UK MULTICENTRE TRIAL IN ATHEROSCLEROTIC RENOVASCULAR DISEASE ASTRAL


- Slides: 18

UK MULTI-CENTRE TRIAL IN ATHEROSCLEROTIC RENOVASCULAR DISEASE ASTRAL Angioplasty and STent for Renal Artery Lesions Philip A Kalra Lead Nephrologist for ASTRAL, Hope Hospital, Salford, UK, On behalf of the ASTRAL TMC and collaborators

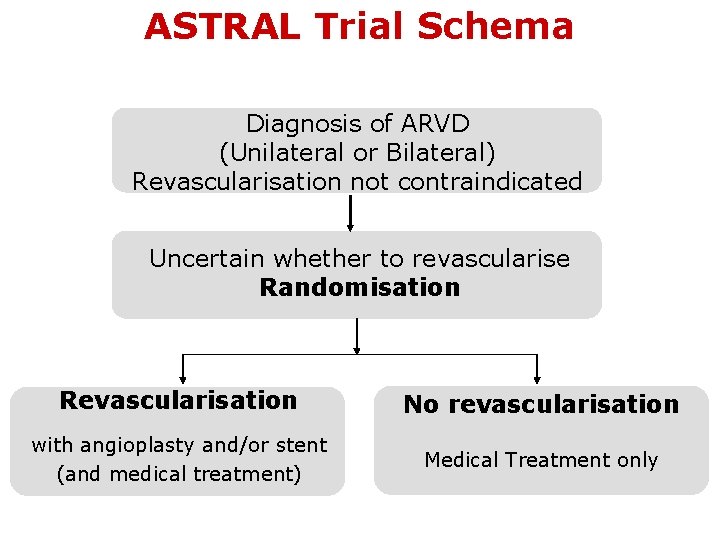
ASTRAL Trial Schema Diagnosis of ARVD (Unilateral or Bilateral) Revascularisation not contraindicated Uncertain whether to revascularise Randomisation Revascularisation No revascularisation with angioplasty and/or stent (and medical treatment) Medical Treatment only

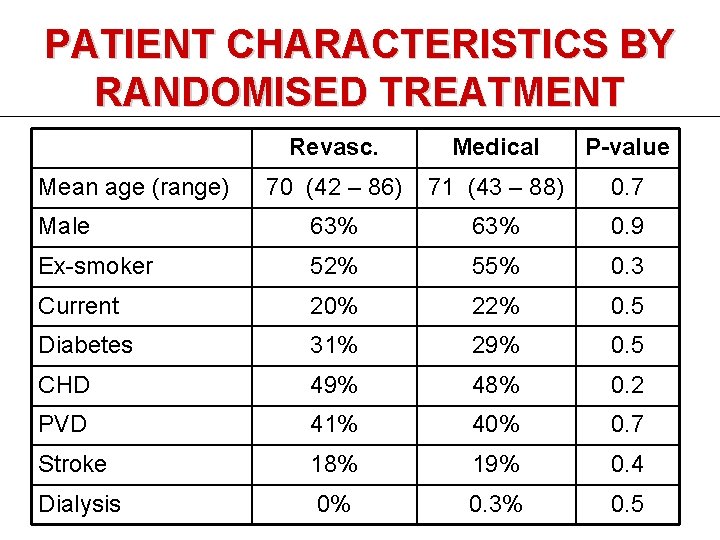
PATIENT CHARACTERISTICS BY RANDOMISED TREATMENT Revasc. Medical P-value 70 (42 – 86) 71 (43 – 88) 0. 7 Male 63% 0. 9 Ex-smoker 52% 55% 0. 3 Current 20% 22% 0. 5 Diabetes 31% 29% 0. 5 CHD 49% 48% 0. 2 PVD 41% 40% 0. 7 Stroke 18% 19% 0. 4 Dialysis 0% 0. 3% 0. 5 Mean age (range)

