u CH 4 CH 3 COOH u carbides
유기화학 u 유기화합물은 탄소원소를 기본으로 구성 된다. (예를 들면, 메탄 CH 4, 초산 CH 3 COOH) u 예외: carbides (Ca. S 2 = calcium carbide), COX (carbon monoxide, dioxide), carbonates, carbon disulphide (CS 2), cyanide salts (sodium cyanide) C C

탄화수소의 명명법 u Prefix # of Carbons u Suffix Bond Type Meth-- One -ane Single bond eth-- Two prop-- Three but-- Four pent-- Five hex-- Six hept-- Seven oct-- Eight one carbon=meth and single non-- Nine bonds= ane: methane dec-- Ten (C-C) -ene Double Bond (C==C) -yne Triple bond (C C) * Example: CH 4,
유기화학 족(family) C C C u 18, 000 화합물이 등록 (Chemical Abstract) C u 약 65, 000 종이 활용됨 C C H O

유기화학 족(family) - Alcohols: R-O-H - Alkanes: Cn. H 2 n+2 - Ethers: R-O-R - Alkenes: Cn. H 2 n - Aldehydes: R-CH=O - Alkynes: Cn. H 2 n-2 - Ketones: R-CR=O - Amines: R-NH 2, R 2 NH, R 3 N - Esters: R-COR=O - Nitriles: R-CN - Organic acids: R-COH=O – Nitro: R-NO 2 - Peroxides: R-O-O-R - Alkyl halides: R-X

무기화학물질 명명법 u Salts Hypo--ite --ate per--ate u Acids Hypo--ous --ic per--ic Na. Cl. O 2 Na. Cl. O 3 Na. Cl. O 4 sodium chlorite sodium chlorate sodium hypochlorite perchlorate Na 2 SO 3 Na 2 SO 4 sodium sulfite sodium sulfate Na 2 PO 3 Na 3 PO 4 sodium phosphite sodium phosphate Na. NO 2 Na. NO 3 sodium nitrite sodium nitrate �Metal with non metal: metal --ide

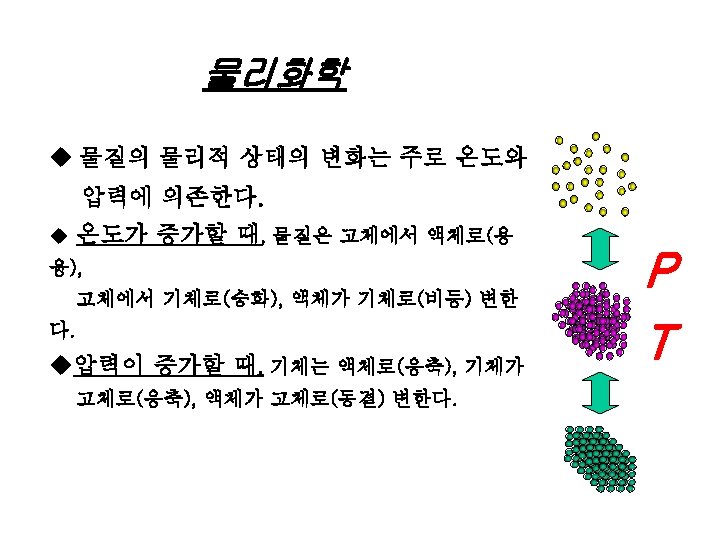
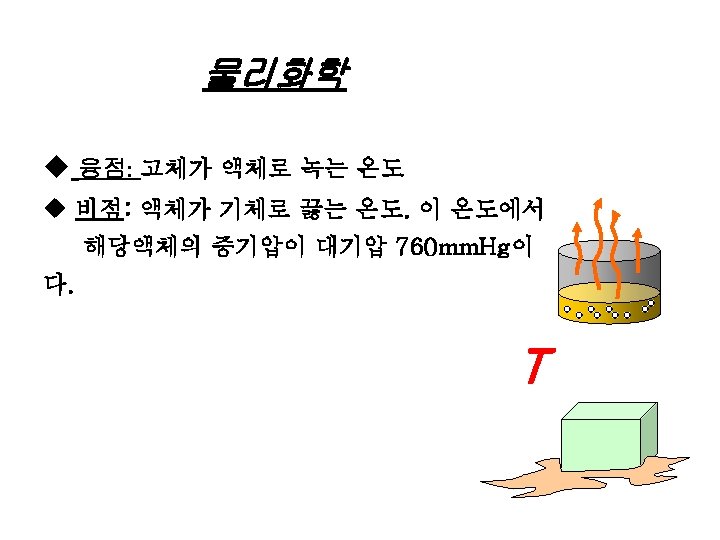

