UCB Immunology Patient Value Unit November 2015 2

UCB Immunology Patient Value Unit November 2015

2 An international executive team Iris Löw-Friedrich Development and Medical Ismail Kola New Medicines Detlef Thielgen Finance Fabrice Enderlin Talent & Company Reputation Emmanuel Caeymaex Immunology Jeff Wren Neurology Jean-Christophe Tellier CEO Anna Richo Legal Mark Mc. Dade Patient Value Operations Bharat Tewarie Strategic Marketing
3

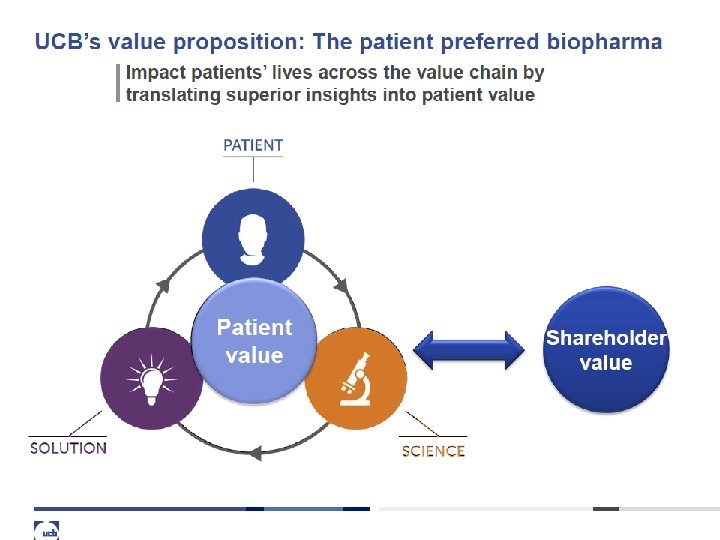
4 What UCB stands for
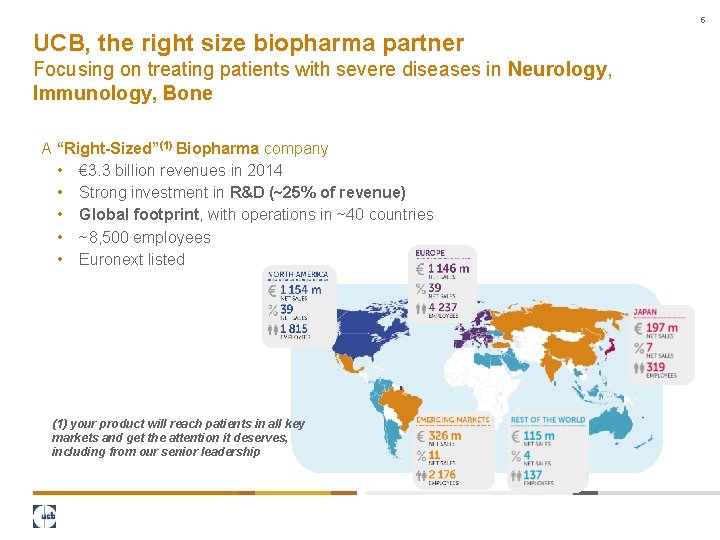
5 UCB, the right size biopharma partner Focusing on treating patients with severe diseases in Neurology, Immunology, Bone A “Right-Sized”(1) Biopharma company • € 3. 3 billion revenues in 2014 • Strong investment in R&D (~25% of revenue) • Global footprint, with operations in ~40 countries • ~8, 500 employees • Euronext listed (1) your product will reach patients in all key markets and get the attention it deserves, including from our senior leadership

6 3 core medicines for patients with severe diseases ■ Rheumatoid arthritis • psoriatic arthritis (PSA) • ankylosing spondylitis (AS) – US • axial spondyloarthritis (Ax. Sp. A) – EU ■ Crohn’s disease ■ Epilepsy adjunctive therapy ■ Parkinson’s disease ■ Restless legs syndrome

