Ultrahighresolution Laser Spectroscopy and The Zeeman Effect of






- Slides: 32

Ultrahigh-resolution Laser Spectroscopy and The Zeeman Effect of Naphthalene S 1←S 0 Transition c (z) Kazuto Yoshida, Shunji Kasahara Kobe University, Japan b (y) a (x) Masaaki Baba Kyoto University, Japan

Introduction PAHs (Polycyclic Aromatic Hydrocarbons) benzene naphthalene anthracene TG 10 TG 11 Ultrahigh-resolution laser spectroscopy Molecular Constants Molecular Structure Linewidth Energy Shift Excited-State Dynamics Zeeman Effect

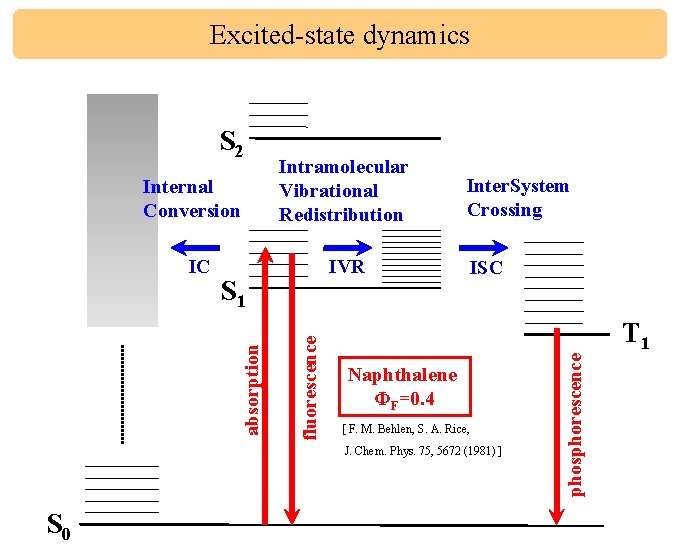
Excited-state dynamics Internal Conversion IVR fluorescence S 1 absorption IC Intramolecular Vibrational Redistribution Inter. System Crossing ISC Naphthalene ΦF=0. 4 [ F. M. Behlen, S. A. Rice, J. Chem. Phys. 75, 5672 (1981) ] S 0 phosphorescence S 2 T 1

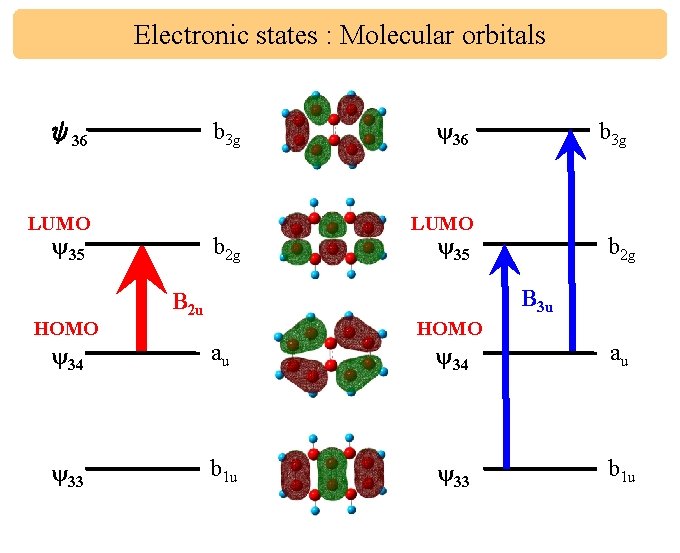
Electronic states : Molecular orbitals b 3 g ψ36 LUMO ψ35 HOMO b 2 g ψ36 b 3 g LUMO ψ35 b 2 g B 3 u B 2 u ψ34 au ψ33 b 1 u HOMO ψ34 au ψ33 b 1 u

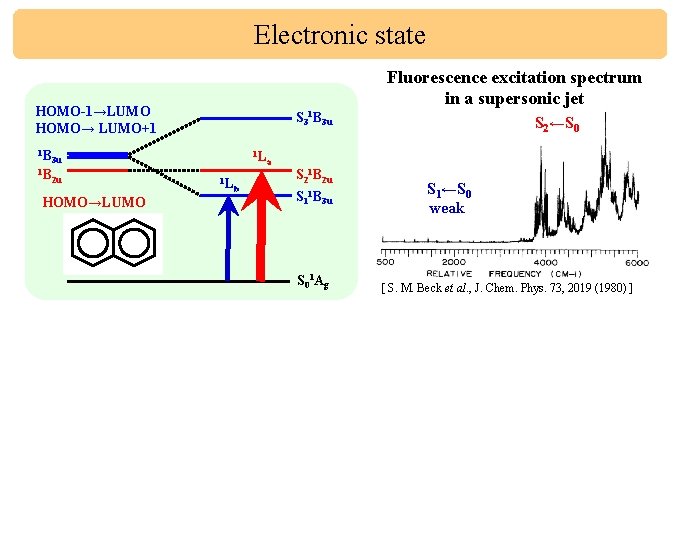
Electronic state Fluorescence excitation spectrum in a supersonic jet HOMO-1→LUMO HOMO→ LUMO+1 1 B 3 u 1 B 2 u S 31 B 3 u 1 L 1 L a b S 21 B 2 u S 11 B 3 u HOMO→LUMO S 0 1 Ag HOMO-1→LUMO HOMO→ LUMO+1 1 B 3 u 1 B S 3 1 L b 1 L 2 u a 1 B S 2←S 0 S 1←S 0 weak [ S. M. Beck et al. , J. Chem. Phys. 73, 2019 (1980) ] S 1←S 0 TG 11 3 u S 21 B 3 u S 11 B 2 u HOMO→LUMO S 0 1 Ag S 2←S 0 weak (NOT FOUND) RELATIVE FREQUENCY (CM-1)

