Ultrafast Shortrange Electron Transfer Dynamics in Biology TingFang

Ultrafast Short-range Electron Transfer Dynamics in Biology Ting-Fang He Dr. Dongping Zhong Programs of Ohio State Biochemistry, Biophysics, and Chemical Physics, and Departments of Physics, Chemistry, and Biochemistry, The Ohio State University, Columbus, Ohio 43210
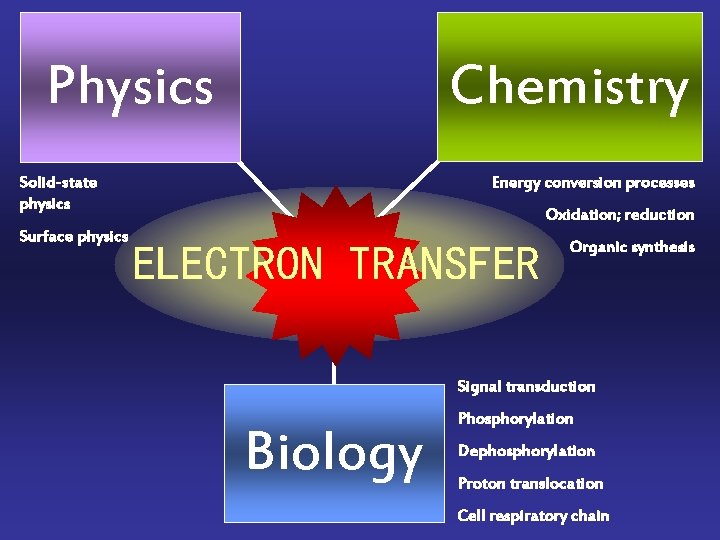
Chemistry Physics Solid-state physics Surface physics Energy conversion processes Oxidation; reduction ELECTRON TRANSFER Organic synthesis Signal transduction Biology Phosphorylation Dephosphorylation Proton translocation Cell respiratory chain
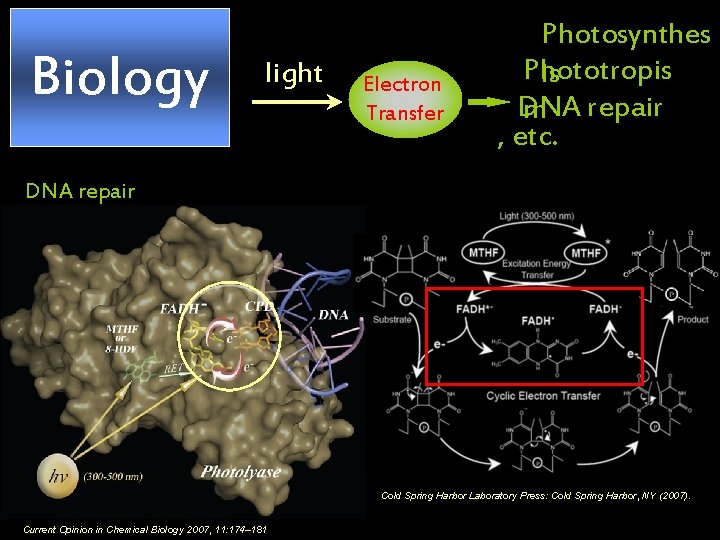
Biology light Electron Transfer Photosynthes Phototropis is DNA repair m , etc. DNA repair Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY (2007). Current Opinion in Chemical Biology 2007, 11: 174– 181
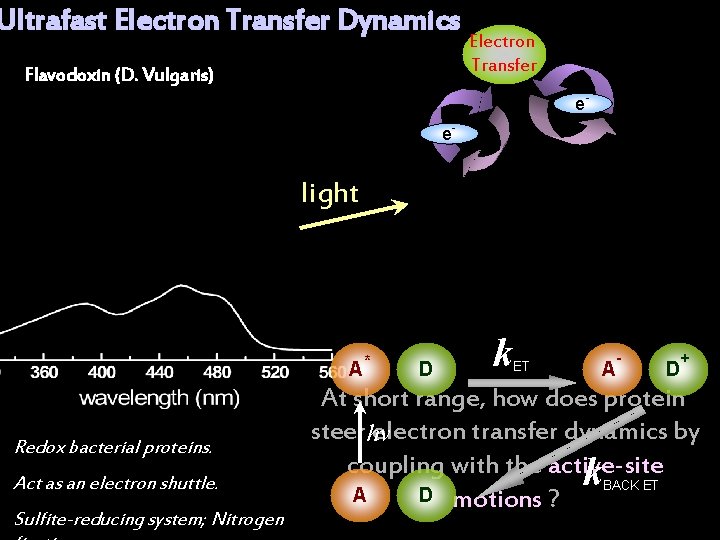
Ultrafast Electron Transfer Dynamics Flavodoxin (D. Vulgaris) Electron Transfer e- e- light A Redox bacterial proteins. Act as an electron shuttle. Sulfite-reducing system; Nitrogen * D k ET A - D + At short range, how does protein steer electron transfer dynamics by hν coupling with the active-site k BACK ET A D motions ?
