Types of Reactions Classifying Reactions l How can




















- Slides: 22

Types of Reactions

Classifying Reactions l. How can you predict the products if you are given the reactants? l. Must know the types of reactions

Types of Reactions l. Addition (Synthesis) l. Decomposition l. Single Displacement (Replacement) l. Double Displacement (Replacement) l. Combustion l. Acid Base

Addition (Synthesis) l. Two or more reactants combine to form a new product l. A + B AB l. Example: Rust formation: Fe(s) + O 2(g) Fe 2 O 3

Decomposition l. Compounds are broken down into two or more smaller compounds l. AB A + B l. Si. Cl 4 Si + Cl 2

Single Displacement l. One element replaces another in a compound l. AX + B BX + A l. Ca + HCl Ca. Cl 2 + H 2

Double Displacement l. Ions switch places; two elements switch places l. AB + CD AD + CB l. Ag. NO 3 + Na. Cl Ag. Cl + Na. NO 3

Combustion l. Carbon compound combines with oxygen to release carbon dioxide and water and energy l. Carbon cmpd + O 2 CO 2 + H 20 + energy! l. C 2 H 6 + O 2 CO 2 + H 20

Acid Base Reactions l. The reactants will always be an acid and a base producing 2 specific products; Water and a Salt. l. Acids always start with an Hl. Bases always end in an –OH l. Water is H 20 l. HCl + Na. OH H 20 + Na. Cl

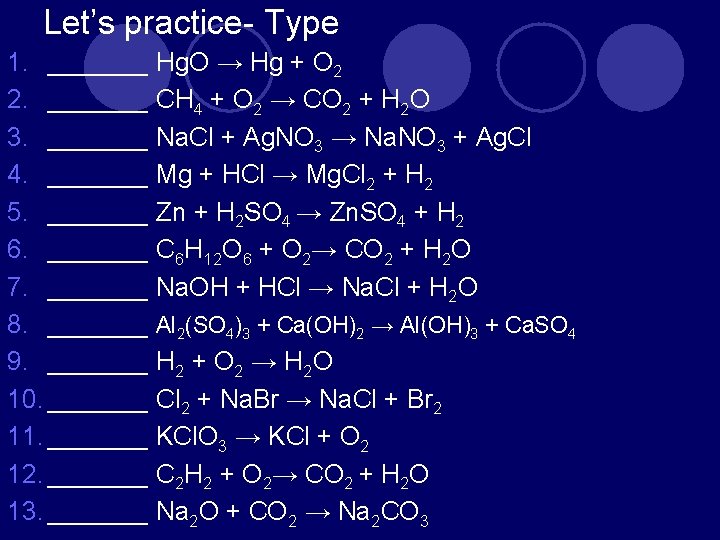
Let’s practice- Type 1. _______ Hg. O → Hg + O 2 2. _______ CH 4 + O 2 → CO 2 + H 2 O 3. _______ Na. Cl + Ag. NO 3 → Na. NO 3 + Ag. Cl 4. _______ Mg + HCl → Mg. Cl 2 + H 2 5. _______ Zn + H 2 SO 4 → Zn. SO 4 + H 2 6. _______ C 6 H 12 O 6 + O 2→ CO 2 + H 2 O 7. _______ Na. OH + HCl → Na. Cl + H 2 O 8. _______ Al 2(SO 4)3 + Ca(OH)2 → Al(OH)3 + Ca. SO 4 9. _______ H 2 + O 2 → H 2 O 10. _______ Cl 2 + Na. Br → Na. Cl + Br 2 11. _______ KCl. O 3 → KCl + O 2 12. _______ C 2 H 2 + O 2→ CO 2 + H 2 O 13. _______ Na 2 O + CO 2 → Na 2 CO 3

Categorizing Chemical Reactions l All chemical reactions involve something moving, so we can categorize the types of reactions we just learned to help us predict the products in a reaction! ¡Moving electrons = synthesis, decomposition, single displacement, combustion. ¡Moving ions = double displacement ¡Moving protons = acid/base reaction (we’ll get into this in a later unit)

Moving Electrons l In the following reactions, one or more electrons move from one atom to another l This allows the atoms to act differently than in their original compounds, and we form new substances. l. Ex: ¡Mg + Br 2 Mg. Br 2 ¡Na. Cl Na + Cl 2 ¡Zn + Cu. SO 4 Cu + Zn. SO 4

l. In the first example, think about the Lewis Structures that we’ve drawn. l. The Magnesium atom loses electrons and the Bromine atom gains electrons. l. There is a transfer of electrons to create the ionic bond.

