Types of Nuclear Decay Topic 7 2 The














- Slides: 33

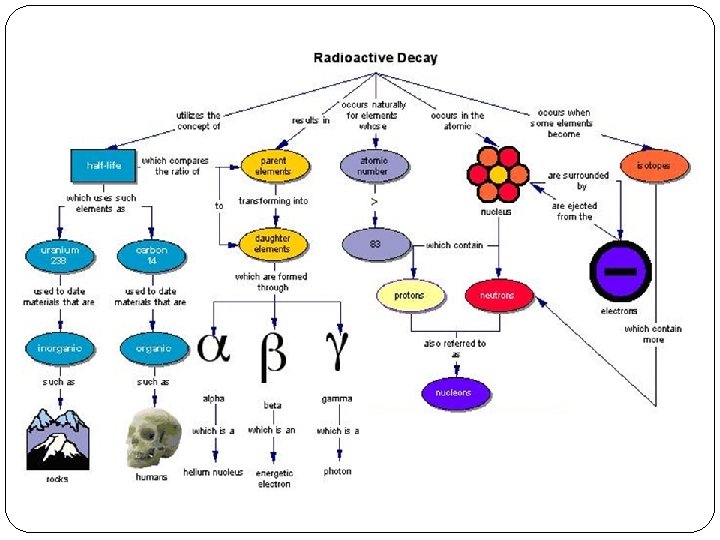
Types of Nuclear Decay Topic 7. 2 The ABC’s of Radioactivity

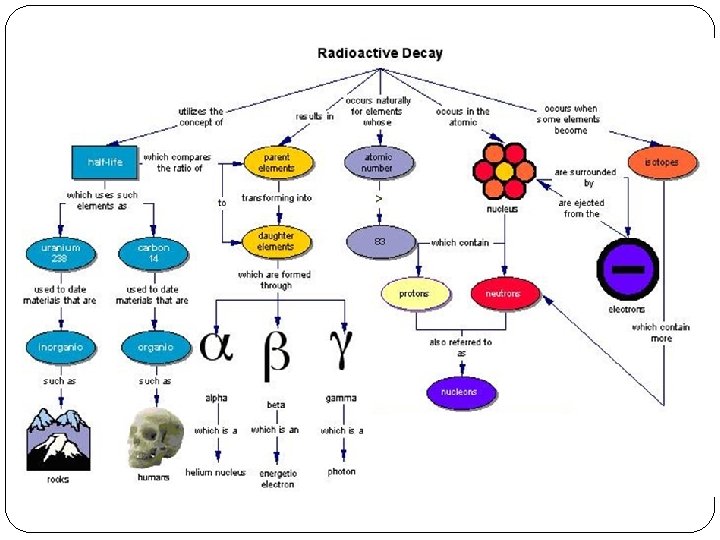
Nuclear Decay Review �Describe the nuclear decay process. �Name the 2 key items involved �What is the relationship between the rate of decay and the number nuclei in the sample? �Describe the term half-life �Why is the nuclear activity is more meaningful than the number of particles in the nuclei?


During the decay process… �the nucleus is ‘broken up’ into smaller pieces �How does this happen? �Where does the initial energy come from? �How would the masses compare before and after a decay? �Are there certain types of particles that come out of a decay or is it random?

