Types of Measurements Different Measurements By Tiffany and

Types of Measurements: Different Measurements By Tiffany and Samantha Qualitative Quantitative The appearance of a sample in words (QUALITY) The amount with numbers (QUANTITY) Ex. Cold, Light, etc. Ex. 18. 3 cm, 10 g, 4 min. SI Units are used to measure in science • Temp. K Kelvin • Length M Meters • Mass Kg Kilograms • Volume L Liters • Time S Seconds Good Measurements are true (accurate) and repeatable (precise)

Can you make a qualitative and quantitative measurement of the following? . 5 grams 20 grams

Accuracy and Precision: Information By charlie zoeller, John Cornfield, & dylan Brooke • When measuring, a measurement should be true and repeatable. • This means that the information is accurate and precise.

Sample • A textbook says that the density of an object is 10 g/cm^3 but a student finds out its actually 8 g/cm^3. • Is it accurate? Yes- The student measured all sides of the book and multiplied them to get 8 g/cm^3 – Is it repeatable? » No- Both the textbook and the students had different answers

Practice Problem • Student one measures a sample of iron to be 3. 34 g. Students two and three measure it to be 3. 3 g. Is student one correct?

Practice problem • Yes, although the student rounded, the info is precise and accurate.

Sig Figs in Measurements By Ian Quinn, Brian Sayre, and Jack Weinberger • Significant Figures are used to reduce the amount of guessing when making measurements. • When measuring, you take all of the certain numbers, plus 1 estimated number.

Measuring using Sig Figs • The last certain number on this graduated cylinder is to the tens place. • We are certain that it goes to 17, but we have to take 1 estimated number past the last certain number. So we would get a measurement of 17. 1
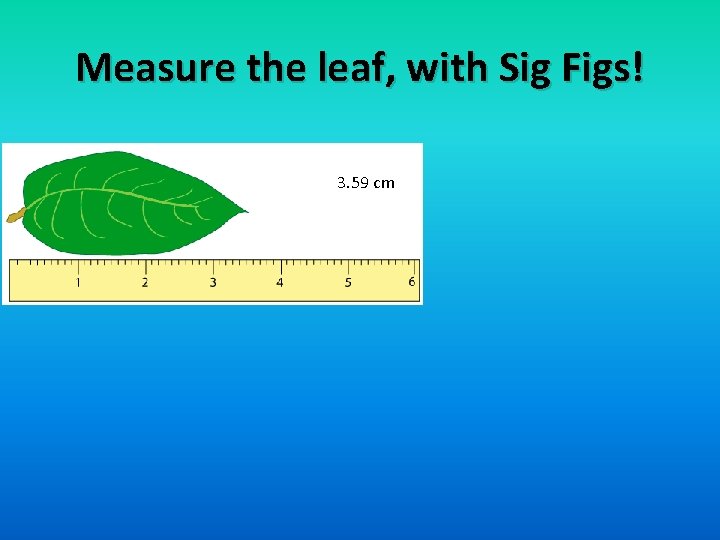
Measure the leaf, with Sig Figs! 3. 59 cm

Multiplying and Dividing Sig. Figs: Rules Mackenzie Saturn, Leah Edmonds and Katelyn Ewell • When multiplying and dividing, the number of Sig. Figs in your answer is the same as the least number of Sig. Figs in your measurement.

