Types of Reactions Single Displacement Double displacement Synthesis

Types of Reactions

• Single Displacement • Double displacement • Synthesis • Decomposition • Combustion

Single Displacement • A Metal or hydrogen replaces the positive portion of a compound of a nonmetal does it for the negative portion

Double Displacement • Two compounds exchange negative portions • Change of phase a must

Synthesis • Smaller molecules or elements combine to make larger molecules

Decomposition • Larger molecules break down to form smaller molecules or elements

Combustion • A carbon compound is combined rapidly with oxygen producing carbon dioxide & water
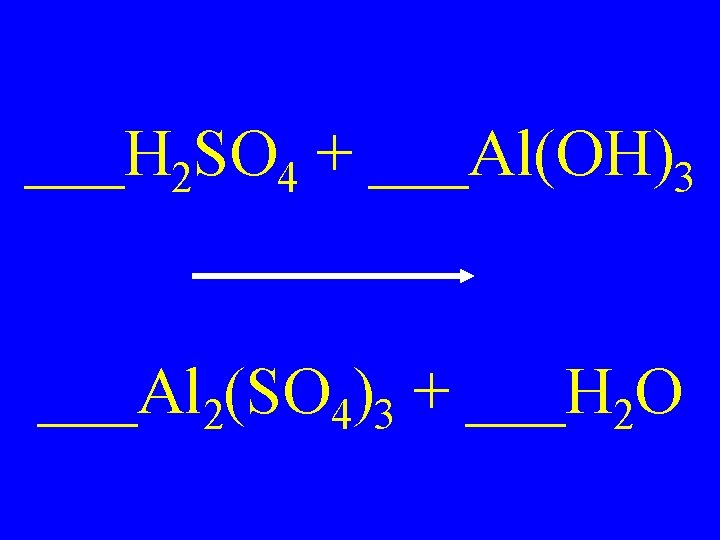
___H 2 SO 4 + ___Al(OH)3 ___Al 2(SO 4)3 + ___H 2 O
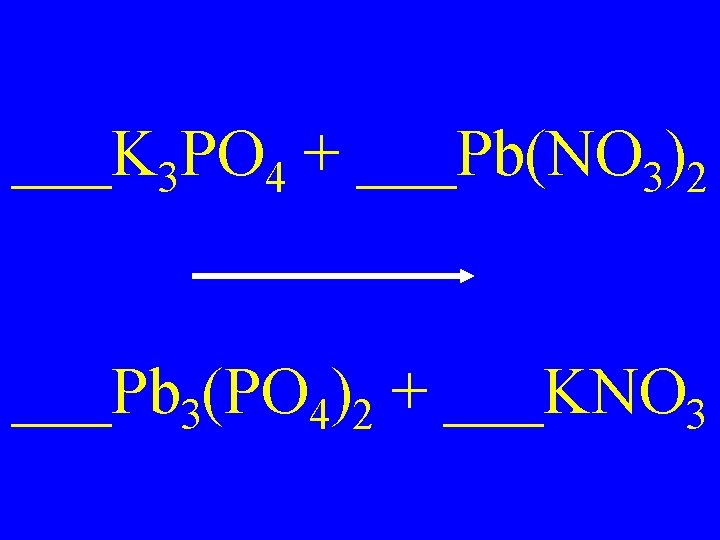
___K 3 PO 4 + ___Pb(NO 3)2 ___Pb 3(PO 4)2 + ___KNO 3
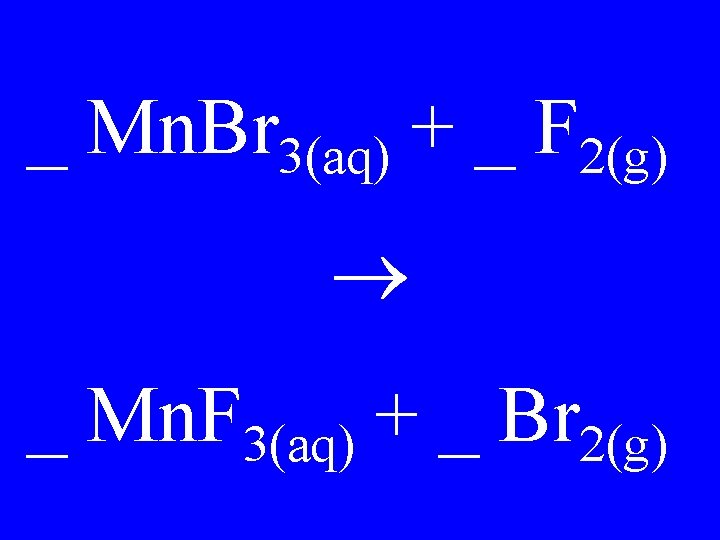
_ Mn. Br 3(aq) + _ F 2(g) _ Mn. F 3(aq) + _ Br 2(g)
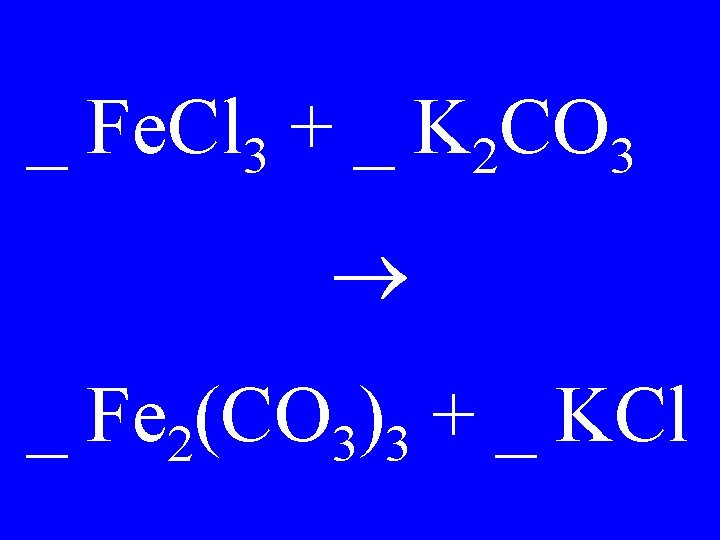
_ Fe. Cl 3 + _ K 2 CO 3 _ Fe 2(CO 3)3 + _ KCl
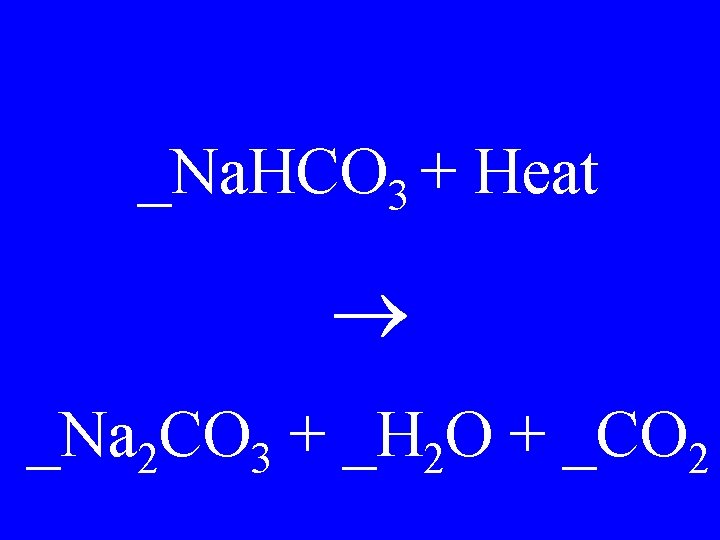
_Na. HCO 3 + Heat _Na 2 CO 3 + _H 2 O + _CO 2
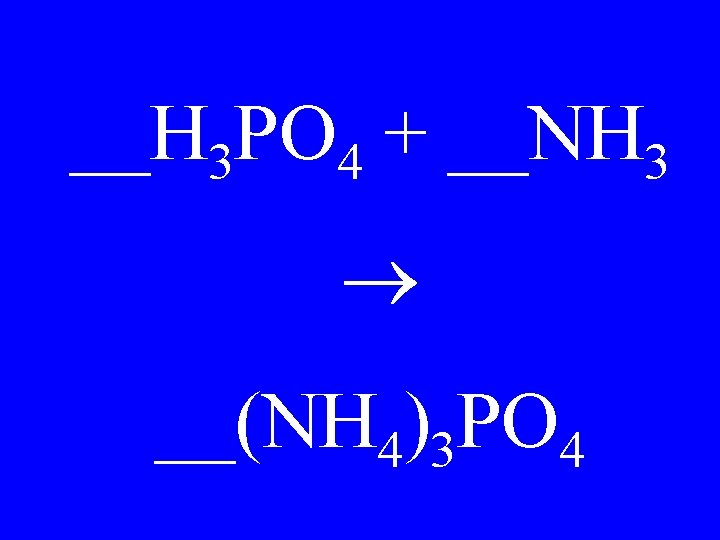
__H 3 PO 4 + __NH 3 __(NH 4)3 PO 4
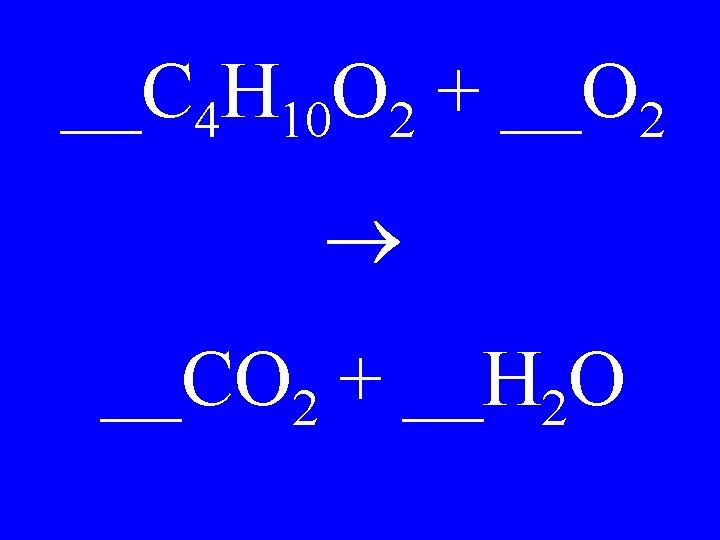
__C 4 H 10 O 2 + __O 2 __CO 2 + __H 2 O