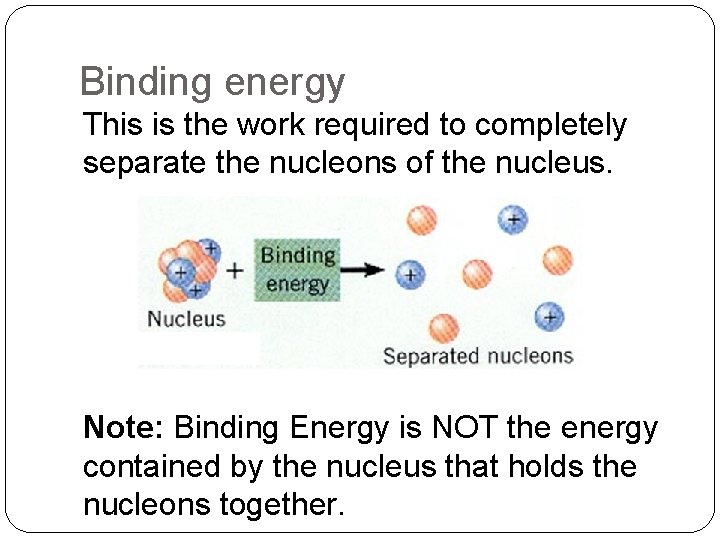
Binding energy This is the work required to completely separate the nucleons of the nucleus. Note: Binding Energy is NOT the energy contained by the nucleus that holds the nucleons together.

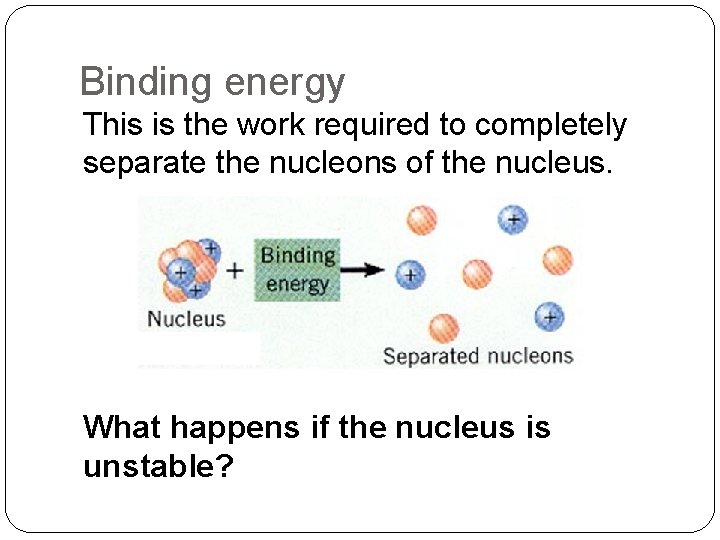
Binding energy This is the work required to completely separate the nucleons of the nucleus. What happens if the nucleus is unstable?

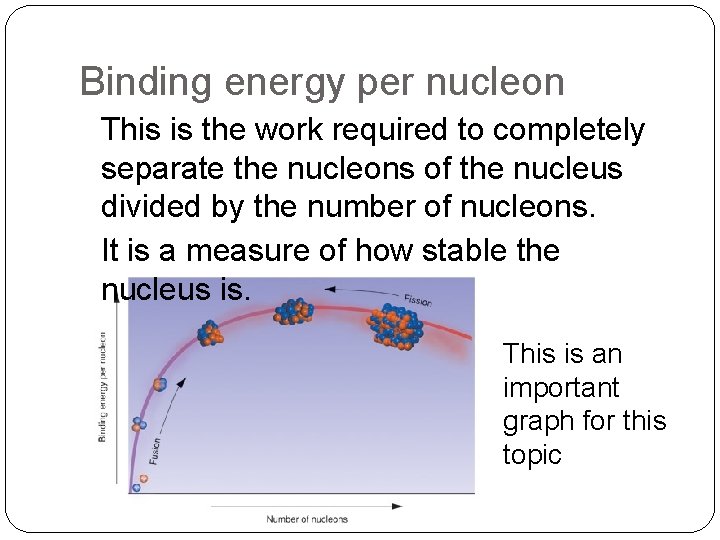
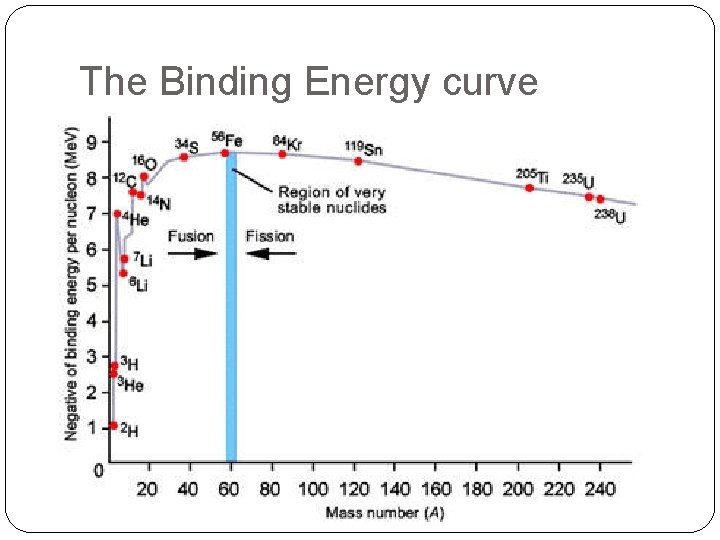
Binding energy per nucleon This is the work required to completely separate the nucleons of the nucleus divided by the number of nucleons. It is a measure of how stable the nucleus is. This is an important graph for this topic