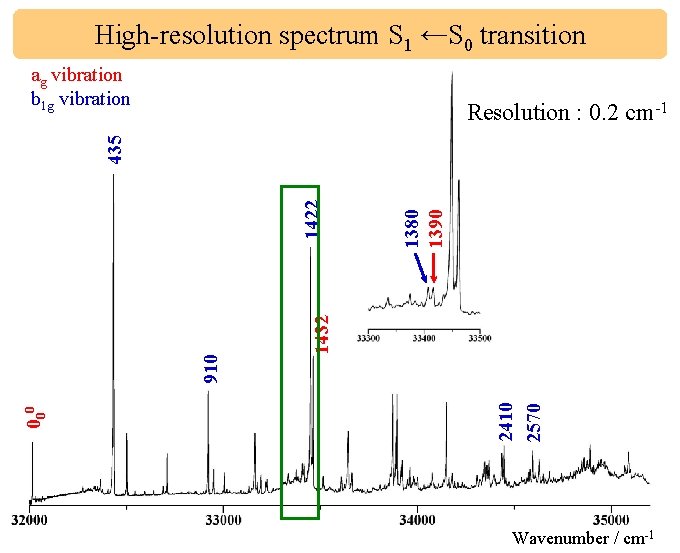
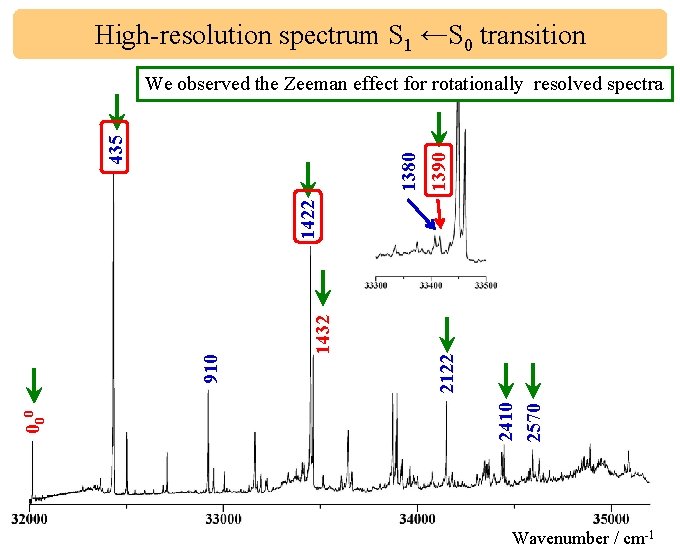
High-resolution spectrum S 1 ←S 0 transition ag vibration b 1 g vibration 2122 1432 1380 1390 2410 2570 000 910 1422 435 Resolution : 0. 2 cm-1 Wavenumber / cm-1

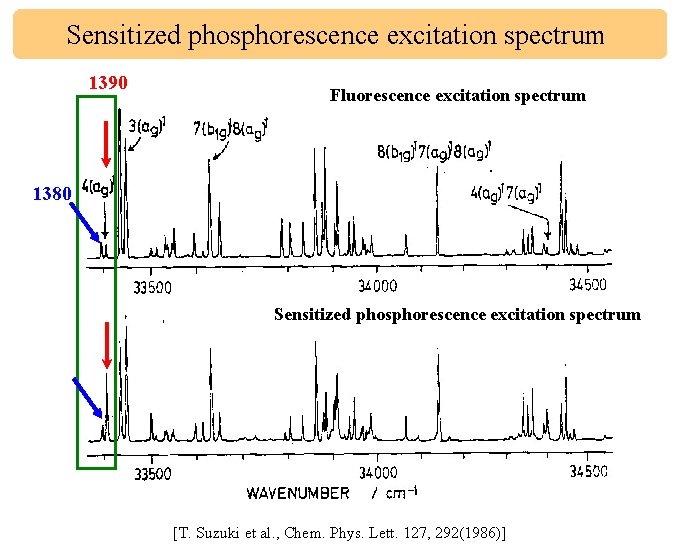
Sensitized phosphorescence excitation spectrum 1390 Fluorescence excitation spectrum 1380 Sensitized phosphorescence excitation spectrum [T. Suzuki et al. , Chem. Phys. Lett. 127, 292(1986)]