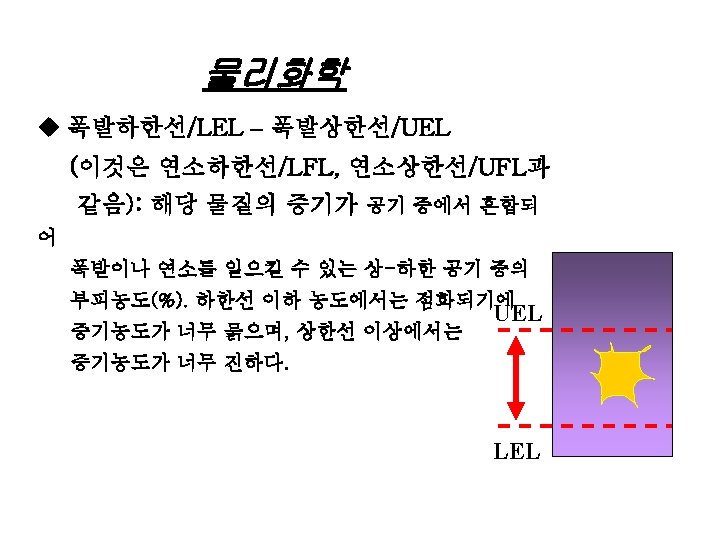
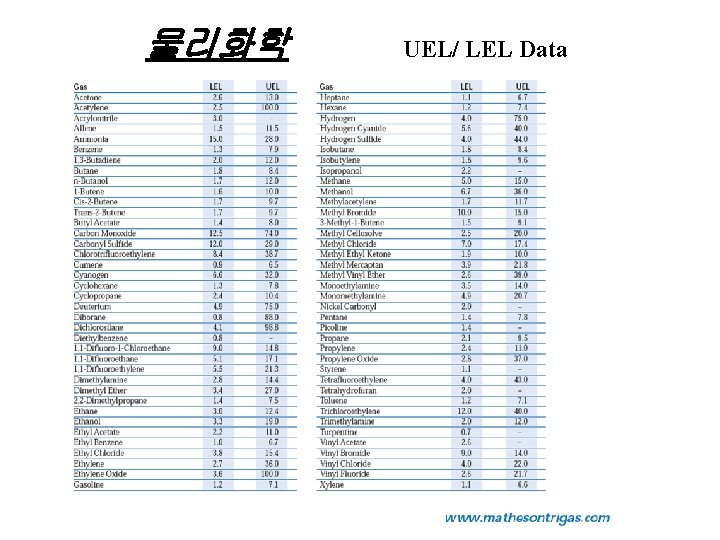
물리화학 UEL/ LEL Data






공기보다 가벼운 기체 (기타 다른 기체는 모두 공기보다 무겁다) u acetylene (C 2 H 2) u hydrogen (H 2) u ammonia (NH 3) u hydrogen cyanide (HCN) u carbon monoxide (CO) u hydrogen fluoride (HF) u deuterium (D 2) u methane (CH 4) u diborane (B 2 H 6) u nitrogen (N 2) u ethylene (C 2 H 4) u helium (He)

- Slides: 20