Types of Mixtures Solutions Suspensions Colloids Solutions are

















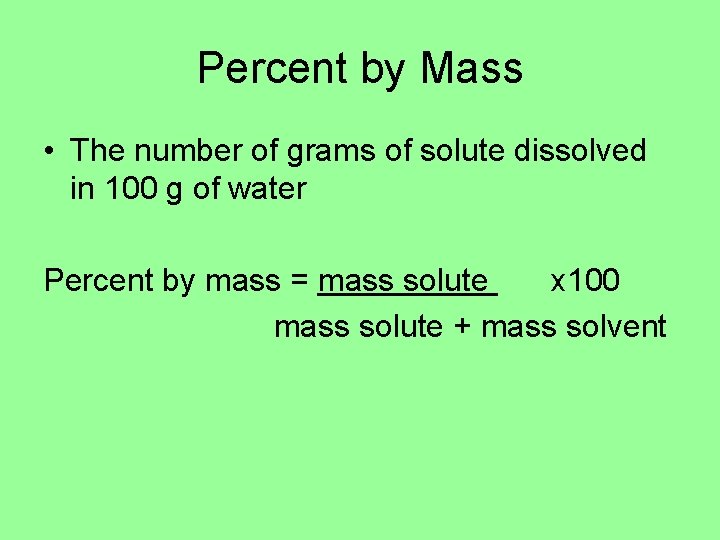














- Slides: 33


Types of Mixtures • Solutions • Suspensions • Colloids

Solutions are: • A homogeneous • mixture of two or 1. more substances in a single phase Composed of: Solvent- the substance that does the dissolving 2. Solute- the substance that is being dissolved Example: In sugar water, water is the solvent and sugar is the solute.

Suspensions • A mixture in which the particles are so large that they settle out unless the mixture is constantly stirred or agitated. • Ex. a jar of muddy water

Colloids • A mixture in which the particles are intermediate in size between those in solutions and suspensions. Ex. paints, milk, mayonaise, fog, chesse

Tyndall Effect • Colloids appear to be homogeneous mixtures (aka solutions) because the individual particles can not be seen…however, they are not true solutions. • The particles are large enough to scatter light. • This effect is known as the Tyndall effect.

Types of Solutions: • Type of solution is determined by the solvent. • Gaseous-mixture of two or more gases • Liquid- solvent is a liquid • Solid- solvent is a solid Examples of: 1. Gaseous- air, scuba tanks 2. Liquid- tea, Koolaid, cokes, salt water 3. Solid- metal alloys, amalgams (dental fillings)

Some solutions conduct electricity: • Electrolytes- a solution that conducts electricity as a result of the formation of ions in solution (examples: salt water, vinegar) • Nonelectrolyte- a solution that does not conduct electricity because there is no formation of ions in solution (example: sugar in oil)

Electrolyte Nonelectrolyte

Factors that Affect Rates of Dissolving Solids in Liquids: 1. Surface Area- increasing surface area increases the rate of dissolving 2. Agitation- stirring or shaking increases rate of dissolving 3. Heat- heating the solvent will increase the rate of dissolving

Solubility • The amount of a substance that is dissolved at solution equilibrium in a specific amount of solvent at a specified temperature. • Factors Affecting Solubility: 1. Nature of Solute and Solvent“like dissolves like”- polar dissolves polar-nonpolar dissolves nonpolar 2. Pressure- changes in pressure have little affect on dissolving solids in liquids but an increase in pressure will increase the solubility of gases in liquids 3. Temperature a) Increasing temp. , decreases gas solubility b) Increasing temp. , increases solid solubility

3 Classifications of Solutions • Saturated- a solution that contains the max. amount of dissolved solute at a given temp. • Unsaturated- a solution that contains less solute than a saturated solution at a given temp. • Supersaturated- a solution that contains more dissolved solute than a saturated solution at a given temp.

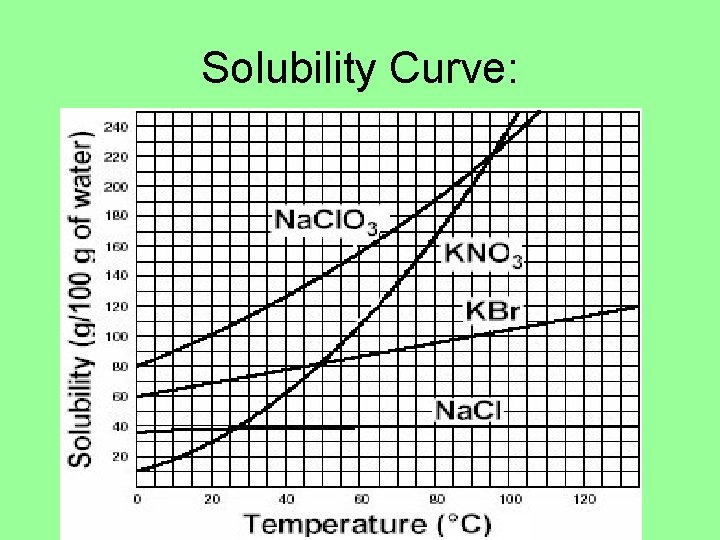
Solubility Curves: • Show much solute can go into solution with a given amount of solvent at different temperatures.