The Binding Energy curve

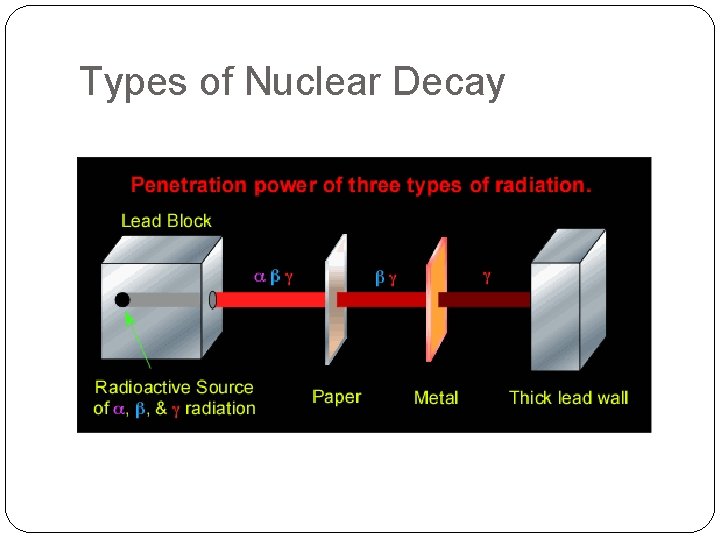
Types of Nuclear Decay They are defined by the 3 main types of particles that can be ejected from an unstable nuclei.

Types of Nuclear Decay What is the main reason that make unstable isotopes ‘decay’?

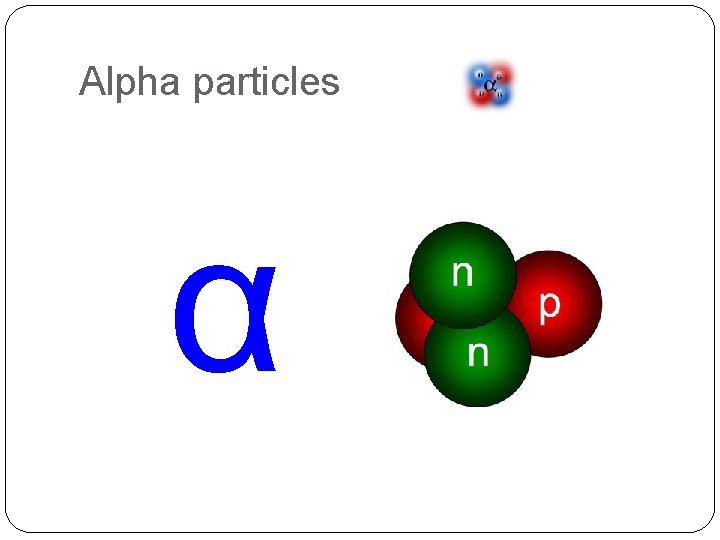
Alpha particles α

Alpha particles � 2 protons and 2 neutrons joined together �The same as the nucleus of a helium atom �Stopped by paper or a few cm of air �Highly ionising �Deflected by electric and strong 2+ 4 magnetic fields 2 He

