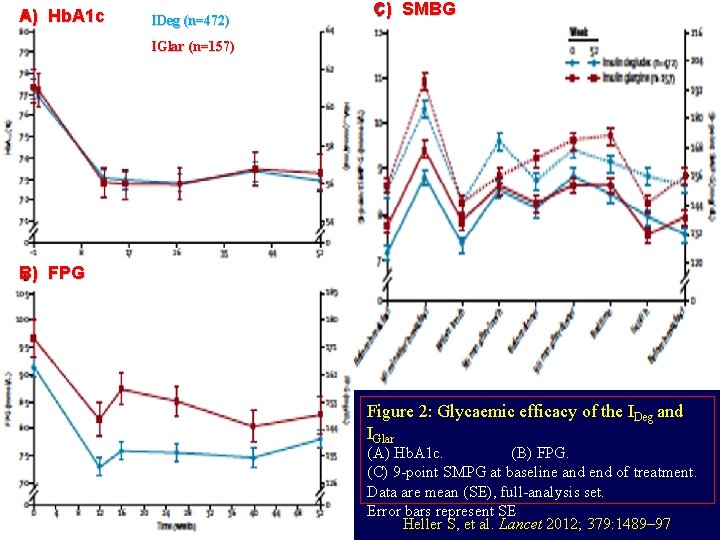
A) Hb. A 1 c IDeg (n=472) C) SMBG IGlar (n=157) B) FPG Figure 2: Glycaemic efficacy of the IDeg and IGlar (A) Hb. A 1 c. (B) FPG. (C) 9 -point SMPG at baseline and end of treatment. Data are mean (SE), full-analysis set. Error bars represent SE Heller S, et al. Lancet 2012; 379: 1489– 97
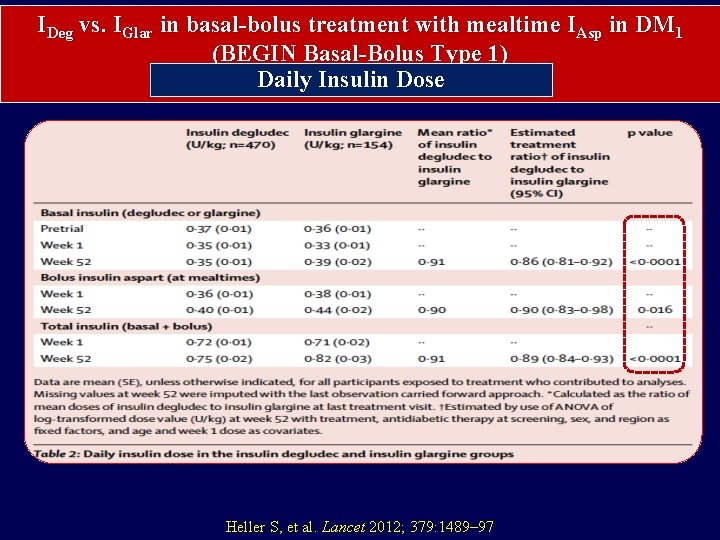
IDeg vs. IGlar in basal-bolus treatment with mealtime IAsp in DM 1 (BEGIN Basal-Bolus Type 1) Daily Insulin Dose Heller S, et al. Lancet 2012; 379: 1489– 97
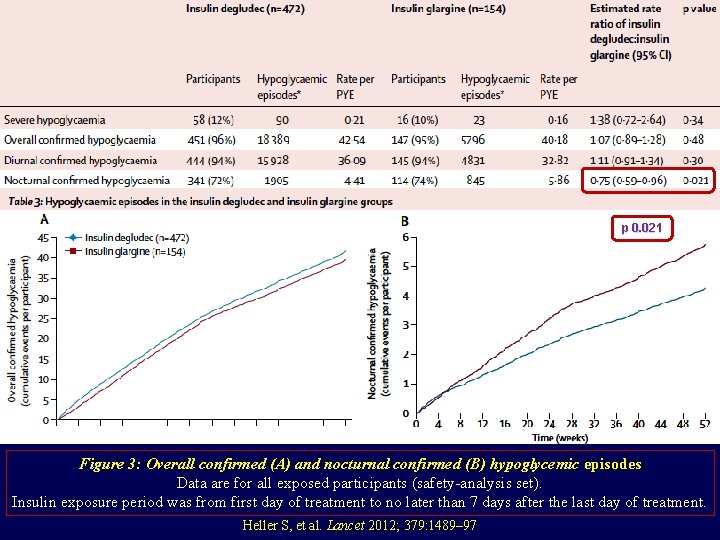
p 0. 021 Figure 3: Overall confirmed (A) and nocturnal confirmed (B) hypoglycemic episodes Data are for all exposed participants (safety-analysis set). Insulin exposure period was from first day of treatment to no later than 7 days after the last day of treatment. Heller S, et al. Lancet 2012; 379: 1489– 97

