Ultrafast Photochemistry of Bromoform in Solution Probed in

Ultrafast Photochemistry of Bromoform in Solution Probed in the Deep-UV- to Near-IR Spectral Range Suman K. Pal, Patrick Z. El-Khoury, Andrey S. Mereshchenko and Alexander N. Tarnovsky Department of Chemistry and the Center for Photochemical Sciences Bowling Green State University, Bowling Green, Ohio

Outline v Objectives v Previous work on bromoform (CHBr 3) v Methods: pump-probe and TD-DFT v Results v Summary

Objectives o Environmental importance: produced by microalgae, 200, 000 tons per year o Atmospheric importance: abundant source of bromine atoms in the earth’s atmosphere causing destruction of the ozone layer o Fundamental interest: structure-selective insights into the condensed-phase photochemistry
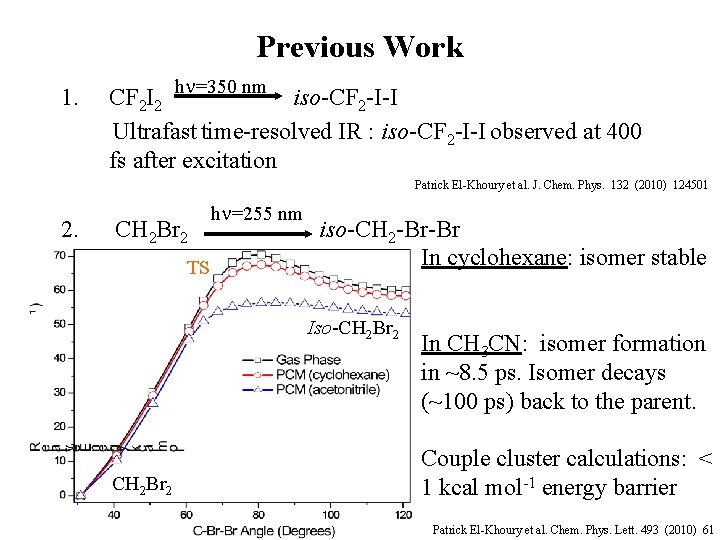
Previous Work 1. CF 2 I 2 h =350 nm iso-CF 2 -I-I Ultrafast time-resolved IR : iso-CF 2 -I-I observed at 400 fs after excitation Patrick El-Khoury et al. J. Chem. Phys. 132 (2010) 124501 2. CH 2 Br 2 TS h =255 nm iso-CH 2 -Br-Br In cyclohexane: isomer stable Iso-CH 2 Br 2 In CH 3 CN: isomer formation in ~8. 