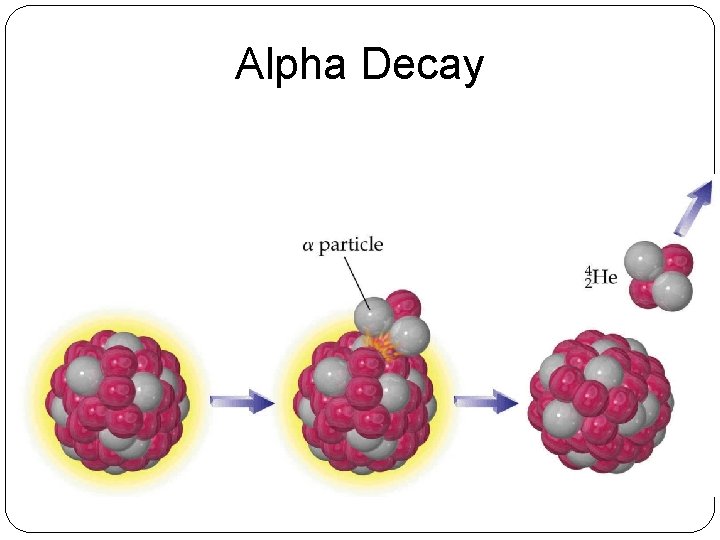
Alpha Decay

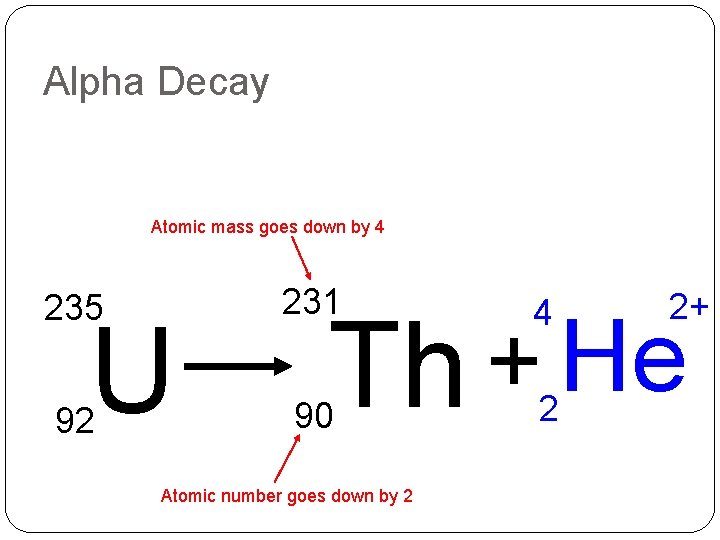
Alpha Decay Atomic mass goes down by 4 235 231 4 92 90 2 U 2+ He + Th Atomic number goes down by 2

