Ultrafast Vibrational Cooling Dynamics in 9 Methyladenine Observed
Ultrafast Vibrational Cooling Dynamics in 9 Methyladenine Observed with UV Pump/UV Probe Transient Absorption Spectroscopy Chris Middleton, Boiko Cohen and Bern Kohler The Ohio State University Department of Chemistry
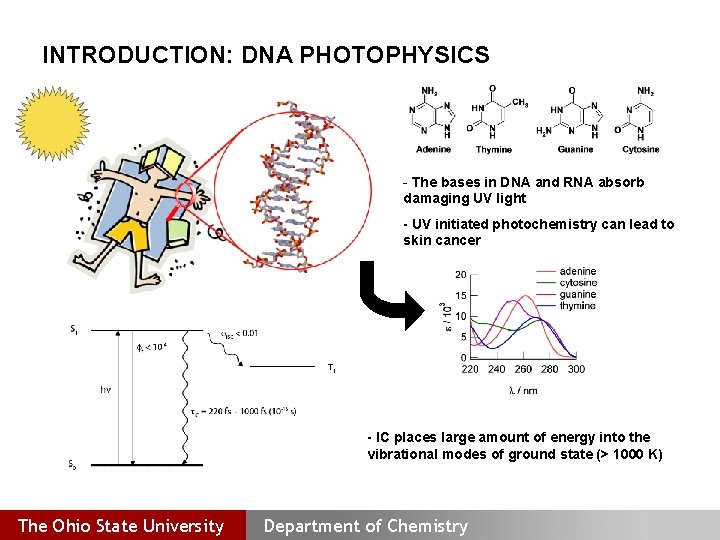
INTRODUCTION: DNA PHOTOPHYSICS - The bases in DNA and RNA absorb damaging UV light UV initiated photochemistry can lead to skin cancer IC places large amount of energy into the vibrational modes of ground state (> 1000 K) The Ohio State University Department of Chemistry
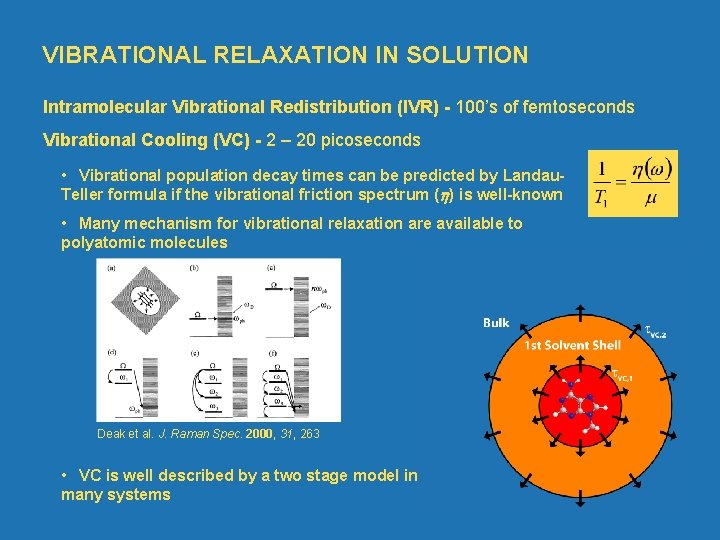
VIBRATIONAL RELAXATION IN SOLUTION Intramolecular Vibrational Redistribution (IVR) 100’s of femtoseconds Vibrational Cooling (VC) 2 – 20 picoseconds • Vibrational population decay times can be predicted by Landau Teller formula if the vibrational friction spectrum ( ) is well known • Many mechanism for vibrational relaxation are available to polyatomic molecules Deak et al. J. Raman Spec. 2000, 31, 263 • VC is well described by a two stage model in many systems
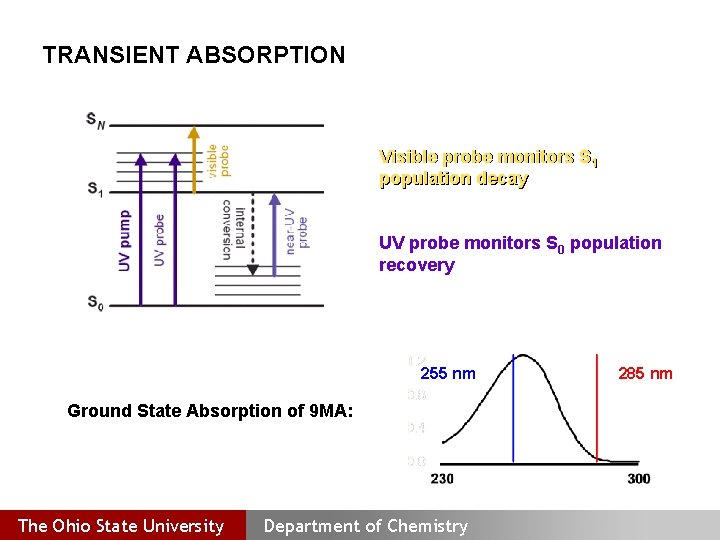