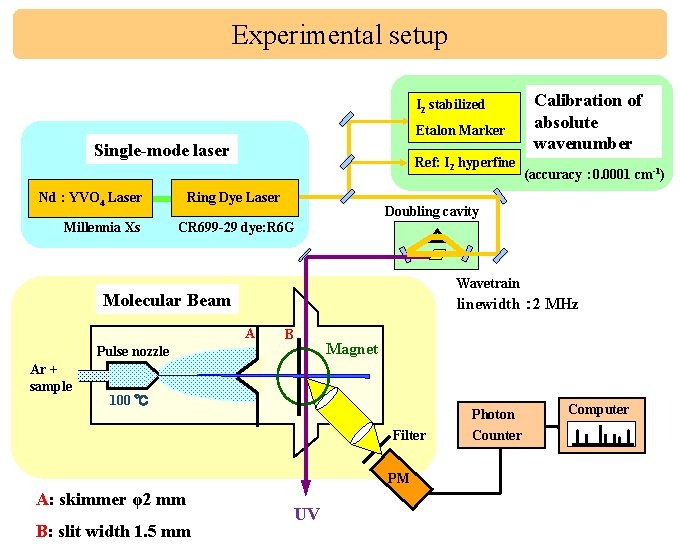
Experimental setup I 2 stabilized Etalon Marker Single-mode laser Ring Dye Laser Nd : YVO 4 Laser Millennia Xs Ref: I 2 hyperfine (accuracy : 0. 0001 cm-1) Doubling cavity CR 699 -29 dye: R 6 G Wavetrain Molecular Beam linewidth : 2 MHz A B Pulse nozzle Ar + sample Calibration of absolute wavenumber Magnet 100 ℃ Photon Filter PM A: skimmer φ2 mm B: slit width 1. 5 mm UV Counter Computer

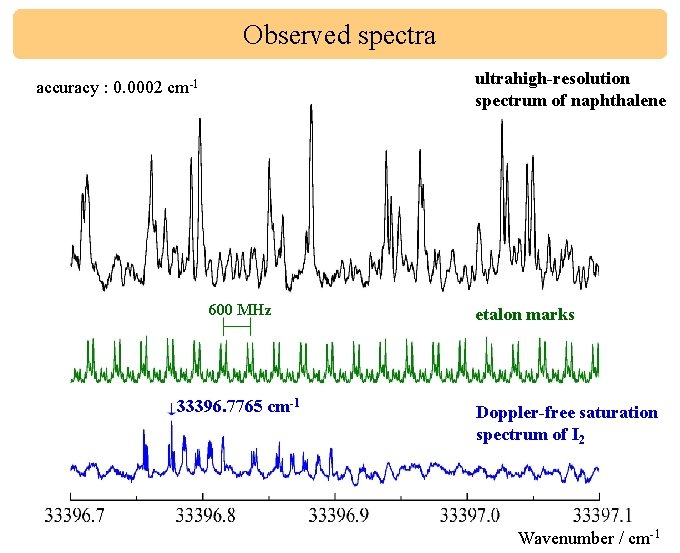
Observed spectra ultrahigh-resolution spectrum of naphthalene accuracy : 0. 0002 cm-1 600 MHz 33396. 7765 cm-1 etalon marks Doppler-free saturation spectrum of I 2 Wavenumber / cm-1

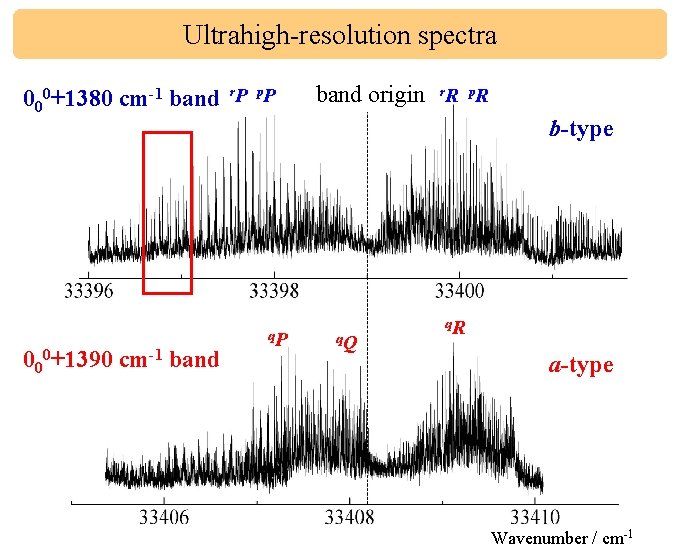
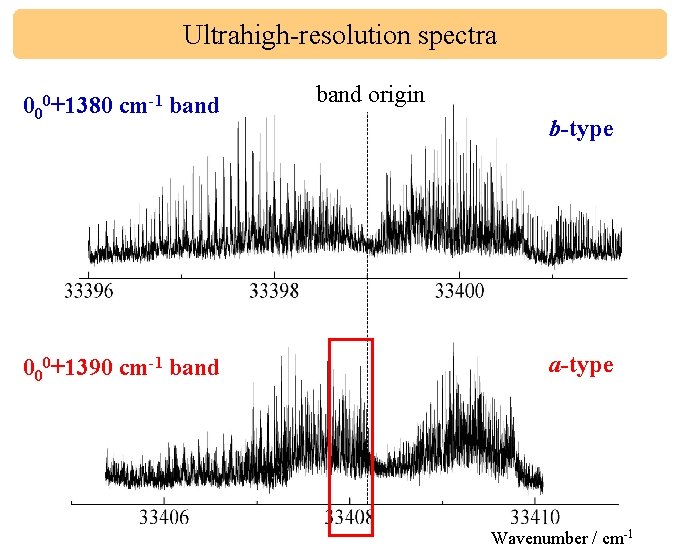
Ultrahigh-resolution spectra 000+1380 cm-1 band r. P p. P 000+1390 cm-1 band q. P band origin r. R p. R b-type q. Q q. R a-type Wavenumber / cm-1

