Types of Chemical Reactions Solution Stoichiometry Chapter 4























































- Slides: 65

Types of Chemical Reactions & Solution Stoichiometry Chapter 4

Aqueous Solutions Water is the dissolving medium, or solvent.

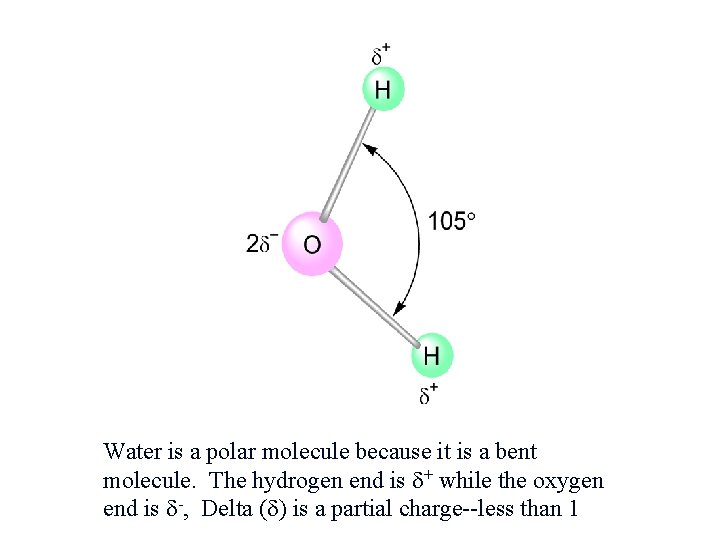
Some Properties of Water is able to dissolve so many substances because: - Water is “bent” or V-shaped. - 105° bond angle - The O-H bonds are covalent. - Water is a polar molecule. - Hydration occurs when salts dissolve in water.

Water is a polar molecule because it is a bent molecule. The hydrogen end is + while the oxygen end is -, Delta ( ) is a partial charge--less than 1.

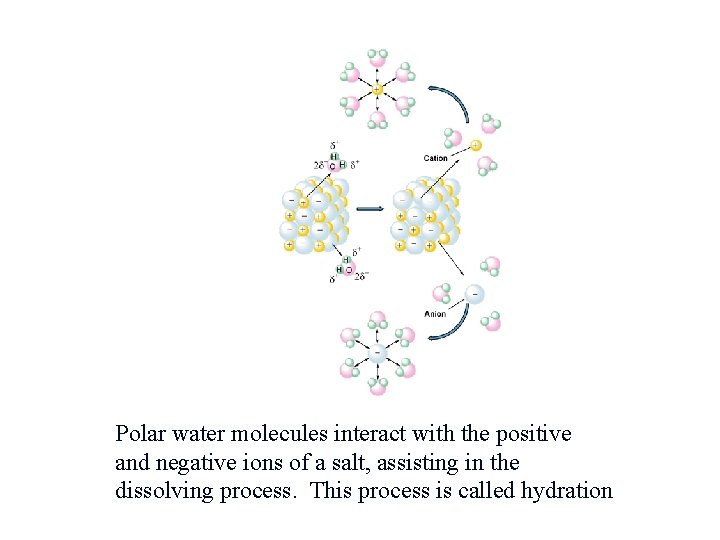
Polar water molecules interact with the positive and negative ions of a salt, assisting in the dissolving process. This process is called hydration.

A Solute - dissolves in water (or other “solvent”) - changes phase (if different from the solvent) - is present in lesser amount (if the same phase as the solvent)

A Solvent - retains its phase (if different from the solute) - is present in greater amount (if the same phase as the solute)

Solubility The general rule for solubility is: “Like dissolves like. ” Polar water molecules can dissolve other polar molecules such as alcohol and, also, ionic substances such as Na. Cl. Nonpolar molecules can dissolve other nonpolar molecules but not polar or ionic substances. Gasoline can dissolve grease.

Miscibility Miscible -- two substances that will mix together in any proportion to make a solution. Alcohol and water are miscible because they are both polar and form hydrogen bonds. (compounds that have hydrogen bonding readily dissolve) Immiscible -- two substances that will not dissolve in each other. Oil and vinegar are immiscible because oil is nonpolar and vinegar is polar.