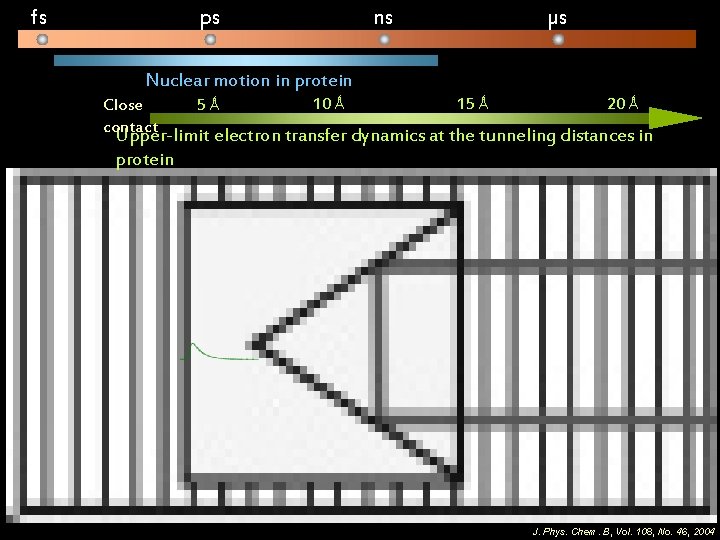
fs ps ns µs Nuclear motion in protein Close contact 5 Ǻ 10 Ǻ 15 Ǻ 20 Ǻ Upper-limit electron transfer dynamics at the tunneling distances in protein J. Phys. Chem. B, Vol. 108, No. 46, 2004
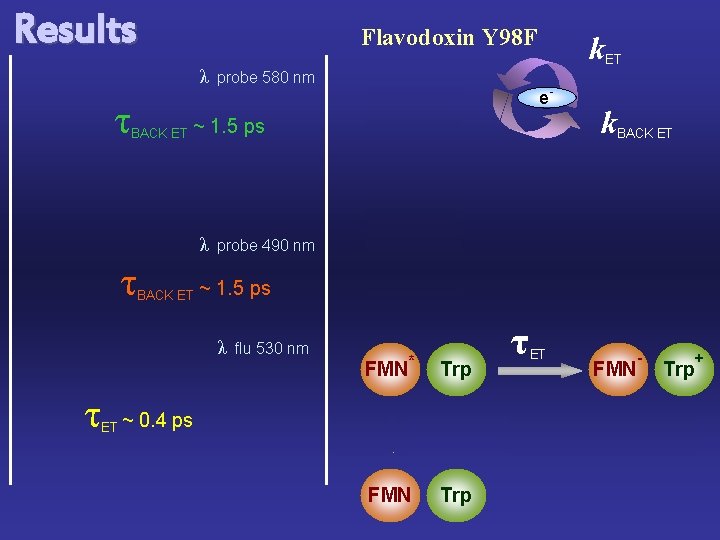
Results Flavodoxin Y 98 F λ τ BACK ET ET ~ e- BACK ET 0. 4 ps k. BACK ET probe 490 nm ~ 1. 5 ps λ τ e- ~ 1. 5 ps λ τ probe 580 nm k. ET flu 530 nm FMN * Trp 400 nm FMN τ ET FMN τ Trp - Trp BACK ET +
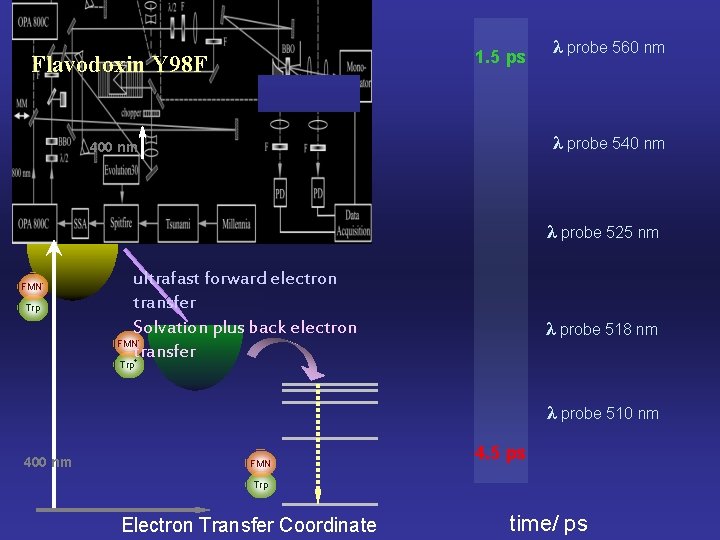
Results 1. 5 ps Flavodoxin Y 98 F λ probe 560 nm λ probe 540 nm 400 nm λ probe 525 nm FMN* Trp ultrafast forward electron transfer Solvation plus back electron FMN transfer λ probe 518 nm - Trp+ λ probe 510 nm 400 nm FMN 4. 5 ps Trp Electron Transfer Coordinate time/ ps
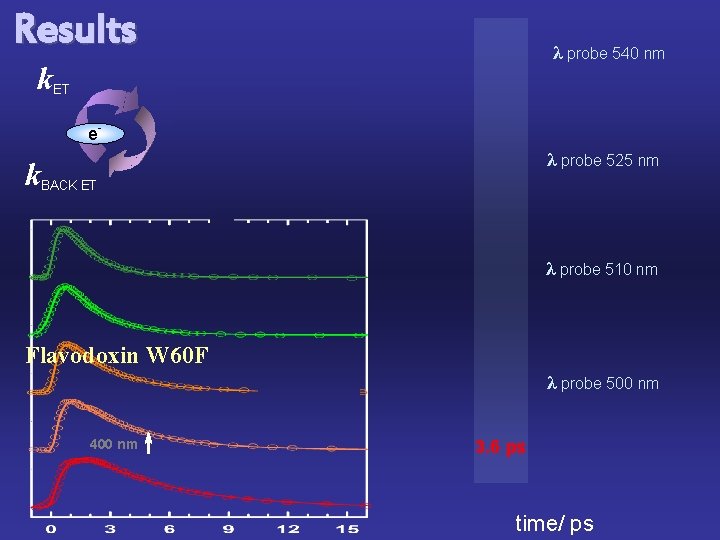
Results k. ET λ probe 540 nm e- e- λ probe 525 nm k. BACK ET λ probe 510 nm Flavodoxin W 60 F λ probe 500 nm 400 nm 3. 6 ps time/ ps
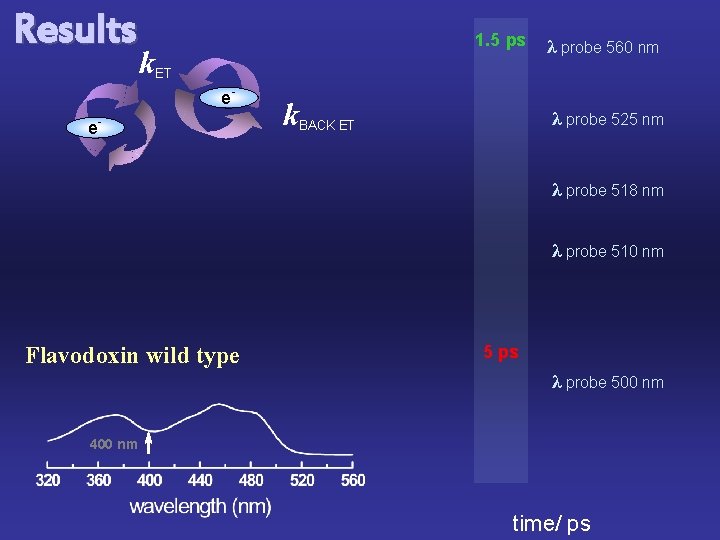
Results 1. 5 ps k. ET e- e- k. BACK ET λ probe 560 nm λ probe 525 nm λ probe 518 nm λ probe 510 nm Flavodoxin wild type 5 ps λ probe 500 nm 400 nm time/ ps