IDeg vs. IGlar in basal-bolus treatment with mealtime IAsp in DM 1 (BEGIN Basal-Bolus Type 1) Interpretation § IDeg provides similar glycaemic control to IGlar with a lower rate of nocturnal hypoglycemia, hypoglycemia which is a major limitation of insulin therapy. § There might be a potential role, as a useful basal insulin, for IDeg in helping patients with DM 1 to reach and maintain tight glucose targets Heller S, et al. Lancet 2012; 379: 1489– 97

Degludec: Experience in High-Doses l In a meta-analysis of 5, phase 3, open-label, randomised, 26 or 52 -week trials in patients with DM 2 comparing once-daily IDeg (n=2, 262) and IGlar (n=1, 110), 35% of patients required > 60 U of IDeg , and 33. 7% of cases >60 U of IGlar. Comparing these two subgroups: § Hb. A 1 C levels were similar at the end of the studies (P =0. 44) § FPG was lower (5. 9 mg/d. L) with IDeg than IGlar (P <0. 04) § Overall confirmed and nocturnal hypoglycaemia rates were lower in patients on IDeg than on IGlar. • Overall confirmed hypoglycemia- RR: IDeg/IGlar 0. 79, P =. 02 • Nocturnal confirmed hypoglycemia- RR: IDeg/IGlar 0. 48, P <. 0001 l IDeg is a safe and effective basal-insulin choice for patients with DM 2 across the spectrum of insulin requirements. Rodbard H, et al. Reduced risk of hypoglycemia with IDeg vs IGlar in patients with DM 2 requiring high doses of basal insulin: meta-analysis of 5 randomized trials. Abstract 241, presented at the 21 st annual meeting and clinical congress of AACE, May 2012.

q A 16 -week, multinational, open-label, randomized trial. q Methods: Once-daily IDeg (n=59) or IGlar (n=59), with IAsp Health status assessment utilized the validated SF-36. at meals. q Results: Degludec resulted in: § Comparable glycaemic control § Less confirmed nocturnal hypoglycemia [RR: 0. 42 (95% CI 0. 25– 0. 69)] § Less overall hypoglycemia [RR: 0. 72 (95% CI 0. 52– 1. 00)] § A significant improvement in SF-36 mental component score; indicating a small-to-medium clinically meaningful difference, attributable to significant changes in the social functioning and mental health domains q Conclusions: Considering the comparable overall glycaemic control with IGlar , IDeg improved mental well-being as measured using the mental component score of the SF-36. It may relate to the observed reduction in hypoglycemic events. Home PD, et al. Improved health status with IDeg compared with IGlar in people with T 1 D. Diabet Med. 2012; 29: 716– 720

Which Properties of Currently Available Long-Acting Analogues Could Be Improved? § § § § Predictable flat or near-flat time–action profile Low or no variability Longer duration of action Soluble to allow mixing with rapid-acting analog Low costs Less or no weight gain No signal of increased mitogenicity Simon ACR, et al. The Future of Basal Insulin Supplementation. Diabetes Technology & Therapeutics 2011; 13, Suppl. 1, S 103 -8

CONCLUSION l Degludec is not a revolution but an evolution of insulin therapy. l IDeg is a safe and effective basal-insulin for patients with type 1 and type 2 diabetes. l Lower rate of important hypoglycemic events is one of the most valuable points that should be considered in its use. l If accessible at a reasonable price after getting official approvals, it will replace for Glargine in the near coming years.

Thanks for your patience

Attributes Of The Ideal Basal Insulin § Delivers a steady, stable, peak less, continuous insulin concentration for ≥ 24 hrs, in a predictable manner, with low intraindividual and interindividual variability § Does not cause side effects such as weight gain or hypoglycemia § Does not induce mitogenicity § Can be used as monotherapy, as part of basal-bolus therapy, or in combination with OADs § Equally efficacious, safe, and well-tolerated in patients with DM 1 or DM 2 Meneghini L, et al. Insulin Detemir: A historical perspective on a modern basal insulin analogue. Prim Care Diabetes. 2010; 4(Suppl 1): S 31– 42.
Confirmed hypoglycemic episodes in the safety analysis set (E) Distribution of confirmed hypoglycemic episodes. Garber AJ, et al. Lancet 2012; 379: 1498– 507.
- Slides: 57