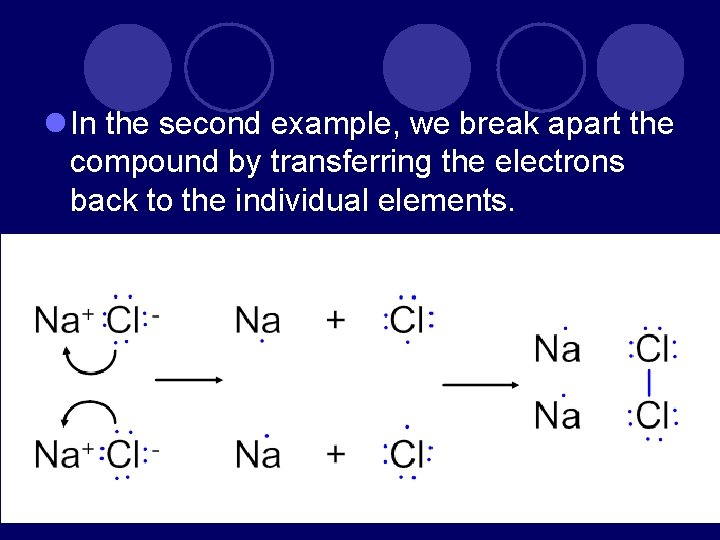
l In the second example, we break apart the compound by transferring the electrons back to the individual elements.

l. Zn + Cu. SO 4 Cu + Zn. SO 4 l Remember Cu. SO 4 is an ionic compound, so it really is made of Cu+2 and SO 4 -2 ions. l Zn is starting off neutral in the reactants. l During this reaction, Zn transfers its electrons to Cu+2 ion. l Zinc loses electrons and becomes a Zn+2 ion and Cu gains electrons to become neutral. l Now that Zn+2 has a charge, it will form the ionic compound with SO 4 -2 because the opposite charges ions will attract.

l In this case, did the sulfate ion see an change of electrons? l No, so these electrons were not involved in the reaction and were not shown. l This is an example of something called a “Spectator Ion” and we’ll talk about it later.

Reduction and Oxidation l. In these reactions, one atom or element is reduced and the other is oxidized. So you’ll hear these called Redox. l. A substance that is reduced has gained electrons and the charge is reduced. (more negative) l. A substance that is oxidized has lost electrons and the charge is increased. (more positive)

2 Fun Ways to Remember OIL RIG l Oxidation l Is l Losing elections l Reduction l Is l Gaining electrons LEO the lion says GER l Losing l Electrons is l Oxidation l Gaining l Electrons is l Reduction

And now, the tricky part… l. A substance that has been oxidized is called a reducing AGENT because it caused another element to take their electrons. l. A substance that has been reduced is called an oxidizing AGENT because it caused another element to give up its own electrons

l Br starts with 0 charge and ends with -1 ¡The charge has gone down, because Br has gained electrons. l Mg starts with 0 charge and ends with +2 ¡The charge has gone up, because Mg has lost electrons. l This means Br has been ______ and is the _______ agent. reduced oxidizing l This means Mg has been ______ and is the _______ agent. oxidized reducing

l Na starts with a +1 charge, ends with 0 charge ¡Charge has gone down because it gained electrons l Cl starts with a -1 charge, ends with 0 charge ¡Charge has gone up because it lost electrons l This means Na has been _______ and is the ______ agent. reduced oxidizing l This means Cl has been ______ and is the _______ agent. oxidized reducing

l Zn started with 0 charge, ended with +2 ¡Charge has gone up because it lost electrons l Cu started with +2 charge, ended with 0 ¡Charge has gone down because it gained electrons l SO 4 started and ended with -2, no electrons changed here l Zn has been oxidized and is the reducing agent l Cu has been reduced and is the oxidizing agent l SO 4 wasn’t really involved in the reaction. (Spectator)