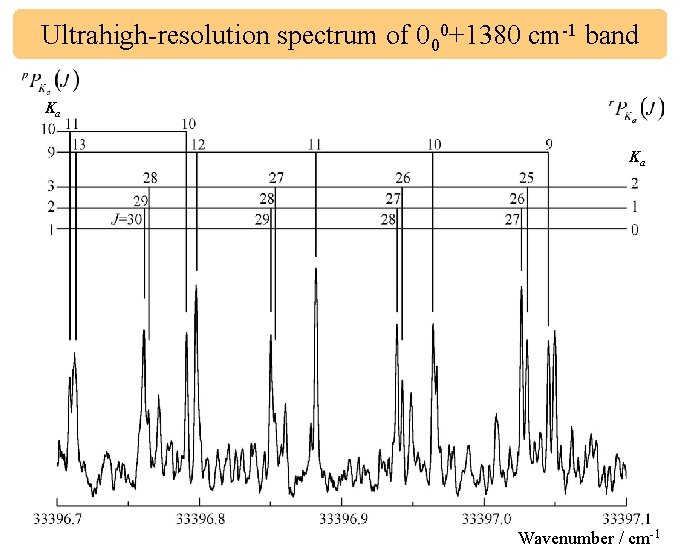
Ultrahigh-resolution spectrum of 000+1380 cm-1 band Ka Ka Wavenumber / cm-1

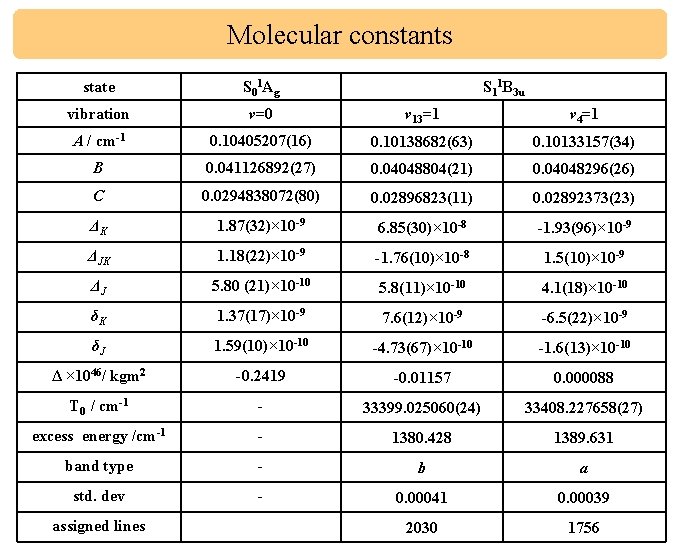
Molecular constants state S 01 Ag vibration ν=0 ν 13=1 ν 4=1 A / cm-1 0. 10405207(16) 0. 10138682(63) 0. 10133157(34) B 0. 041126892(27) 0. 04048804(21) 0. 04048296(26) C 0. 0294838072(80) 0. 02896823(11) 0. 02892373(23) ΔK 1. 87(32)× 10 -9 6. 85(30)× 10 -8 -1. 93(96)× 10 -9 ΔJK 1. 18(22)× 10 -9 -1. 76(10)× 10 -8 1. 5(10)× 10 -9 ΔJ 5. 80 (21)× 10 -10 5. 8(11)× 10 -10 4. 1(18)× 10 -10 δK 1. 37(17)× 10 -9 7. 6(12)× 10 -9 -6. 5(22)× 10 -9 δJ 1. 59(10)× 10 -10 -4. 73(67)× 10 -10 -1. 6(13)× 10 -10 Δ × 1046/ kgm 2 -0. 2419 -0. 01157 0. 000088 T 0 / cm-1 - 33399. 025060(24) 33408. 227658(27) excess energy /cm-1 - 1380. 428 1389. 631 band type - b a std. dev - 0. 00041 0. 00039 2030 1756 assigned lines S 11 B 3 u