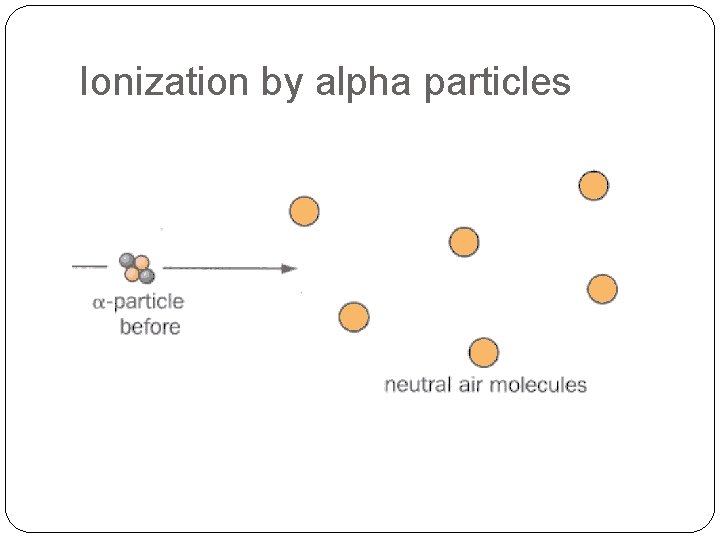
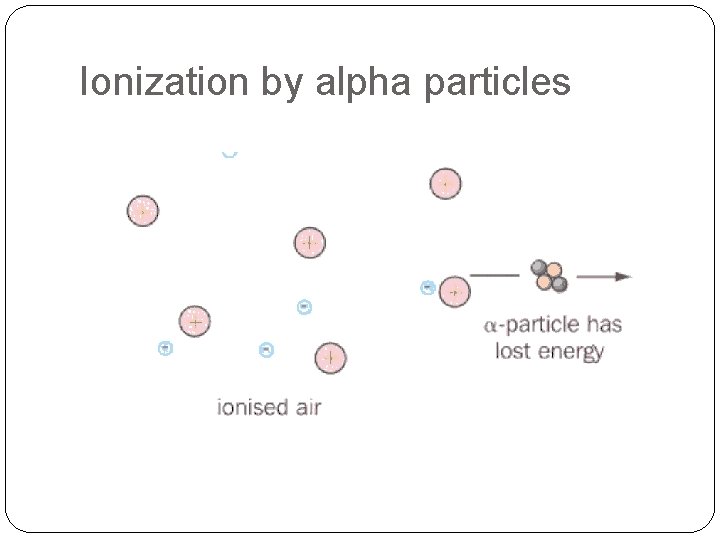
Ionization by alpha particles

Ionization by alpha particles

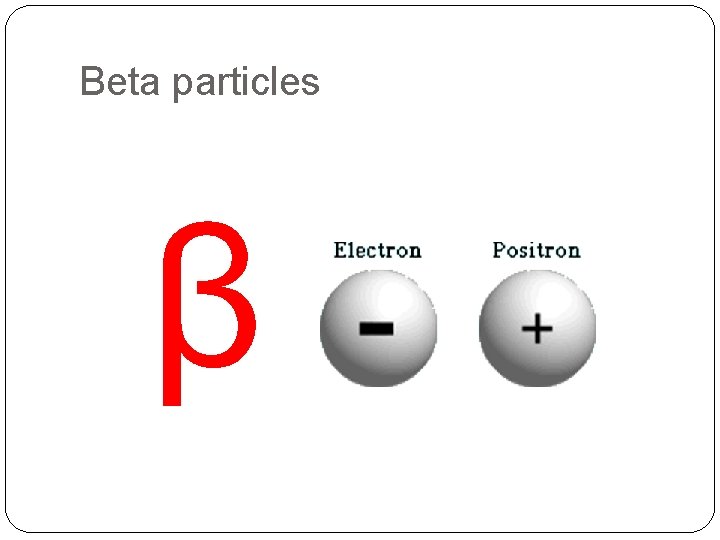
Beta particles β

Beta particles �Fast moving electrons �Effectively massless �Stopped by about 3 mm of aluminium �Weakly ionising �Deflected by electric and magnetic fields e 0 -1