TRANSIENT ABSORPTION Visible probe monitors S 1 population decay UV probe monitors S 0 population recovery 255 nm Ground State Absorption of 9 MA: The Ohio State University Department of Chemistry 285 nm
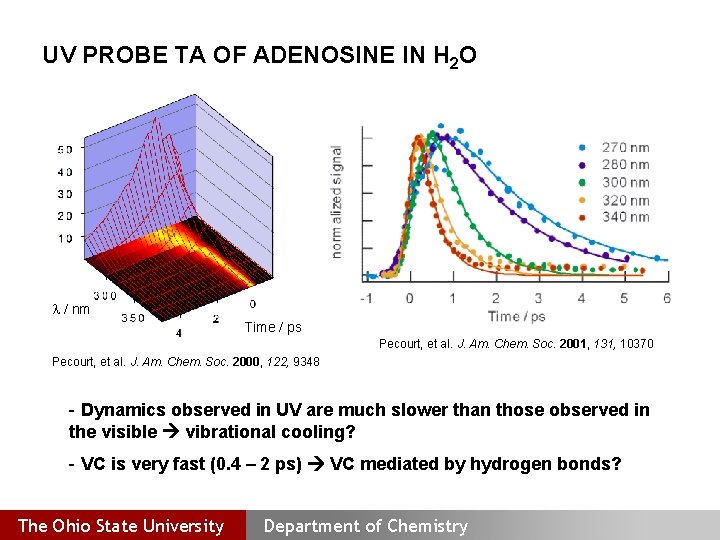
UV PROBE TA OF ADENOSINE IN H 2 O l / nm Time / ps Pecourt, et al. J. Am. Chem. Soc. 2001, 131, 10370 Pecourt, et al. J. Am. Chem. Soc. 2000, 122, 9348 - Dynamics observed in UV are much slower than those observed in the visible vibrational cooling? - VC is very fast (0. 4 – 2 ps) VC mediated by hydrogen bonds? The Ohio State University Department of Chemistry
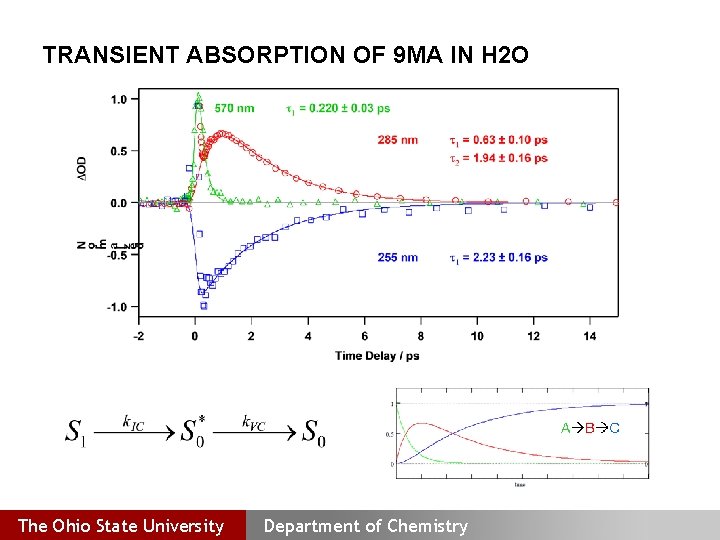
TRANSIENT ABSORPTION OF 9 MA IN H 2 O The Ohio State University Department of Chemistry
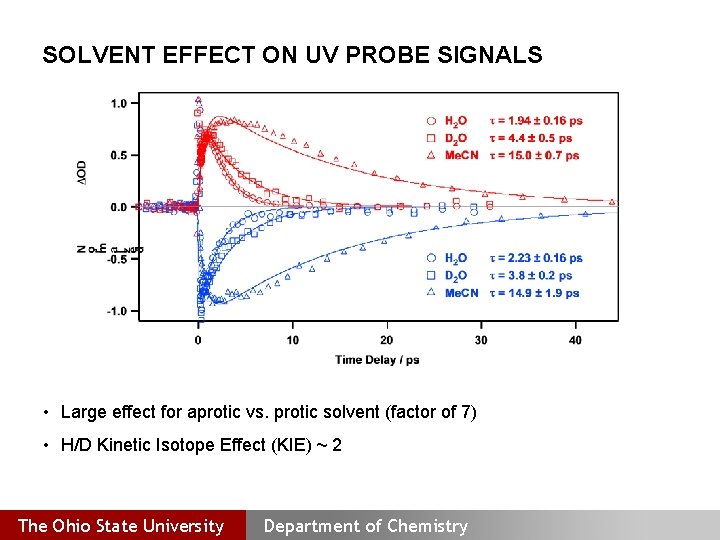
SOLVENT EFFECT ON UV PROBE SIGNALS • Large effect for aprotic vs. protic solvent (factor of 7) • H/D Kinetic Isotope Effect (KIE) ~ 2 The Ohio State University Department of Chemistry
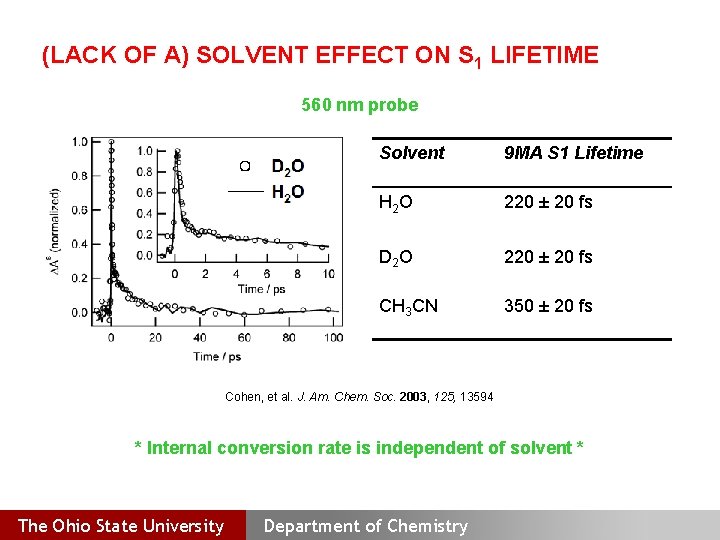
(LACK OF A) SOLVENT EFFECT ON S 1 LIFETIME 560 nm probe Solvent 9 MA S 1 Lifetime H 2 O 220 ± 20 fs D 2 O 220 ± 20 fs CH 3 CN 350 ± 20 fs Cohen, et al. J. Am. Chem. Soc. 2003, 125, 13594 * Internal conversion rate is independent of solvent * The Ohio State University Department of Chemistry