5 ps. Isomer decays (~100 ps) back to the parent. Couple cluster calculations: < 1 kcal mol-1 energy barrier Patrick El-Khoury et al. Chem. Phys. Lett. 493 (2010) 61

Bromoform: Early Gas-Phase Work • Excitation at ~200 nm exclusively causes direct two-body decay followed by the C—Br bond dissociation in vibrationally hot radicals CHBr 3* CHBr 2·* + Br· CHBr 2·* CHBr·+ Br· Excitation at 193 nm: HBr elimination from vibrationally hot CHBr 2 radicals CHBr 2·* CBr+ HBr • Excitation in the 234 -267 nm range: molecular bromine elimination CHBr 3* CHBr + Br 2 Simons et al. Trans. Faraday Soc. 57 (1961) 2167 Mc. Givern et al. J. Phys. Chem. A 104 (2002) 10085 Zou et al. J. Phys. Chem. A 108 (2004) 1482 Xu and co-workers J. Chem. Phys. 117 (2002) 2578

Bromoform: Previous Liquid-Phase Work • Excitation at ~267 nm in water and water/acetonitrile mixtures causes formation of iso-bromoform, which finally reacts with water to form HBr, CO and HCO. Phillips and co-workers J. Chem. Phys. 120 (2004) 3323; J. Am. Chem. Soc. 126 (2004) 3119 • Neat CHBr 3 and concentrated ( 0. 23 M) cyclohexane solutions upon excitation at 267 nm: iso-bromoformation: prompt (<0. 5 ps) hot iso-bromoform decay: 13 ps lifetime relaxed iso-bromoform: metastable release of Br atoms from hot iso-bromoform: rapid formation of CHBr 3 -Br complex in concentrated solutions Crim and co-workers J. Phys. Chem. A 114 (2010) 1548

Ultrafast (100 fs) Laser System We Built: Gap-Free Tunable 208 nm – 22 mm Visible probe 0. 32 -0. 78 mm 0. 23 -2. 6 mm UV/vis/near-IR pump UV/vis/IR probe (IR: with Dr. K. Glusac) 266 nm pump/probe 0. 208 -22 mm 0. 266 mm Accessible Spectral Range Continuum probe TOPAS/pump TOPAS/probe Tripler 800/266 Hurricane (100 fs, 800 nm, 0. 8 m. J, 1 KHz)
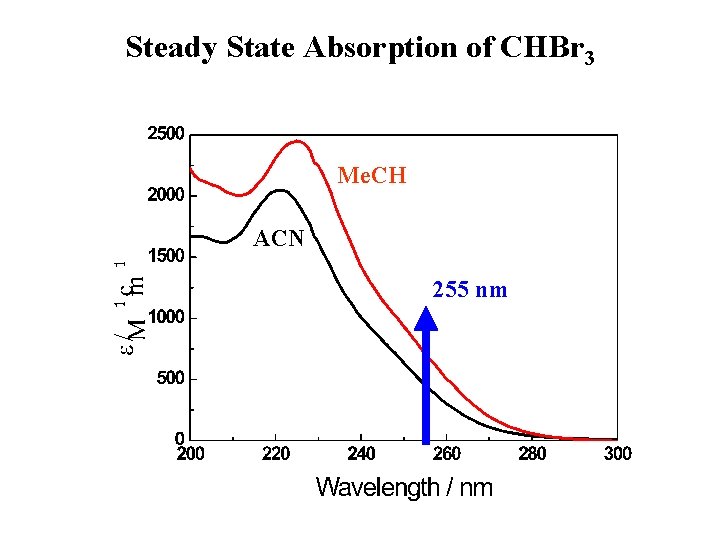
Steady State Absorption of CHBr 3 Me. CH ACN 255 nm
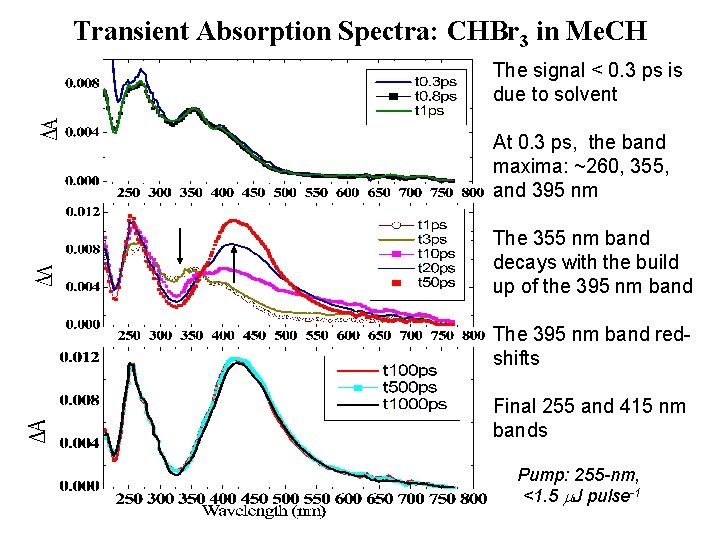