Make the following conversions • 50. 0 g Ca. CO 3 to molecules • 5. 6 L SO 2 to mass • 4. 0 mg Fe 2 O 3 to atoms
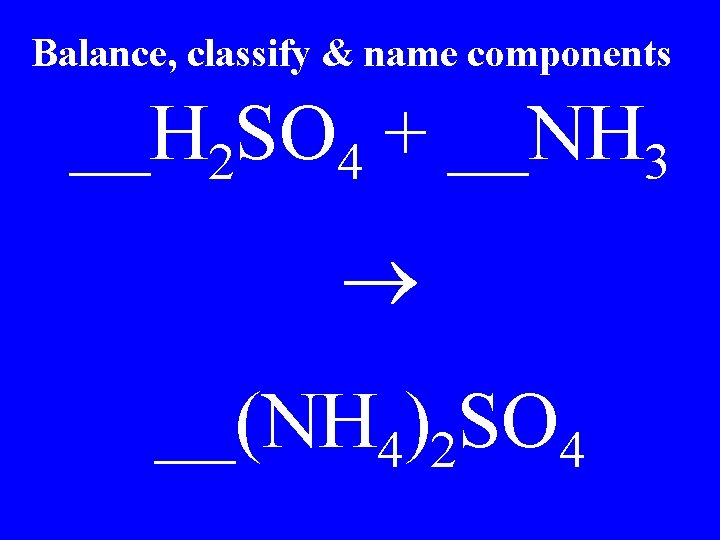
Balance, classify & name components __H 2 SO 4 + __NH 3 __(NH 4)2 SO 4