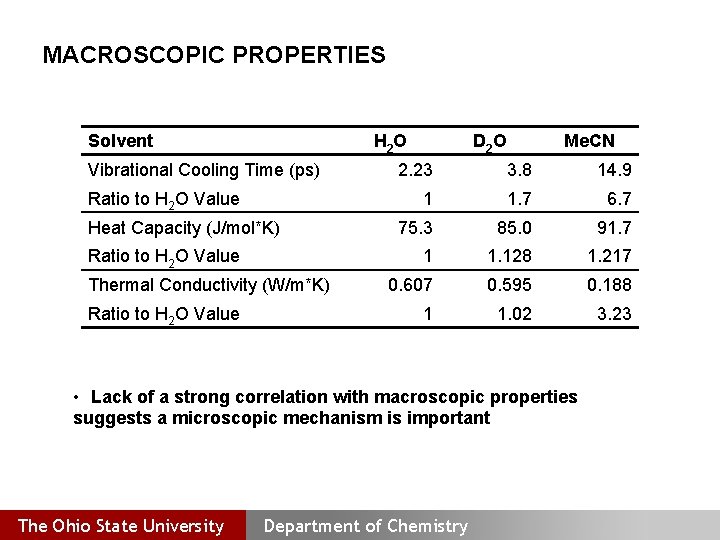
MACROSCOPIC PROPERTIES Solvent H 2 O Vibrational Cooling Time (ps) Ratio to H 2 O Value Heat Capacity (J/mol*K) Ratio to H 2 O Value Thermal Conductivity (W/m*K) Ratio to H 2 O Value D 2 O Me. CN 2. 23 3. 8 14. 9 1 1. 7 6. 7 75. 3 85. 0 91. 7 1 1. 128 1. 217 0. 607 0. 595 0. 188 1 1. 02 3. 23 • Lack of a strong correlation with macroscopic properties suggests a microscopic mechanism is important The Ohio State University Department of Chemistry
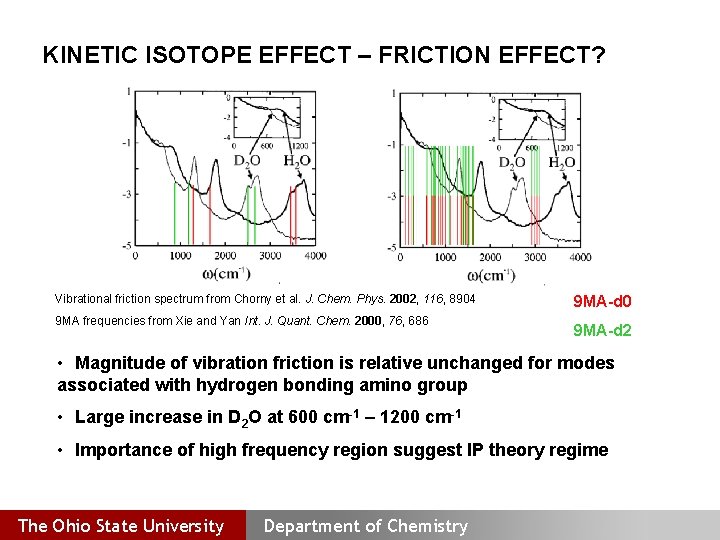
KINETIC ISOTOPE EFFECT – FRICTION EFFECT? Vibrational friction spectrum from Chorny et al. J. Chem. Phys. 2002, 116, 8904 9 MA frequencies from Xie and Yan Int. J. Quant. Chem. 2000, 76, 686 9 MA d 0 9 MA d 2 • Magnitude of vibration friction is relative unchanged for modes associated with hydrogen bonding amino group • Large increase in D 2 O at 600 cm 1 – 1200 cm 1 • Importance of high frequency region suggest IP theory regime The Ohio State University Department of Chemistry