Examples-Multiplication Questions: 1. 250. m. L x 5. 6 m. L 2. 50. 54 cm x 30 cm 3. 0. 25 g x 45 g Answers: 1. =1400 m. L^2 2. =2000 cm^2 3. =11 g^2
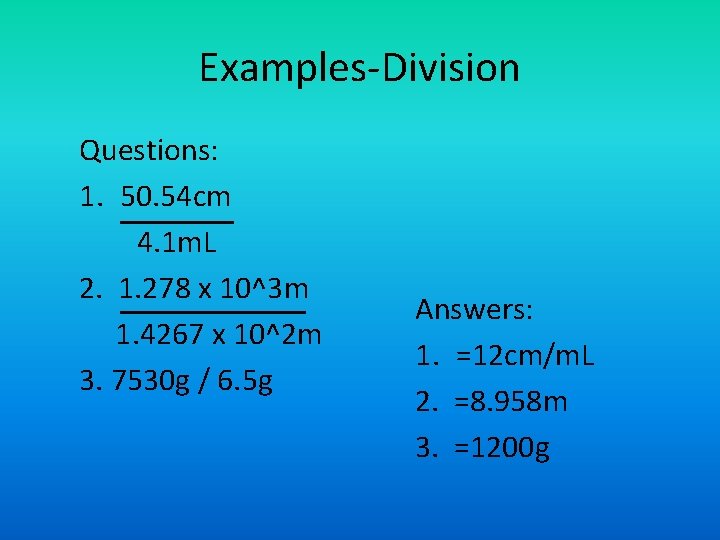
Examples-Division Questions: 1. 50. 54 cm 4. 1 m. L 2. 1. 278 x 10^3 m 1. 4267 x 10^2 m 3. 7530 g / 6. 5 g Answers: 1. =12 cm/m. L 2. =8. 958 m 3. =1200 g

Try it! Questions: 1. 8. 91 x 2700 2. 5000. x 0. 23 3. 2. 90 1. 7 4. 678 4 Answers: 1. =24000 2. =1200 3. =1. 7 4. =200

Metric Prefix Conversions By: Julia Broadbent and Carolyn Belardinelli What is a Prefix? • Prefixes are used to adjust the size of a metric unit.

Positive Prefixes • • • Tera: T-10^12 (1, 000, 000) Giga: G-10^9 (1, 000, 000) Mega: M-10^6 (1, 000) Kilo: k-10^3 (1, 000) Hecto: h-10^2 (100) Deca: da-10^1 (10)

Base Units • Grams, Liters, Meters, etc. • They equal 1 unit

Negative Prefixes • • • Deci: d-10^-1 (. 1) centi: c-10^-2 (. 01) milli: m-10^-3 (. 001) micro: -10^-6 (. 0000001) nano: n-10^-9 (. 000001) pico: p- 10^-12 (. 0000001)

Sample Problems 1) 1 nm=10^-9 m 2) 14 cm*10^-2 m =1. 4*10^-1 m 1 cm -write down # and unit given -put units to get rid of in denominator. Put units wanted in numerator -fill in the equality with correct units

Other Unit Conversions: Information/ How To by Mark And Mike • Converting a unit of measure to another unit of measure such as: inches-miles.

Sample Problem • 3 feet=1 yard • 12 feet to___ yards. • 12 feet/ 3 feet 1 yard • Answer= 4 yards

Practice Problem • ¼ mile= 15, 840 inches • 100, 000 inches to ____ miles.

Answer • 1. 57 miles

What is Density? Maria Weck & Jessica Chu Density is a ratio of mass to volume Density can be used to identify substances. The density of a substance is always the same. You can find density by dividing mass by volume. • The units of each are… density=g/cm^3 or g/m. L, mass=g, volume=cm^3 or m. L • •

Sample Problem • If a 96. 5 g newspaper has a density of 2. 7 g/cm 3, what is its volume? • V=M÷D

Answer • V= 35. 74 cm^3

Percent Error By Daniel Yu and Julian Wolak • Percent error is stated as a percentage of the change between an approximate or measured value and an exact or known value. • To calculate Percent error, you must use this formula: |Accepted Value-Experimental Value| Accepted Value x 100%

Sample Problem You calculate the density of an aluminum block and it is 2. 68 g/cm 3. Then you calculate the density in room temperature and the density is 2. 70 g/cm 3. Calculate the percent of error.

Sample Problem 2. 68 – 2. 70 X 100%. 02/ 2. 70=. 0074074 X 100% =. 74% Percent of Error is. 74% (expressed using 2 Sig. Figs)

Problem • At a track meet, you time a friend running 100 m in 11. 00 seconds. The officials time her at 10. 67 seconds. What is your percentage error?

Answer 10. 67 -11. 00 = -. 33/10. 67=. 03093 X 100% Percent of error is 3. 08%
- Slides: 30