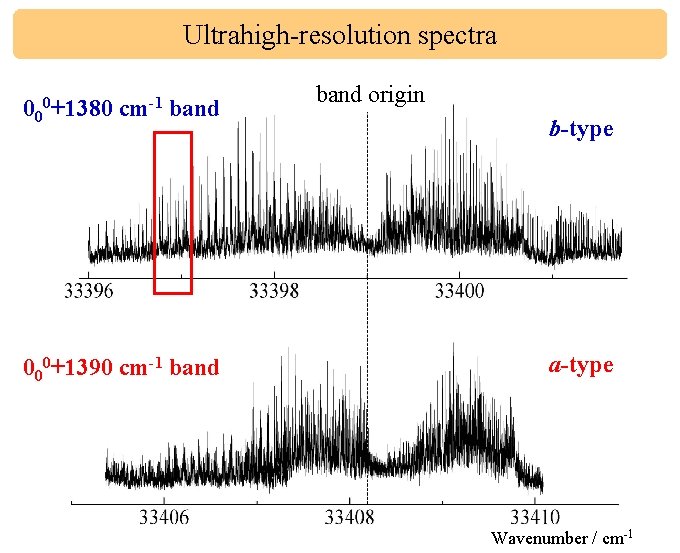
Ultrahigh-resolution spectra 000+1380 cm-1 band 000+1390 cm-1 band origin b-type a-type Wavenumber / cm-1

Comparison between observed and calculated spectrum Ka 000+1380 cm-1 band Ka obs. calc. Wavenumber / cm-1

Ultrahigh-resolution spectra 000+1380 cm-1 band 000+1390 cm-1 band origin b-type a-type Wavenumber / cm-1

Comparison between observed and calculated spectrum 000+1390 cm-1 band obs. calc. Wavenumber / cm-1

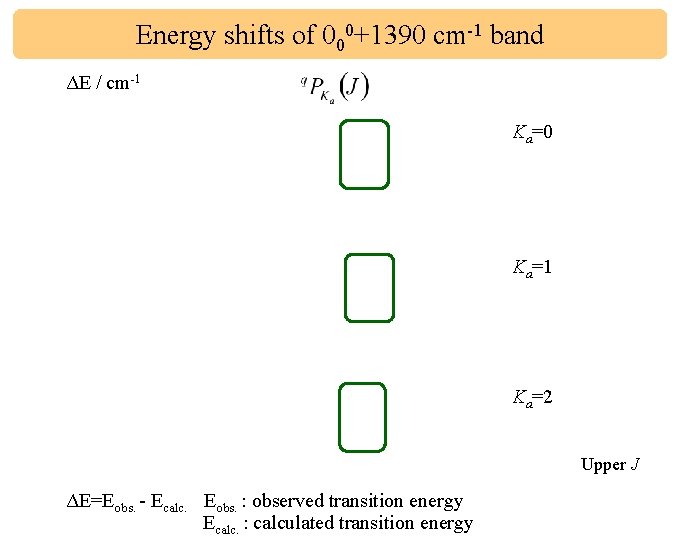
Energy shifts of 000+1390 cm-1 band ΔE / cm-1 Ka=0 Ka=1 Ka=2 Upper J ΔE=Eobs. - Ecalc. Eobs. : observed transition energy Ecalc. : calculated transition energy

The Zeeman Effect

High-resolution spectrum S 1 ←S 0 transition 1432 2122 2410 2570 000 910 1422 1380 1390 435 We observed the Zeeman effect for rotationally resolved spectra Wavenumber / cm-1

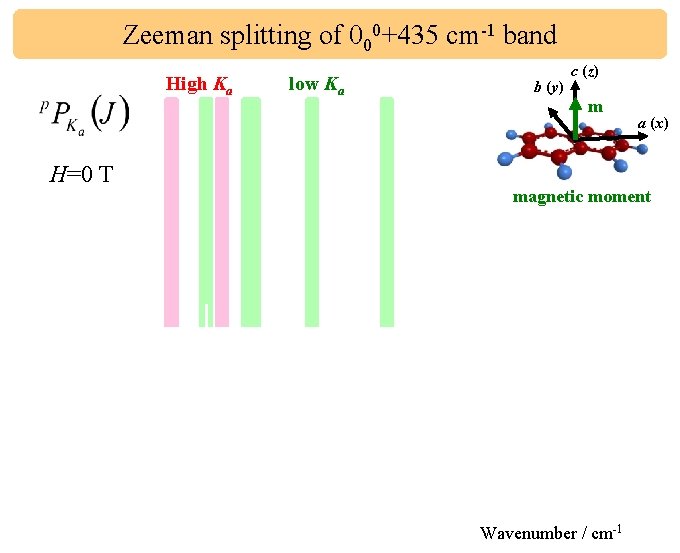
Zeeman splitting of 000+435 cm-1 band High Ka H=0 T low Ka b (y) c (z) m a (x) magnetic moment H=0. 50 T Wavenumber / cm-1