7 Other key products Keppra® (levetiracetam) ■ Epilepsy adjunctive therapy (also in monotherapy in Europe) – Sold as E Keppra® in Japan Keppra XR® (levetiracetam – Extended-release tablets) – U. S. only ■ Epilepsy adjunctive therapy Zyrtec® (cetirizine HCl) ■ Seasonal allergic rhinitis Xyzal® (levocetirizine dihydrochloride) ■ Allergies and chronic idiopatic urticaria
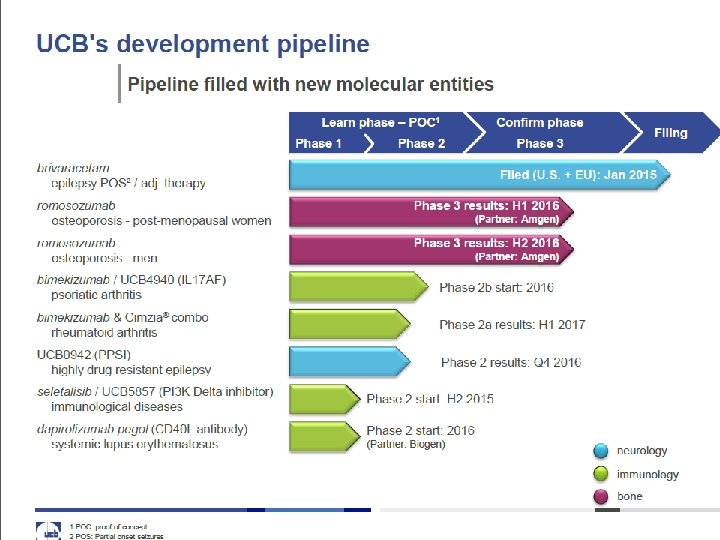
8 Moving forward - seeking additional R&D partnerships Phase 1 Phase 2 Phase 3 romosozumab(1) osteoporosis - postmenopausal women Phase 3 results: H 1 2016 romosozumab(1) osteoporosis – men Phase 3 results: H 2 2016 UCB 0942 (PPSI) highly drug resistant epilepsy Phase 2 results: Q 4 2016 bimekizumab / UCB 4940 (IL 17 AF) psoriatic arthritis Phase 2 a results: H 2 2015 bimekizumab & Cimzia® combo rheumatoid arthritis Phase 2 a results: H 1 2017 UCB 5857 (PI 3 K Delta inhibitor) immune-inflammatory diseases Phase 2 start: H 2 2015 dapirolizumab pegol (CD 40 L antibody) (2) systemic lupus erythematosus Phase 2 start: 2016 neurology UCB 7665 immunological diseases immunology UCB 4144 / VR 942(3) asthma (1) partner: Amgen bone (2) partner: Biogen (3) partner: Vectura

9 Cutting-edge research and science Addressing unmet patient needs and delivering breakthrough medicines Two main Research Centers World-class expertise in selected therapeutic areas and disease mechanisms Connecting science in new ways - chemistry (small, chemically-derived molecules) and biology (large antibody-based molecules), to leverage the potential of these two disciplines ‘Open innovation’ model based on strong partnerships with other biotech companies and prominent universities Biologics pilot plant – Braine-l’Alleud, Belgium Two main Research Centers Biologics R&D centre - Slough, U. K.
10 Where can you be sure an asset will deliver transformational Benefit? Preclinical Cost Increase Decisions Get More Difficult (Emotional/Financial) Phase 1 Phase 2 Phase 3

11 Partnering in Immunology - leveraging respective strengths in research, development and commercial capabilities 2015 2014 2012 Selected partnerships in Neurology Development & Commercial Cimzia in Ps. O Research & Development Peptide combination to address high unmet need in Type 1 Diabetes Research collaboration Exploring the microbiome

Everything we do starts with a simple question: “How will this make a difference to the lives of people with severe diseases? ” Nele, living with lupus 12
- Slides: 12