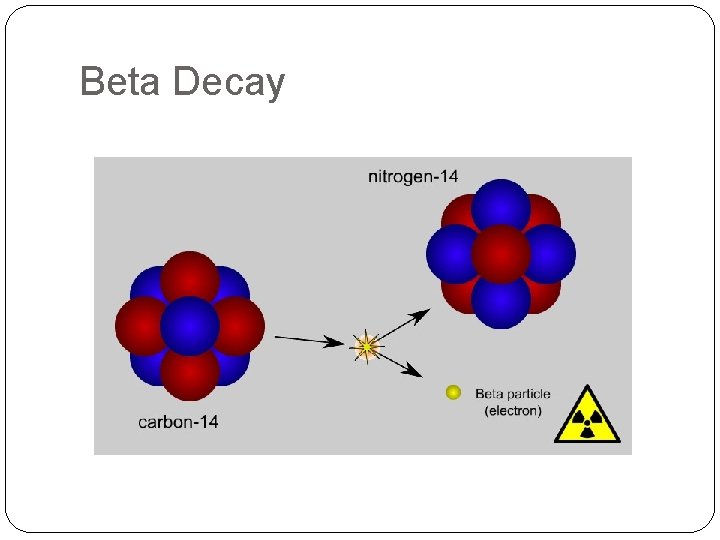
Beta Decay

Beta Decay �In the nucleus a neutron changes into an electron (the beta particle which is ejected) and a proton (which stays in the nucleus) �During beta decay the mass number stays the same but the proton number goes up by 1. 231 Remember the 231 electron comes 0 from the nucleus! 90 Th Pa + e 91 -1

Beta Decay – Mono energetic? �Unlike alpha particles, beta particles can be emitted with different energies �How do we conserve the energy with each reaction? antimatter 231 Th 90 antineutrino Pa + e + ע e 231 91 0 -1 0 0

Antiparticles �Produced naturally symmetry in the universe! �Particle + Antiparticle = ENERGY (photons) antimatter �How did matter dominate in our universe? 231 Th 90 positron neutrino Pa + e + ע e 231 91 0 +1 0 0

Gamma rays

Gamma rays �High frequency electromagnetic radiation �Stopped by several cm of lead �Very weakly ionising �NOT affected by electric or magnetic fields

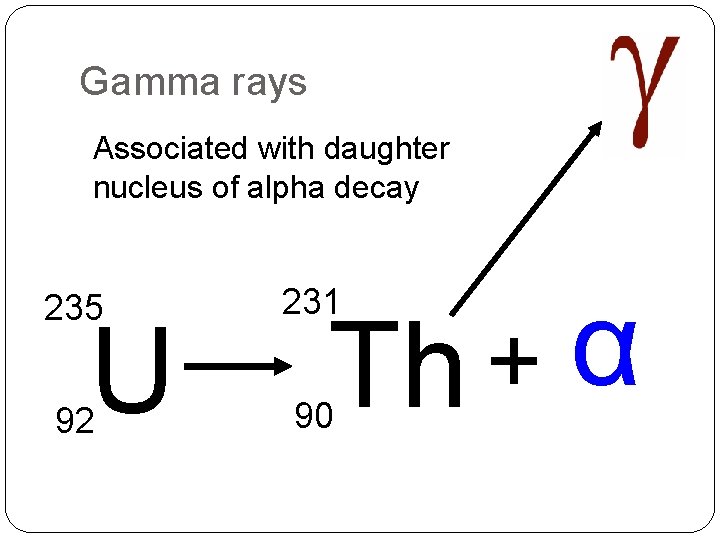
Gamma rays Associated with daughter nucleus of alpha decay 235 231 92 90 U Th + α