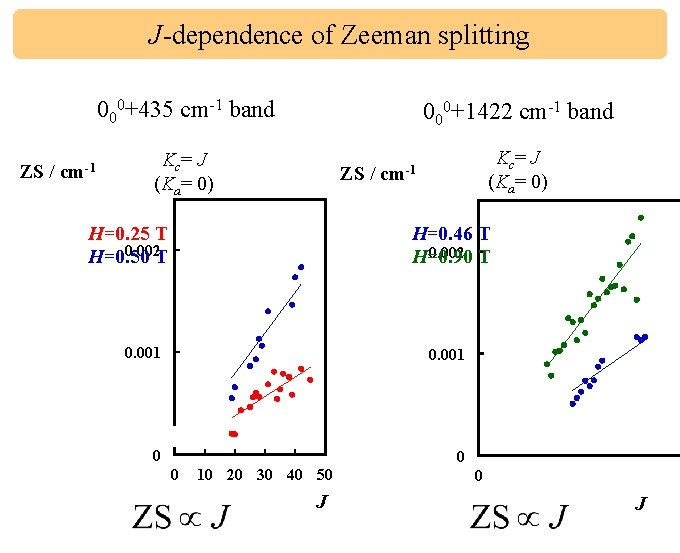
J-dependence of Zeeman splitting 000+435 cm-1 band ZS / cm-1 000+1422 cm-1 band Kc = J (Ka= 0) ZS / H=0. 25 T 0. 002 T H=0. 50 Kc = J (Ka= 0) cm-1 H=0. 46 T 0. 002 T H=0. 90 0. 001 0 0 0 10 20 30 40 50 J

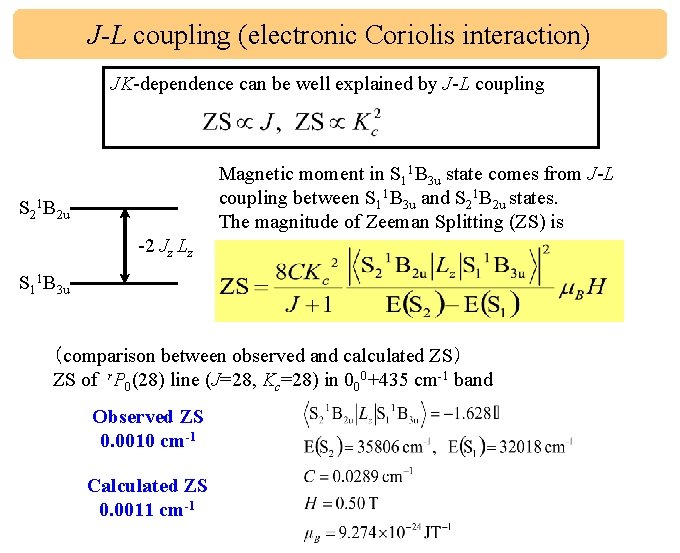
J-L coupling (electronic Coriolis interaction) JK-dependence can be well explained by J-L coupling Magnetic moment in S 11 B 3 u state comes from J-L coupling between S 11 B 3 u and S 21 B 2 u states. The magnitude of Zeeman Splitting (ZS) is S 21 B 2 u -2 Jz Lz S 11 B 3 u (comparison between observed and calculated ZS) ZS of r. P 0(28) line (J=28, Kc=28) in 000+435 cm-1 band Observed ZS 0. 0010 cm-1 Calculated ZS 0. 0011 cm-1

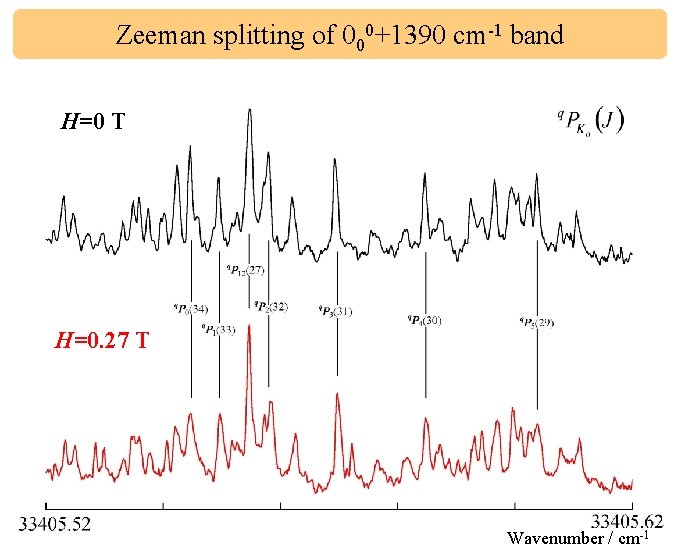
Zeeman splitting of 000+1390 cm-1 band H=0 T H=0. 27 T Wavenumber / cm-1

Summary We observed ultrahigh-resolution spectra of 000+1380 cm-1 and 000+1390 cm-1 vibronic bands of naphthalene S 1←S 0 transition. Several rotational lines of these vibronic bands were assigned and the rotational constants were determined in high accuracy. We determined vibrational energy of v 4, v 13 in high accuracy. In 000+1390 cm-1 band , the local energy shifts were found. The Zeeman splitting was very small and was proportional to J for a given K. The magnetic moment comes from an electron angular momentum induced by the J-L coupling between S 11 B 3 u and S 21 B 2 u states. The main nonradiative process of S 1 state is not intersystem crossing to the triplet state. It is presumed to be internal conversion to ground state.