KINETIC ISOTOPE EFFECT – HYDROGEN BONDING? • Vibrational population decay time is proportional to reduced mass • Vibrational friction for amino group modes is the same in H 2 O and D 2 O • Good agreement with KIE for 9 MA = 1. 72 ± 0. 08 The Ohio State University Department of Chemistry
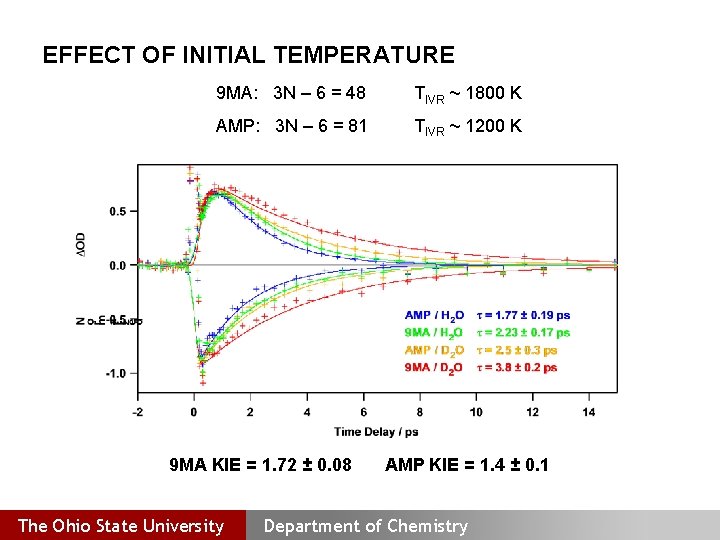
EFFECT OF INITIAL TEMPERATURE 9 MA: 3 N – 6 = 48 TIVR ~ 1800 K AMP: 3 N – 6 = 81 TIVR ~ 1200 K 9 MA KIE = 1. 72 ± 0. 08 The Ohio State University AMP KIE = 1. 4 ± 0. 1 Department of Chemistry

CONCLUSIONS • Long dynamics observed in the UV are assigned to vibrational cooling of the hot ground state • Large acceleration VC rate in protic solvents suggests the hydrogen bonds strongly mediate VC • A KIE isotope effect for VC has been observed with two possible mechanisms QUESTIONS RAISED • Solute solvent vs. solvent hydrogens bonds? • Importance of lower frequency vs. higher frequency modes? The Ohio State University Department of Chemistry

ACKNOWLEDGEMENTS Dr. Bern Kohler Dr. Boiko Cohen (Wasielewski Group, Northwestern University) Kohler Group Gustafson Group NIH International Symposium on Molecular Spectroscopy The Ohio State University Department of Chemistry
- Slides: 14