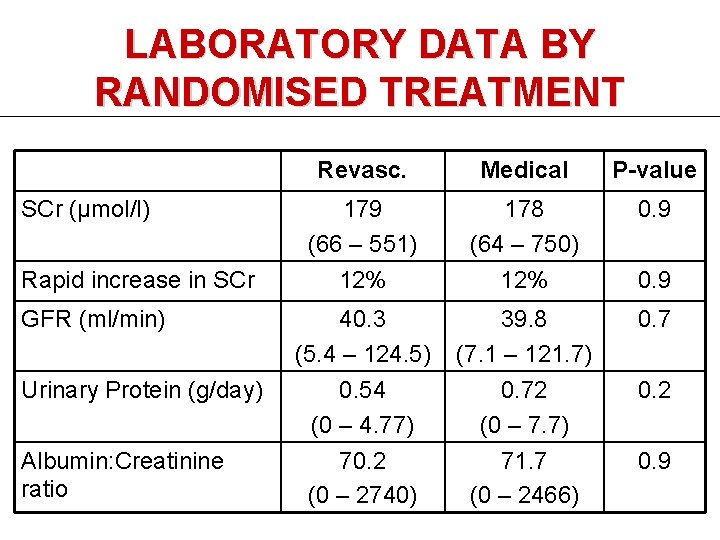
LABORATORY DATA BY RANDOMISED TREATMENT SCr (μmol/l) Rapid increase in SCr GFR (ml/min) Urinary Protein (g/day) Albumin: Creatinine ratio Revasc. Medical P-value 179 (66 – 551) 12% 178 (64 – 750) 12% 0. 9 40. 3 (5. 4 – 124. 5) 0. 54 (0 – 4. 77) 70. 2 (0 – 2740) 39. 8 (7. 1 – 121. 7) 0. 72 (0 – 7. 7) 71. 7 (0 – 2466) 0. 7 0. 9 0. 2 0. 9

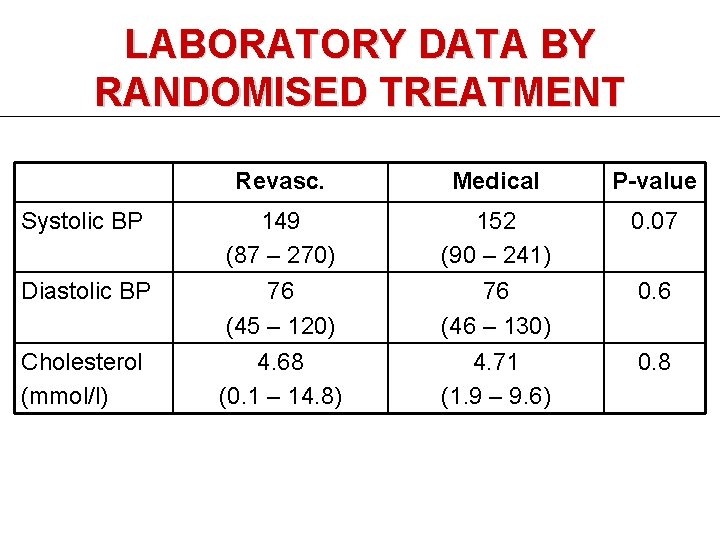
LABORATORY DATA BY RANDOMISED TREATMENT Systolic BP Diastolic BP Cholesterol (mmol/l) Revasc. Medical P-value 149 (87 – 270) 76 (45 – 120) 152 (90 – 241) 76 (46 – 130) 0. 07 4. 68 (0. 1 – 14. 8) 4. 71 (1. 9 – 9. 6) 0. 8 0. 6

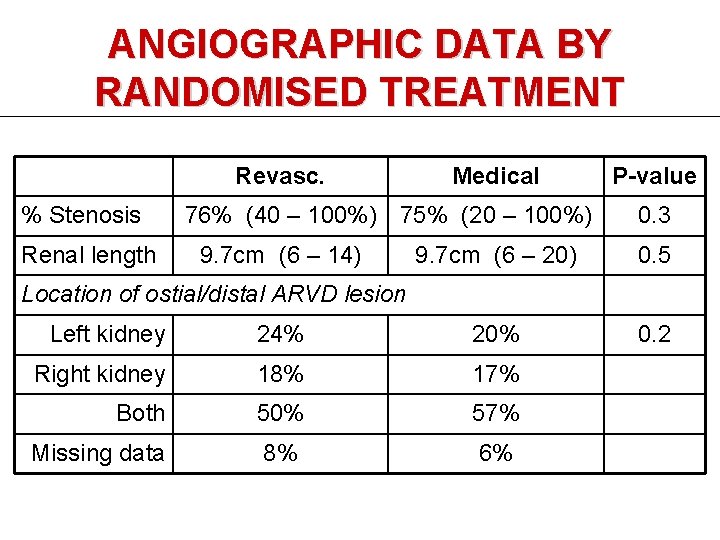
ANGIOGRAPHIC DATA BY RANDOMISED TREATMENT Revasc. % Stenosis Renal length Medical 76% (40 – 100%) 75% (20 – 100%) 9. 7 cm (6 – 14) P-value 0. 3 9. 7 cm (6 – 20) 0. 5 0. 2 Location of ostial/distal ARVD lesion Left kidney 24% 20% Right kidney 18% 17% Both 50% 57% Missing data 8% 6%