Conclusio (a) Under the oxidized flavodoxin, the forward electron transfer n dynamics from the van der Waals contact aromatic amino acids to the excited flavin is determined faster than 0. 5 ps. (b) The back electron transfer turns out being more than two times slower than its forward, ~1. 5 ps. (c) The recombination process is found at the hot ground state before the redox molecules and the protein solvents relax vibrationally to the equilibrated configurations. (d) The subsequent vibrational relaxation (cooling) is determined ~5 ps in the active-site pocket. (e) The study indicates that, for the highly exergonic back electron transfer (the inverted region), at this short range, there appears a hot channel at the vibrational excited electronic ground state to boost the return dynamics. How universal in regulating the biological function?
Acknowledgeme nts Advisor Group Members Funding Dr. Dongping Zhong Qing Ding the National Institutes of Health Dr. Xunmin Guo the Packard Foundation Fellowship Ali Hassanali Dr. Jiang Li Tangping Li Zheyun Liu Jingwei Lu Justin Link Oseoghaghare Okobiah Weihong Qiu Jeff Stevens Chuang Tan Yi Yang Chen Zang Luyuan Zhang Professor Richard P. Swenson Coworkers Lijuan Wang Dr. Lijun Guo Dr. Chaitanya Saxena Ya-Ting Kao
- Slides: 11