Types of Reactions We can abbreviate Reaction to

Types of Reactions We can abbreviate Reaction to Rxn

Reactants and Products • Reactants are the chemicals that react – they are on the left side of the equation • Products are the chemicals that are produced – they are on the right side of the equation Reactant + Reactant => Product + Product
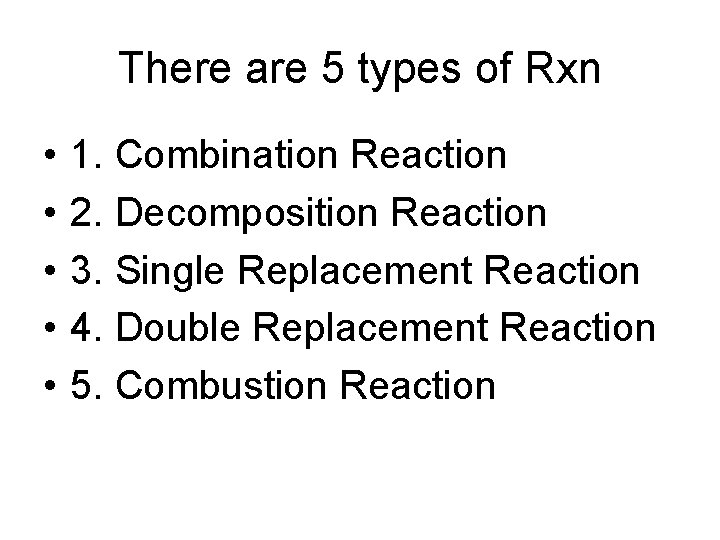
There are 5 types of Rxn • • • 1. Combination Reaction 2. Decomposition Reaction 3. Single Replacement Reaction 4. Double Replacement Reaction 5. Combustion Reaction

Combination Reaction A + B => AB • Two or more substances react and combine to form a single substance • Also called a Synthesis Reaction 2 H 2 + O 2 => 2 H 2 O


Decomposition Reaction AB => A + B • A single compound reacts to form two or more simpler products. 2 NI 3 => N 2 + 3 I 2

Boom!!!!
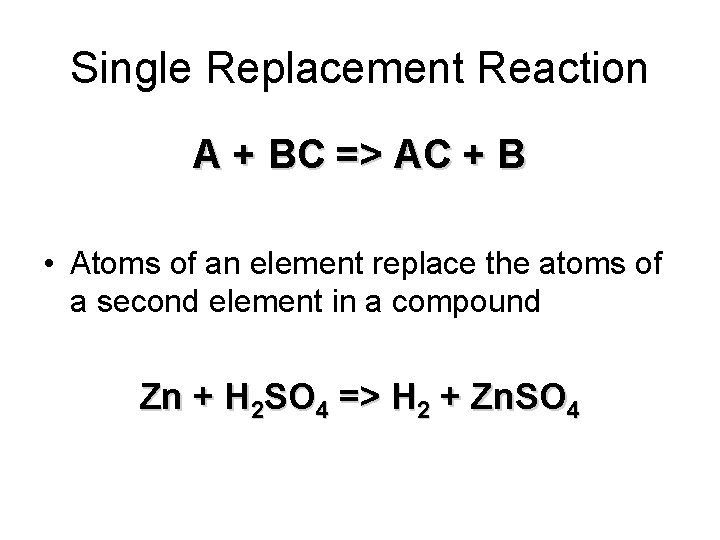
Single Replacement Reaction A + BC => AC + B • Atoms of an element replace the atoms of a second element in a compound Zn + H 2 SO 4 => H 2 + Zn. SO 4


• The higher it is the more reactive. • Metals from Li to Na will replace H from acids and water. • Metals from Mg to Pb will replace H from acids only.

Double Replacement Reaction AB + CD => AD + CB • Positive ions are exchanged between two compounds. Na. OH + HCl => Na. Cl + H 2 O

Calcium carbonate (marble) and Sulfurous Acid (acid rain)

Combustion Reaction Cx. Hy + O 2 => CO 2 + H 2 O • Oxygen reacts with another substance – producing energy (heat and light) – and usually carbon dioxide and water. CH 4 + O 2 => CO 2 + H 2 O 2 CH 3 OH + 3 O 2 => 2 CO 2 + 4 H 2 O




Putting out a Fire (Combustion) • If you need a Fuel and Oxygen to have combustion – how many ways are there to stop combustion. • 1. “Stop” the Fuel • 2. “Stop” the Oxygen • 3. Reduce the energy so it cannot support the initiating energy. (ignition point) – Read Fahrenheit 451 by Ray Bradbury


Disclaimer Aloha I put together these power points for use in my science classes. You may use them in your classes. Some images are public domain, some are used under the fair-use provisions of the copyright law, some are mine. Copyright is retained by the owners! Ted Brattstrom
- Slides: 19