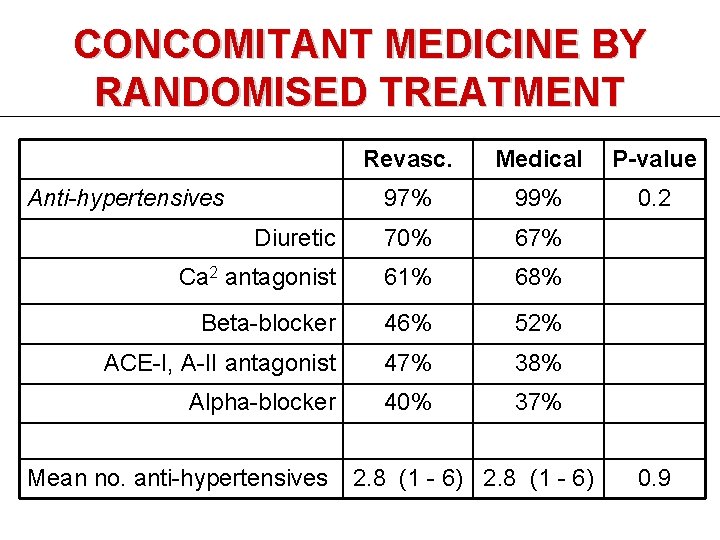
CONCOMITANT MEDICINE BY RANDOMISED TREATMENT Revasc. Medical P-value 97% 99% 0. 2 Diuretic 70% 67% Ca 2 antagonist 61% 68% Beta-blocker 46% 52% ACE-I, A-II antagonist 47% 38% Alpha-blocker 40% 37% Anti-hypertensives Mean no. anti-hypertensives 2. 8 (1 - 6) 0. 9

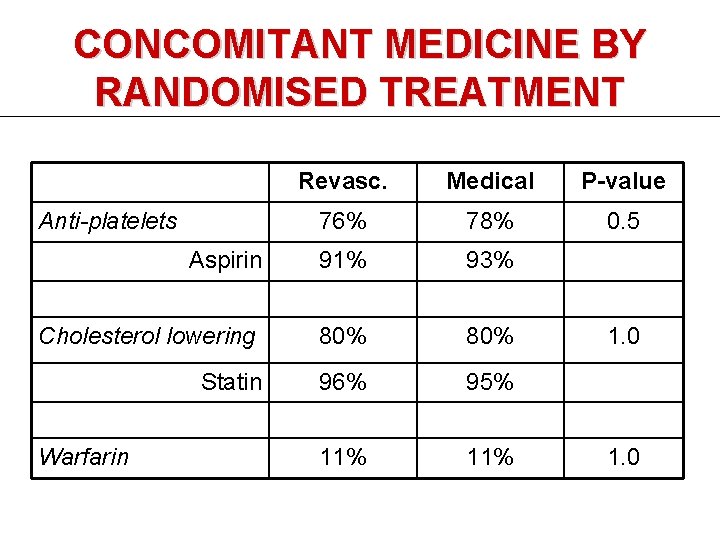
CONCOMITANT MEDICINE BY RANDOMISED TREATMENT Revasc. Medical P-value 76% 78% 0. 5 Aspirin 91% 93% Cholesterol lowering 80% Statin 96% 95% 11% Anti-platelets Warfarin 1. 0