Types of Nuclear Decay

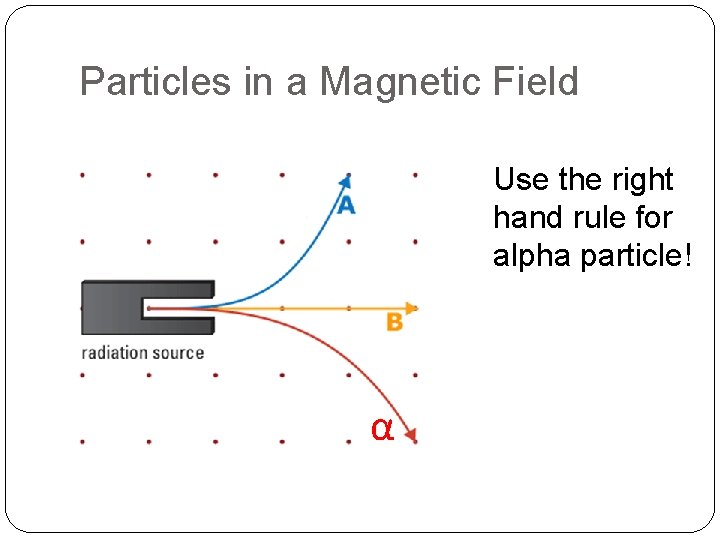
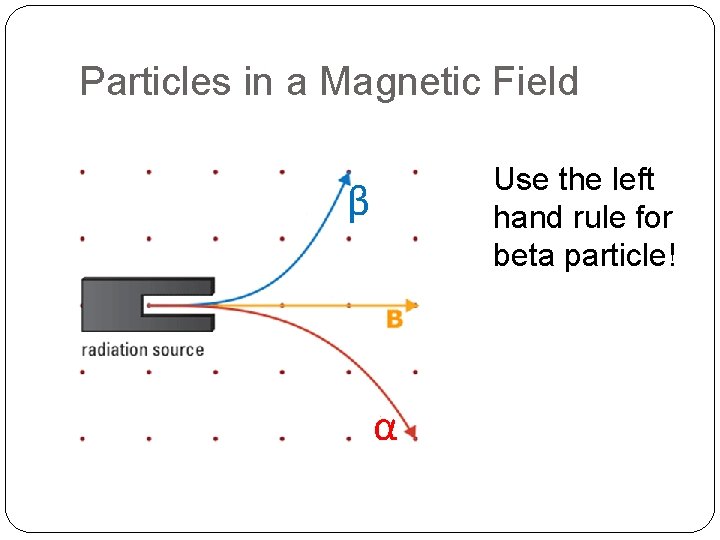
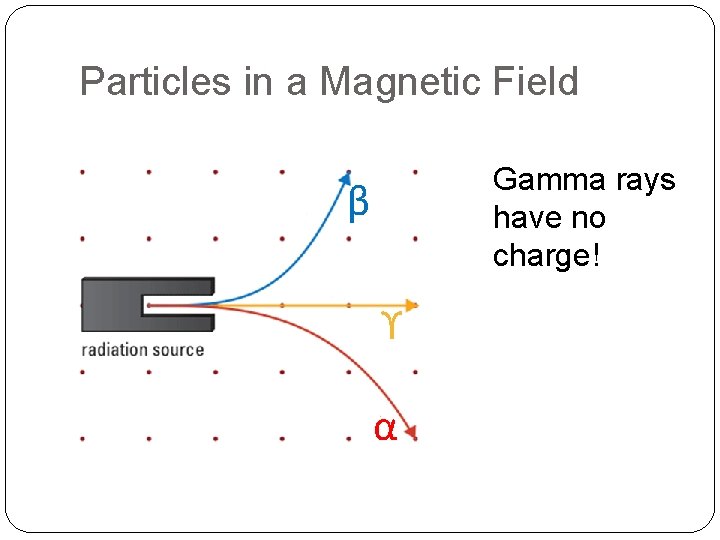
Particles in a Magnetic Field If the magnetic field is coming out of the page, name three types of radiation that’s present in this diagram

Particles in a Magnetic Field Use the right hand rule for alpha particle! α

Particles in a Magnetic Field Use the left hand rule for beta particle! β α

Particles in a Magnetic Field Gamma rays have no charge! β ϒ α

Cloud Chamber �Utilizes ionization properties of alpha & beta particles �Vapour condenses around small particles of ‘dust’ cloud �Similar to vapour trails left by airplanes

Ph. ET Simulations �Alpha Decay �Beta Decay