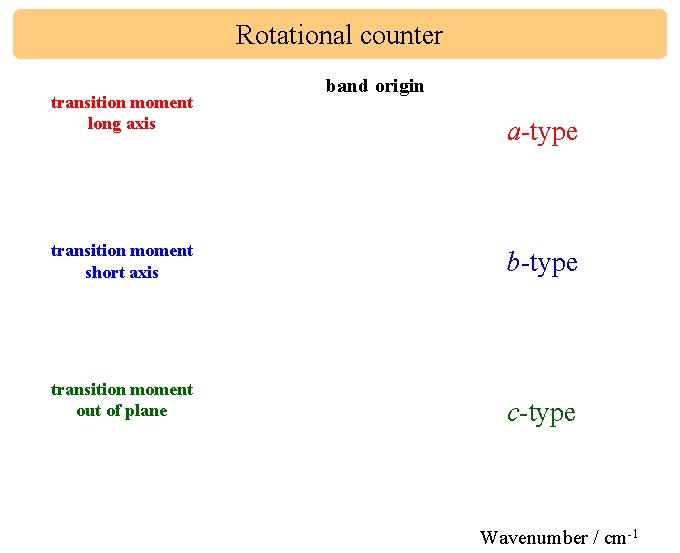
Rotational counter transition moment long axis band origin a-type transition moment short axis b-type transition moment out of plane c-type Wavenumber / cm-1

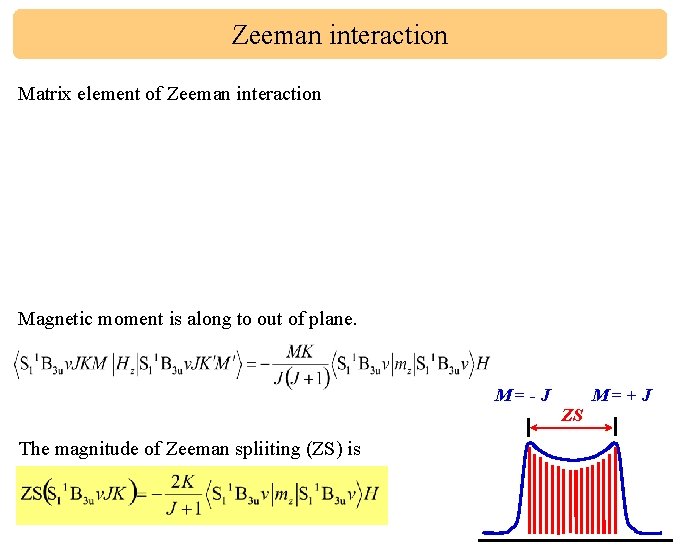
Zeeman interaction Matrix element of Zeeman interaction Magnetic moment is along to out of plane. M=-J M=+J ZS The magnitude of Zeeman spliiting (ZS) is

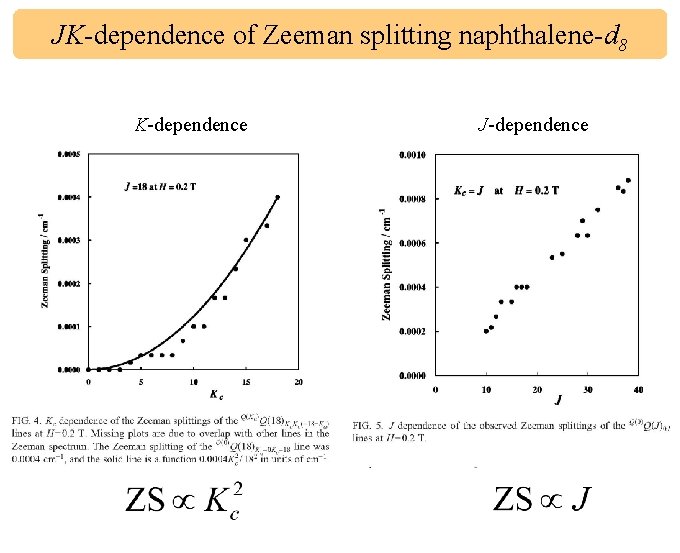
JK-dependence of Zeeman splitting naphthalene-d 8 K-dependence J-dependence

v-dependence of Zeeman splitting Zeeman Splitting / MHz at Ka=0 (Kc=J), J=20, H = 0. 2 T ZS is not v-dependence Excess Energy / cm-1