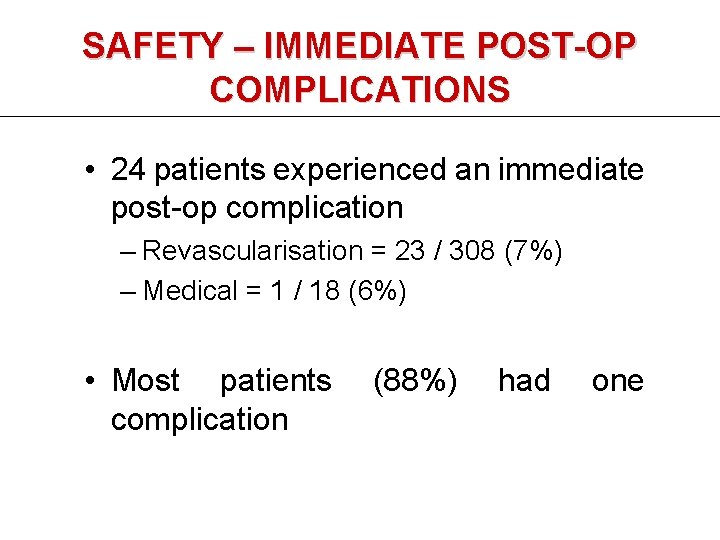
SAFETY – IMMEDIATE POST-OP COMPLICATIONS • 24 patients experienced an immediate post-op complication – Revascularisation = 23 / 308 (7%) – Medical = 1 / 18 (6%) • Most patients complication (88%) had one

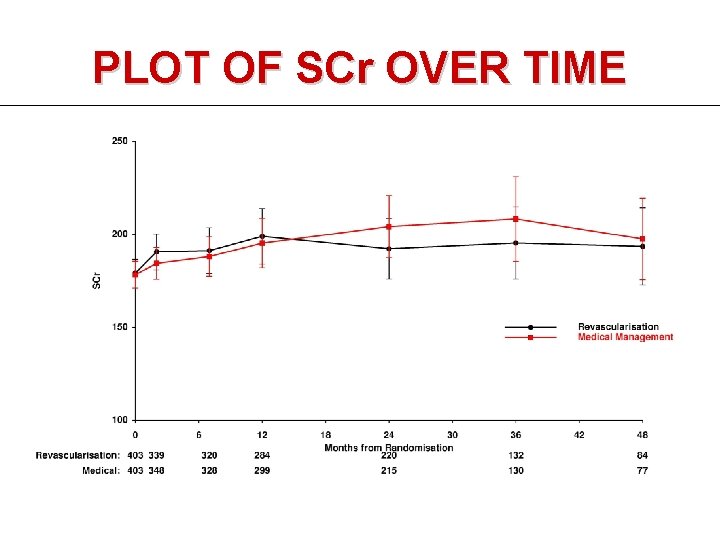
PLOT OF SCr OVER TIME

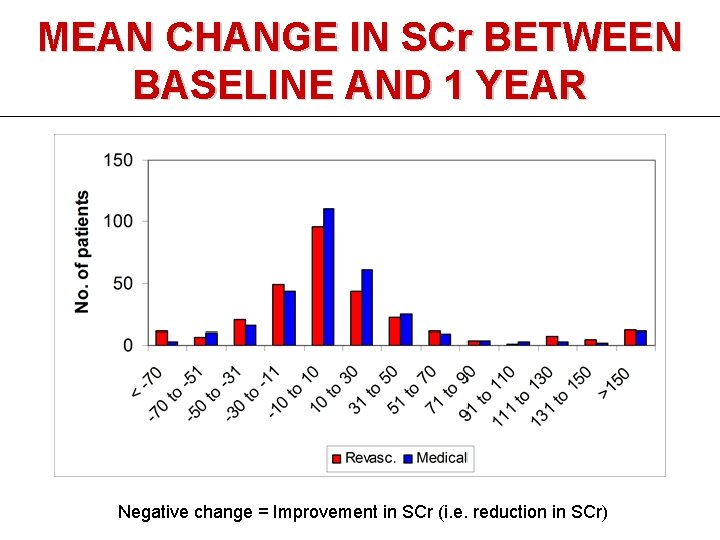
MEAN CHANGE IN SCr BETWEEN BASELINE AND 1 YEAR Negative change = Improvement in SCr (i. e. reduction in SCr)