Transient Absorption Spectra: CHBr 3 in Me. CH The signal < 0. 3 ps is due to solvent At 0. 3 ps, the band maxima: ~260, 355, and 395 nm The 355 nm band decays with the build up of the 395 nm band The 395 nm band redshifts Final 255 and 415 nm bands Pump: 255 -nm, <1. 5 m. J pulse-1
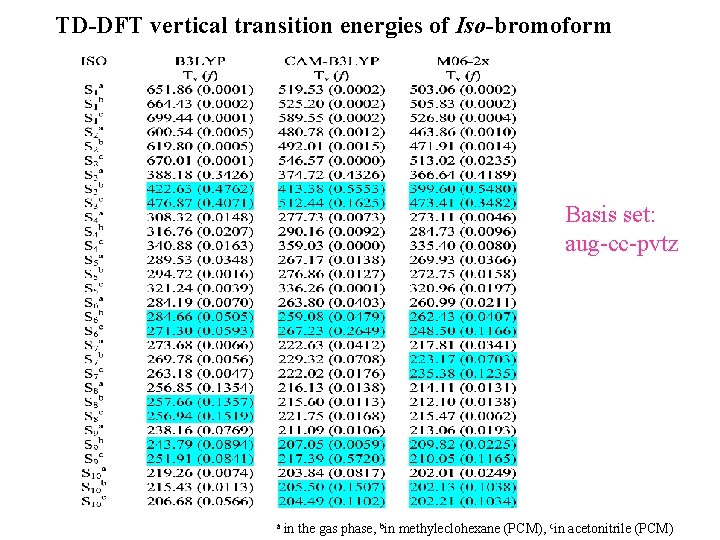
TD-DFT vertical transition energies of Iso-bromoform Basis set: aug-cc-pvtz a in the gas phase, bin methyleclohexane (PCM), cin acetonitrile (PCM)
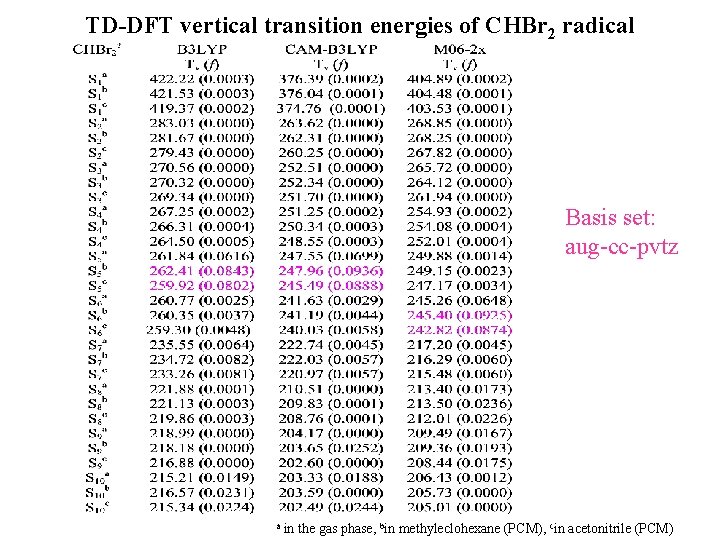
TD-DFT vertical transition energies of CHBr 2 radical Basis set: aug-cc-pvtz a in the gas phase, bin methyleclohexane (PCM), cin acetonitrile (PCM)
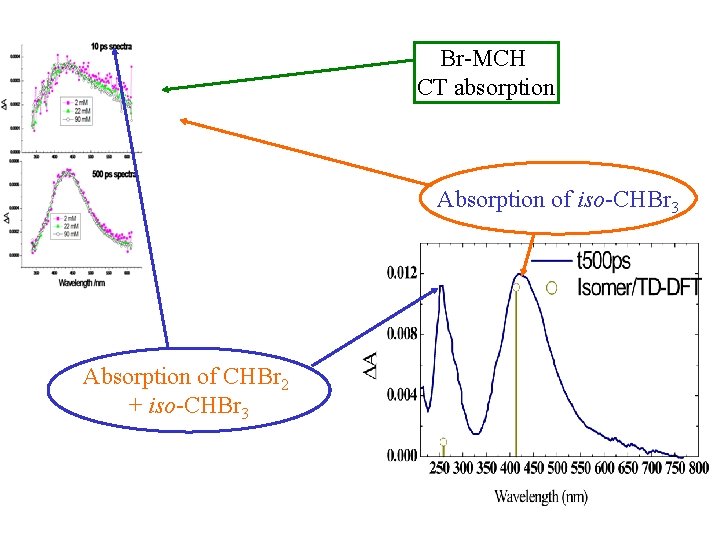
Br-MCH CT absorption Absorption of iso-CHBr 3 Absorption of CHBr 2 + iso-CHBr 3
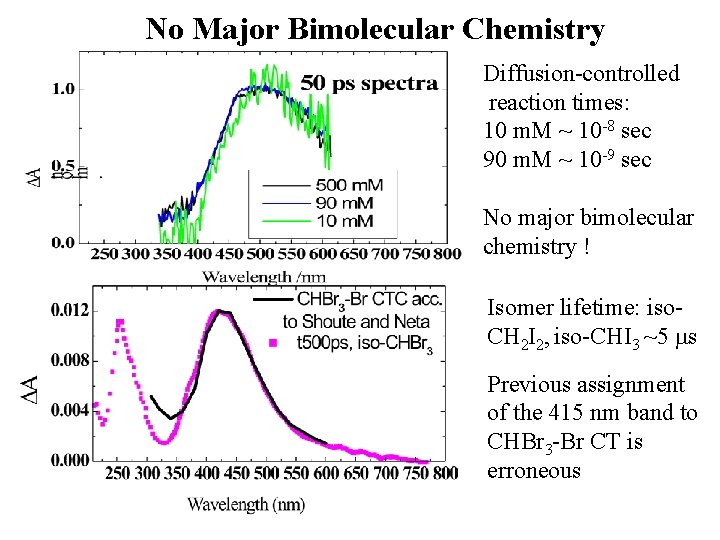
No Major Bimolecular Chemistry Diffusion-controlled reaction times: 10 m. M ~ 10 -8 sec 90 m. M ~ 10 -9 sec No major bimolecular chemistry ! Isomer lifetime: iso. CH 2 I 2, iso-CHI 3 ~5 ms Previous assignment of the 415 nm band to CHBr 3 -Br CT is erroneous