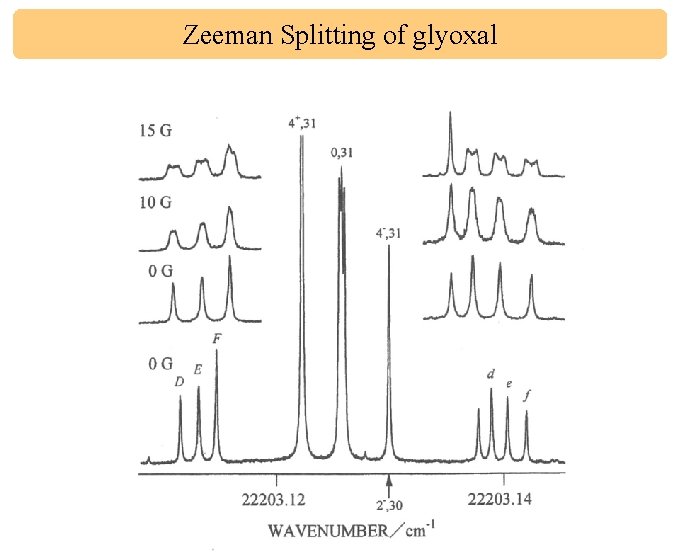
Zeeman Splitting of glyoxal

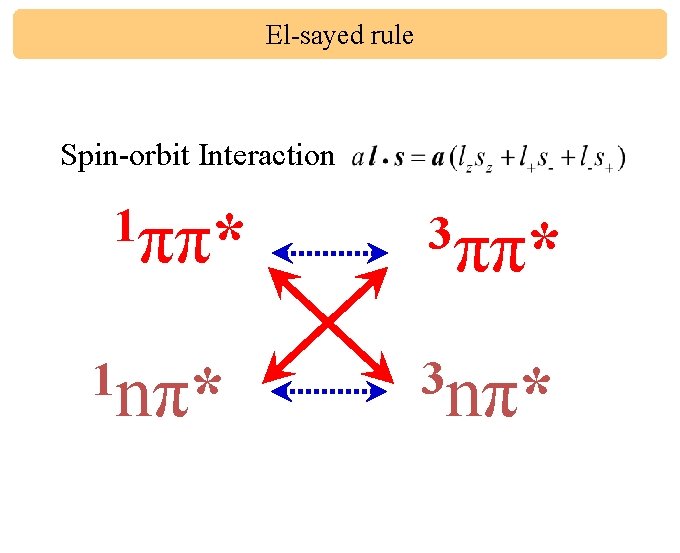
El-sayed rule Spin-orbit Interaction 1ππ* 1 nπ* 3ππ* 3 nπ*

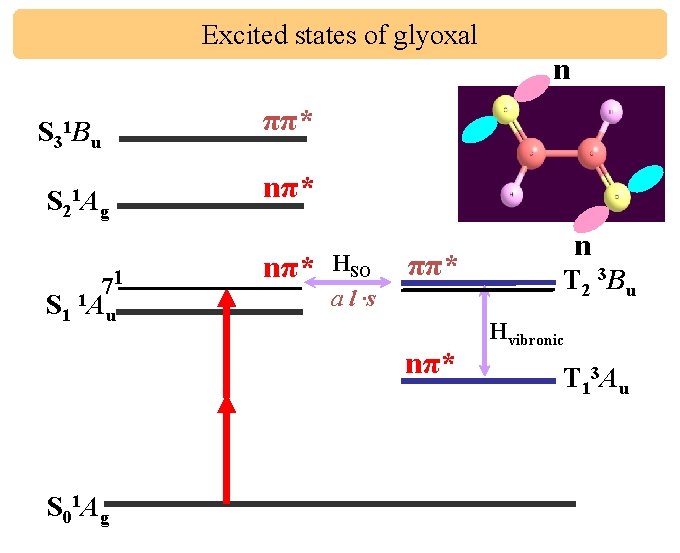
Excited states of glyoxal n ππ* S 31 Bu S 21 Ag 71 S 1 1 Au nπ* HSO a l ·s ππ* nπ* S 01 Ag n T 2 3 Bu Hvibronic T 13 Au

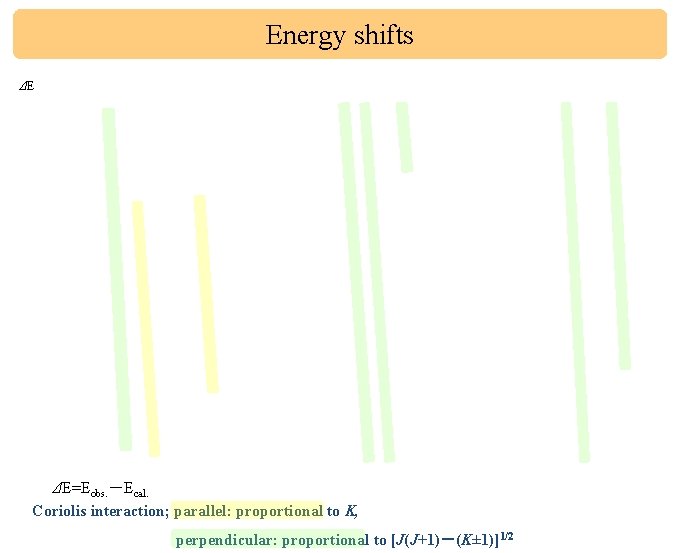
Energy shifts ΔE ΔE=Eobs. -Ecal. Coriolis interaction; parallel: proportional to K, perpendicular: proportional to [J(J+1)-(K± 1)]1/2