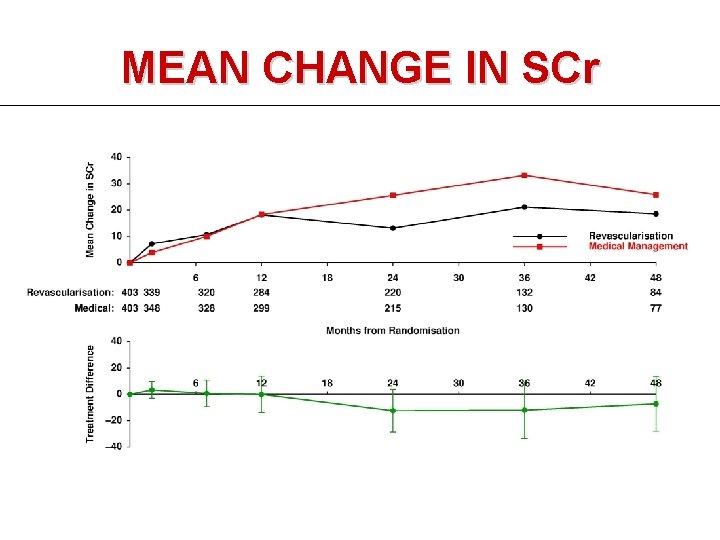
MEAN CHANGE IN SCr

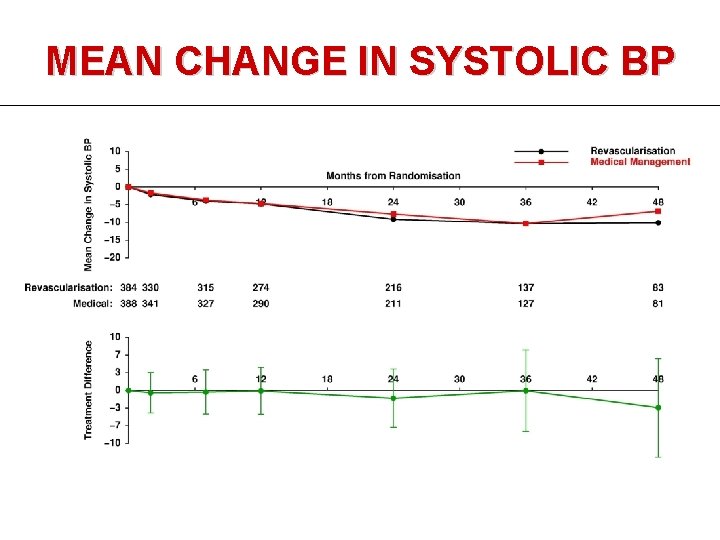
MEAN CHANGE IN SYSTOLIC BP

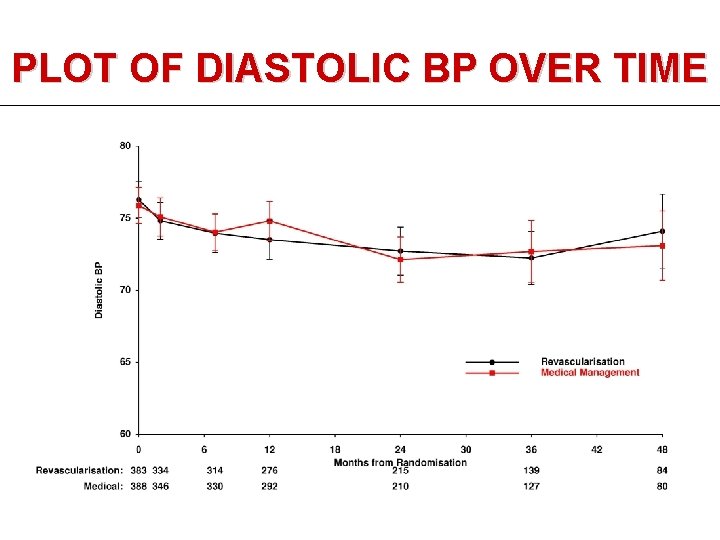
PLOT OF DIASTOLIC BP OVER TIME

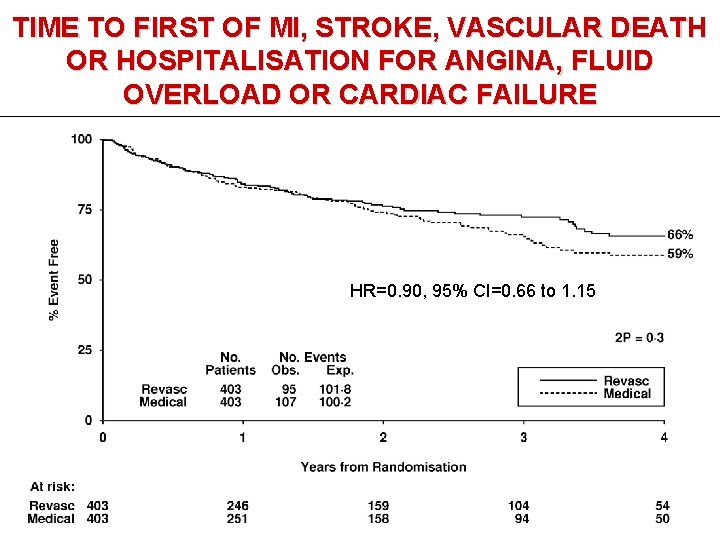
TIME TO FIRST OF MI, STROKE, VASCULAR DEATH OR HOSPITALISATION FOR ANGINA, FLUID OVERLOAD OR CARDIAC FAILURE HR=0. 90, 95% CI=0. 66 to 1. 15

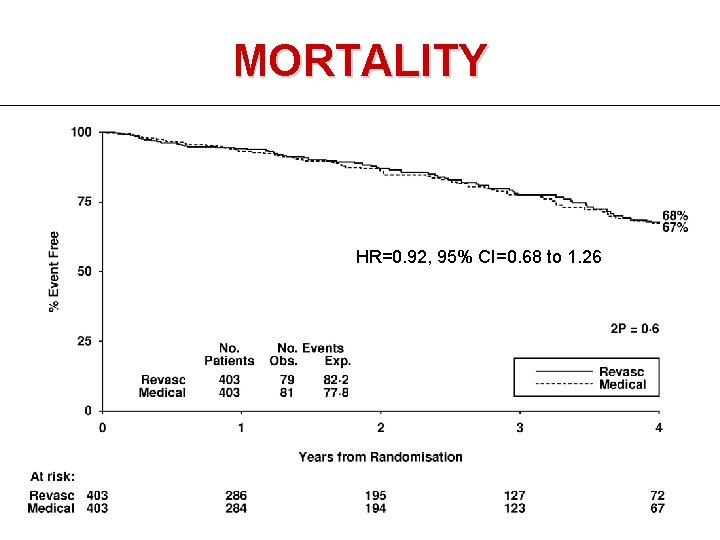
MORTALITY HR=0. 92, 95% CI=0. 68 to 1. 26

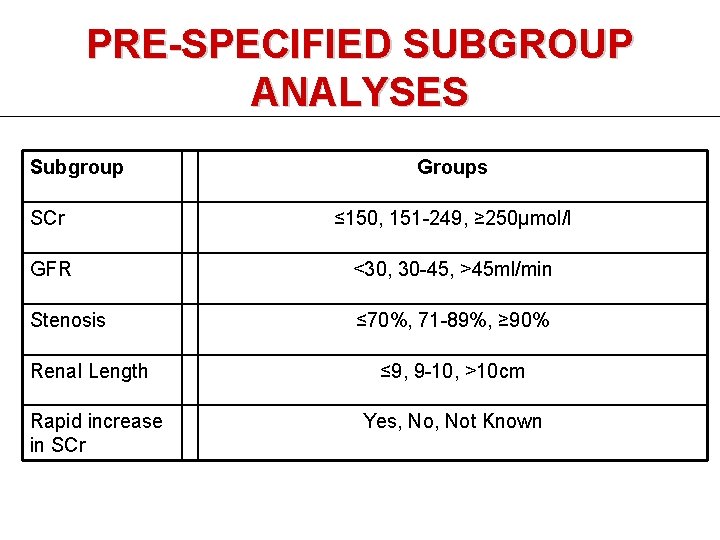
PRE-SPECIFIED SUBGROUP ANALYSES Subgroup Groups SCr ≤ 150, 151 -249, ≥ 250μmol/l GFR <30, 30 -45, >45 ml/min Stenosis ≤ 70%, 71 -89%, ≥ 90% Renal Length Rapid increase in SCr ≤ 9, 9 -10, >10 cm Yes, Not Known

SUMMARY • Currently no evidence of a benefit for revascularisation on renal function in the ARVD patients entered into ASTRAL – those in whom clinicians ‘uncertain’ of whether to revascularise • Also no evidence of differences between the arms for any of the secondary endpoints (i. e. blood pressure, major events) • No evidence of differences in treatment effect across the various subgroups • Longer follow-up is needed • Plan to update meta-analysis published in NDT in 2003 to include ASTRAL and other trials