Solubility Curve:

Colligative Properties • A property that depends on the number of solute particles but is independent of their nature • 3 Colligative Properties 1. Vapor Pressure Lowering 2. Freezing Point Depression 3. Boiling Point Elevation

• Vapor Pressure Lowering- the vapor pressure of a solvent containing a nonvolatile solute is lower than the vapor pressure of the pure solvent at any temp. • Freezing Point Depression- the freezing point of a solvent containing a solute will be lower than the pure solvent • Boiling Point Elevation- the boiling point of a solvent containing a solute will be higher than the pure solvent

Solution Concentration: • Concentration is a measurement of the amount of solute in a given amount of solvent or solution • Can be expressed qualitatively or quantitatively • Qualitative Terms: 1. Dilute- relatively small amount of solute compared to solvent 2. Concentrated- relatively large amount of solute in a solvent

Quantitative Terms: • Percent by Mass • Molarity • Molality
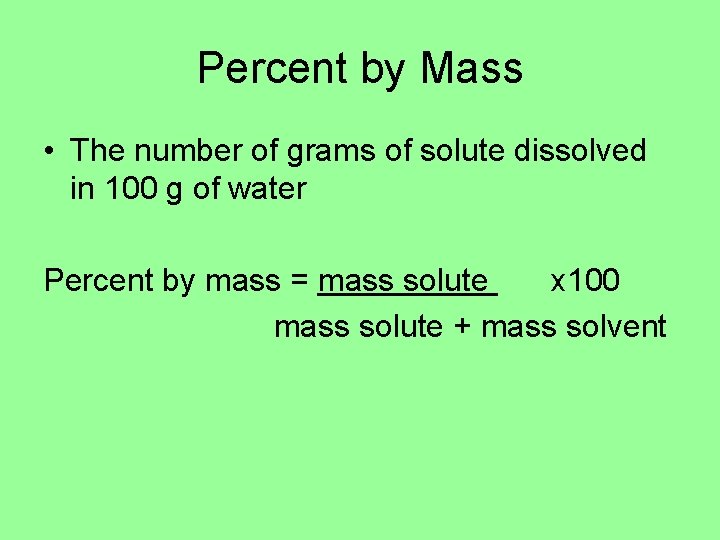
Percent by Mass • The number of grams of solute dissolved in 100 g of water Percent by mass = mass solute x 100 mass solute + mass solvent

Examples: • A solution of sodium chloride is prepared by dissolving 5 g of salt in 550 g of water. What is the concentration as given by percent by mass? Answ: 0. 9% • What is the percent by mass of a solution prepared by dissolving 4 g of acetic acid in 35 g of water. Answ: 10%

Molarity • Symbolized by M • Units of mol L • Describes how many moles of solute are present per liter of solution

Examples: • What is the molarity of 3. 5 L of solution that contains 90 g of sodium chloride? Answ: 0. 44 M • How many moles of HCl are present in 0. 8 L of a 0. 5 M HCl solution? Answ: 0. 4 mol • How many grams of sodium chloride will be required to make 555 m. L of a 1. 45 M solution? Answ: 47. 1 g

More Examples: • How many liters of solution can be prepared if 78. 9 g of sodium chloride is used to make a 3. 00 M solution? Answ: 0. 45 L • What is the molarity of a solution that is prepared by using 20 g of sodium hydroxide in enough water to make a 2 L solution? Answ: 0. 25 M

Dilution Problems • Use the equation: M 1 V 1 = M 2 V 2 Where M 1 = molarity 1, V 1= volume 1…. all volumes must be in liters.

Examples: • What is the molarity of a solution that is made by diluting 50 m. L of a 4. 74 M solution to 250 m. L? Answ: 0. 948 M

Molality Symbolized by m Units are mol solute kg of solvent Describes how many moles of solute are present per kg of solvent.

Examples: • A solution contains 17. 1 g of sucrose (C 12 H 22 O 11) dissolved in 125 g of water. Find the molal concentration. Answ: 0. 400 m • How much iodine must be added to prepare a 0. 480 m solution of iodine in carbon tetrachloride (CCl 4) if 100 g of CCl 4 is used? • Answ: 12. 2 g

More Examples: • What is the molality of a solution composed of 2. 55 g of acetone (CH 3)2 CO, dissolved in 200 g of water? Answ: 0. 219 m • What quantity, in grams, of methanol (CH 3 OH) is required to prepare a 0. 244 M solution in 400 g of water? Answ: 3. 12 g

• How many grams of Ag. NO 3 are needed to prepare a 0. 125 m solution in 250 m. L of water? (Hint. . 1 m. L of water = 1 g and 1 L of water = 1 kg) Answ: 5. 31 g Honors: • What is the molality of a solution that contains 200 g of carbon tetrachloride in 800 m. L of chloroform? The density of chloroform is 1. 489 g/m. L.


Examples: • 2 H 3 PO 4 + 3 Ca(OH)2 Ca 3(PO 4)2 +6 H 2 O 1. If 2 L of 4 M phosphoric acid is used, how many grams of water could be formed? 2. If 6 g of calcium hydroxide is used, how many liters of a 5 M acid solution would be needed?

More Examples: 3. When 200 m. L of 6 M phosphoric acid is used, how many m. L of a 3 M calcium hydroxide solution would be required? 4. What is the molarity of 0. 6 L of calcium phosphate produced when 500 m. L of 3 M phosphoric acid is used?

More Examples: • Na 2 CO 3 + HCl Na. Cl + H 2 O + CO 2 5. If the above reaction was performed at STP and 2 L of carbon dioxide gas was collected, what was the molarity of the 200 m. L of HCl used?