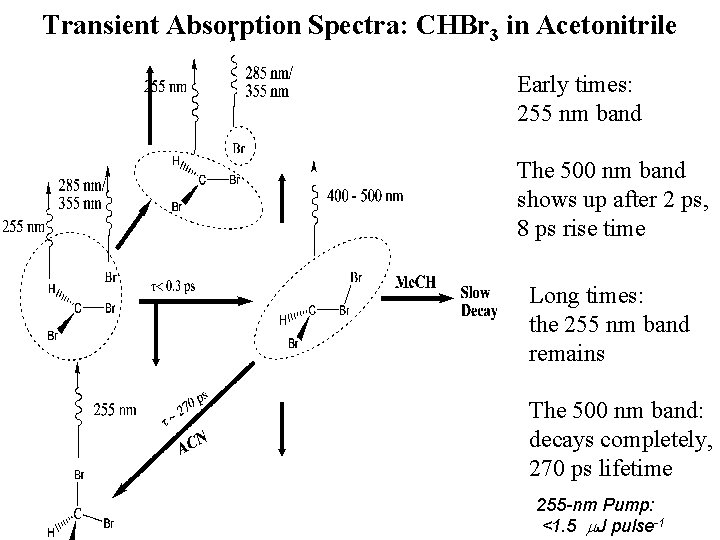
Transient Absorption Spectra: CHBr 3 in Acetonitrile Early times: 255 nm band The 500 nm band shows up after 2 ps, 8 ps rise time Long times: the 255 nm band remains The 500 nm band: decays completely, 270 ps lifetime 255 -nm Pump: <1. 5 m. J pulse-1
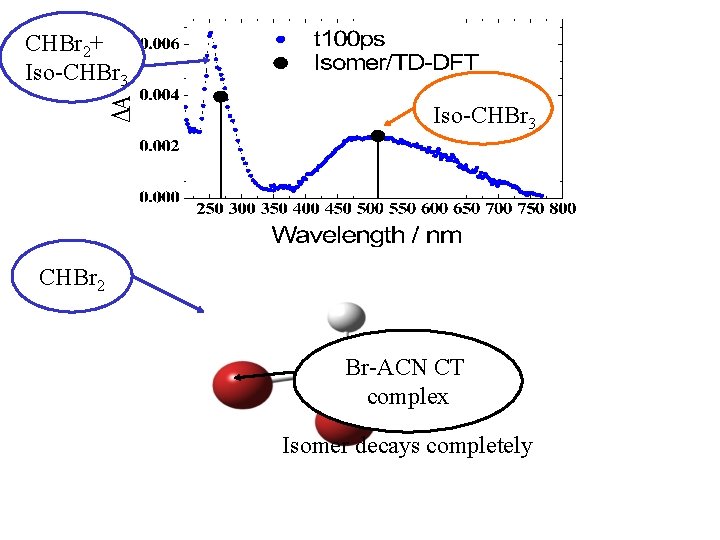
CHBr 2+ Iso-CHBr 3 CHBr 2 Br-ACN CT complex Isomer decays completely
Stabilization of the TS by Polar Solvent 1 TSiso 2 2 1 1 Iso-bromoform readily converts back to the parent in acetonitrile
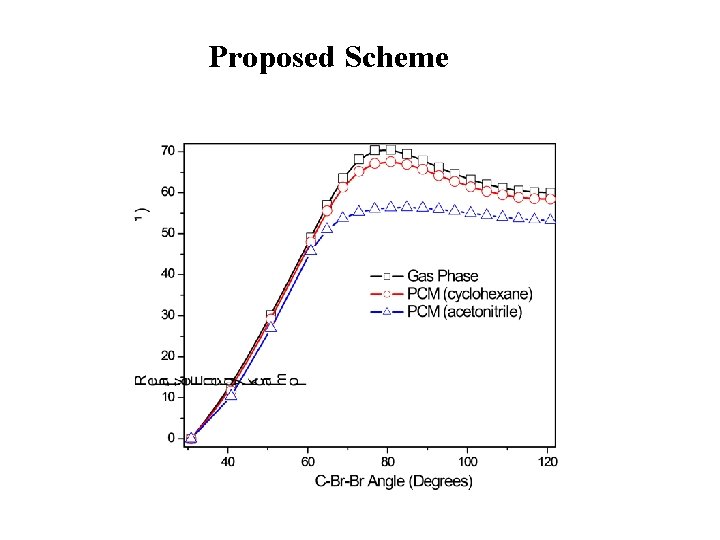
Proposed Scheme

Acknowledgments • Tarnovsky Group • Prof. Scott Reid (Marquette University) • Ohio Supercomputer Center • Funding NSF CAREER (Tarnovsky A. N. )
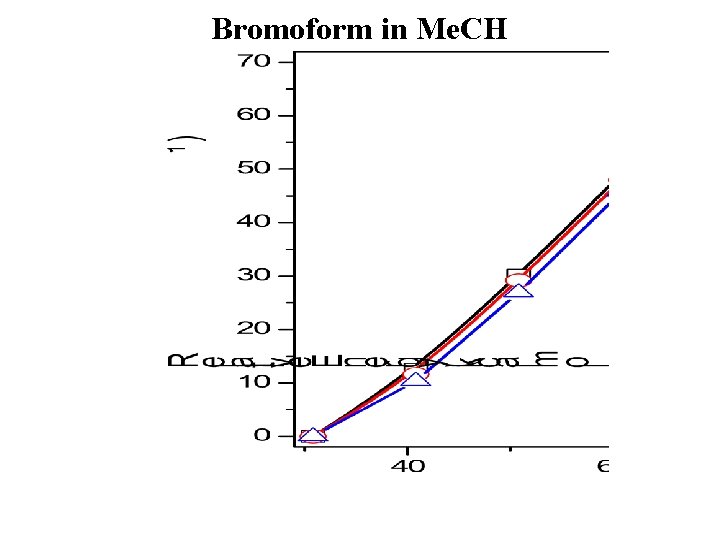
Bromoform in Me. CH
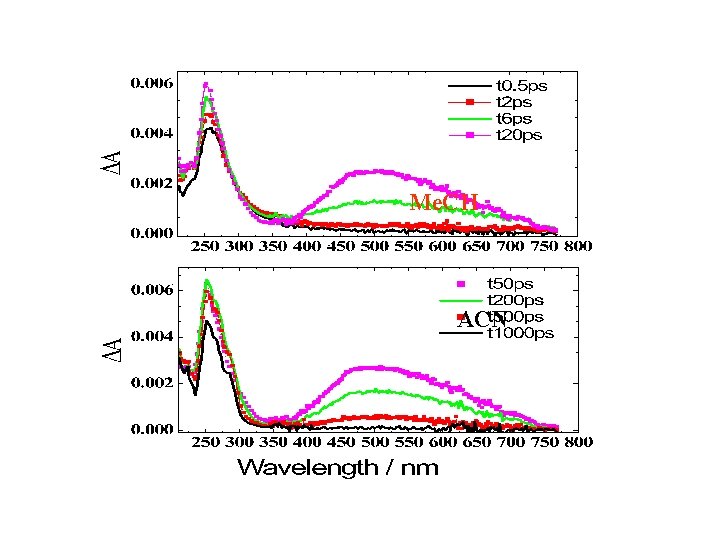
Me. CH ACN
Proposed Scheme
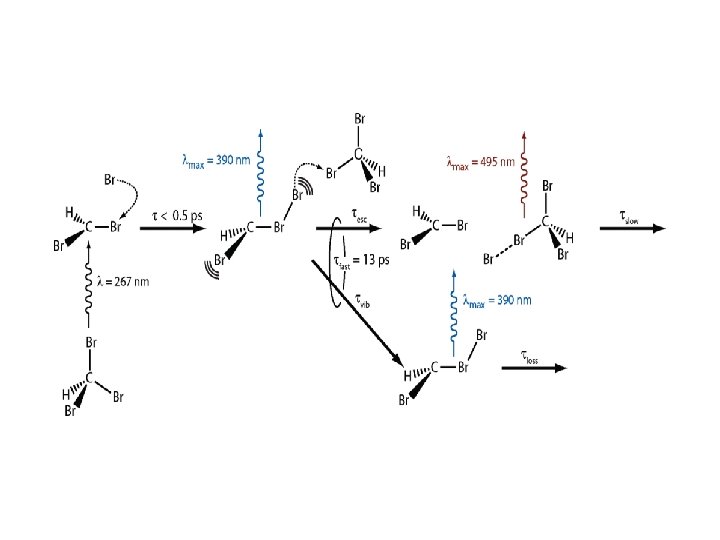
- Slides: 22