Balance, classify & name components _K 2 SO 4 +_Fe. Cl 3 _ Fe 2(SO 4)3 + _ KCl
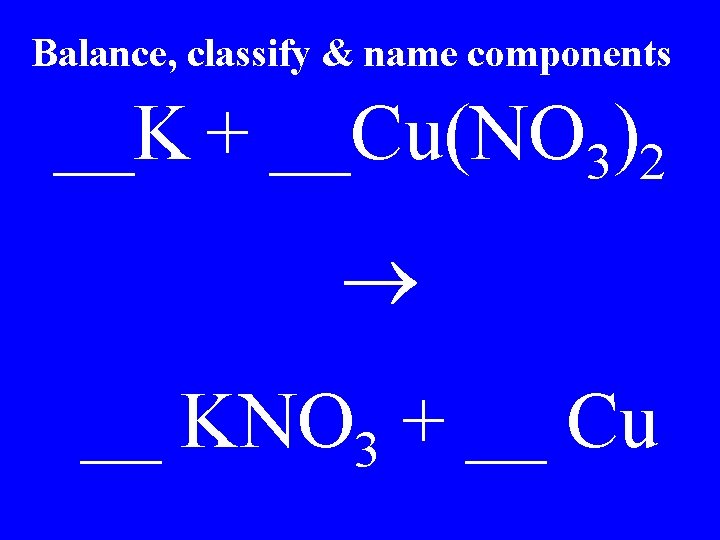
Balance, classify & name components __K + __Cu(NO 3)2 __ KNO 3 + __ Cu
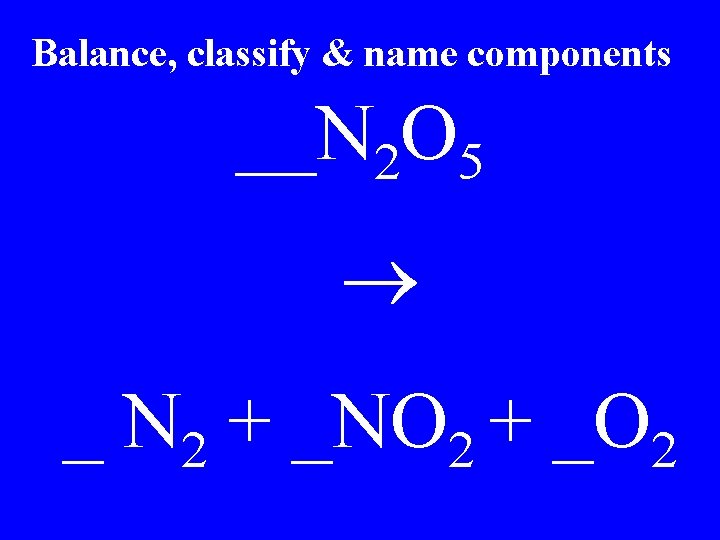
Balance, classify & name components __N 2 O 5 _ N 2 + _NO 2 + _O 2
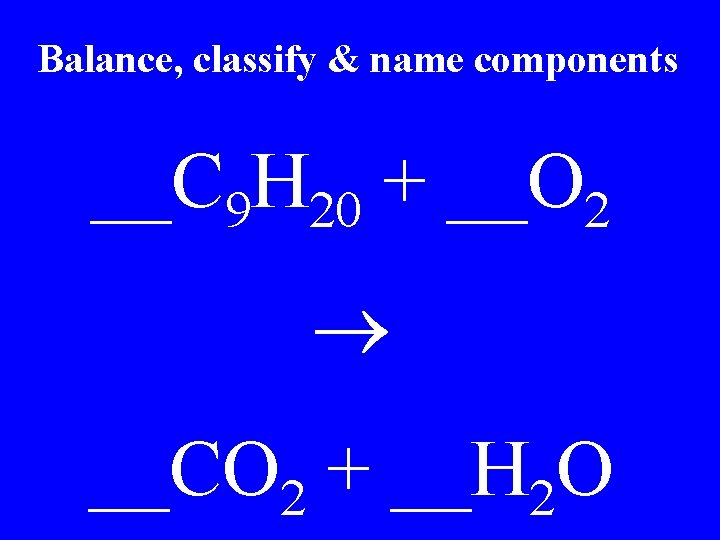
Balance, classify & name components __C 9 H 20 + __O 2 __CO 2 + __H 2 O
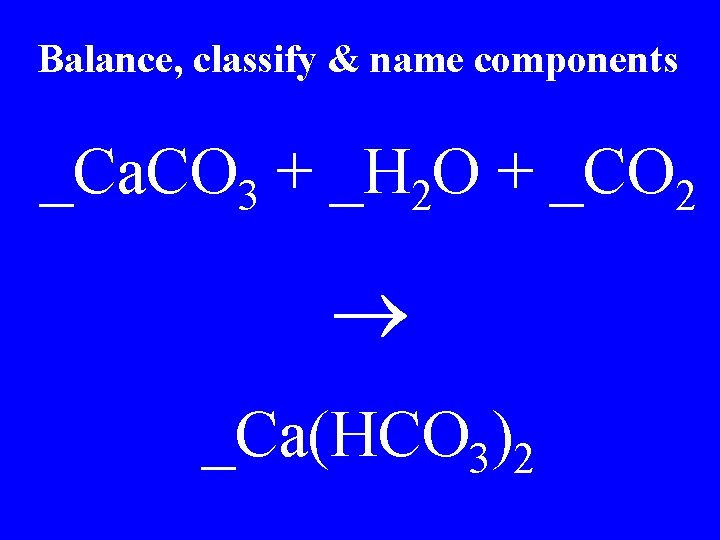
Balance, classify & name components _Ca. CO 3 + _H 2 O + _CO 2 _Ca(HCO 3)2
- Slides: 21