Solubility How does the rule “Like dissolves like. ” apply to cleaning paint brushes used for latex paint as opposed to those used with oil-based paint? C 6 H 14 H 20 I 2 Na. NO 3 H 20 HF H 20

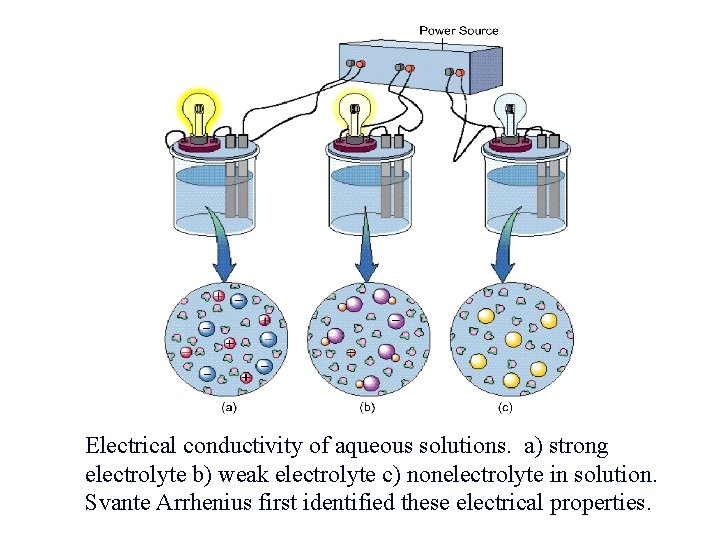
Electrolytes & Nonelectrolytes An electrolyte is a material that dissolves in water to give a solution that conducts an electric current. A nonelectrolyte is a substance which, when dissolved in water, gives a nonconducting solution.

Electrolytes Strong - conduct current efficiently and are soluble salts, strong acids, and strong bases. Na. Cl, KNO 3, HNO 3, Na. OH Weak - conduct only a small current and are weak acids and weak bases. HC 2 H 3 O 2, aq. NH 3, tap H 2 O Non - no current flows and are molecular substances pure H 2 O, sugar solution, glycerol

Electrical conductivity of aqueous solutions. a) strong electrolyte b) weak electrolyte c) nonelectrolyte in solution. Svante Arrhenius first identified these electrical properties.

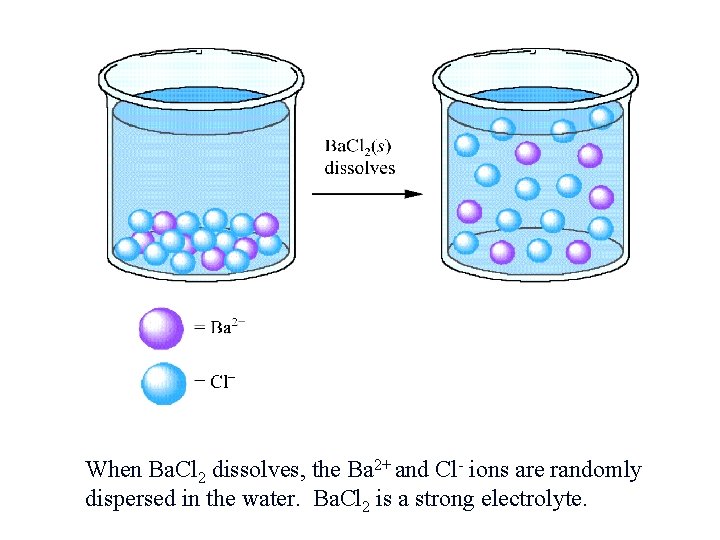
When Ba. Cl 2 dissolves, the Ba 2+ and Cl- ions are randomly dispersed in the water. Ba. Cl 2 is a strong electrolyte.

Acids Strong acids - dissociate completely (~100 %) to produce H+ in solution HCl, H 2 SO 4, HNO 3, HBr, HI, & HCl. O 4 Weak acids - dissociate to a slight extent (~ 1 %) to give H+ in solution HC 2 H 3 O 2, HCOOH, HNO 2, & H 2 SO 3

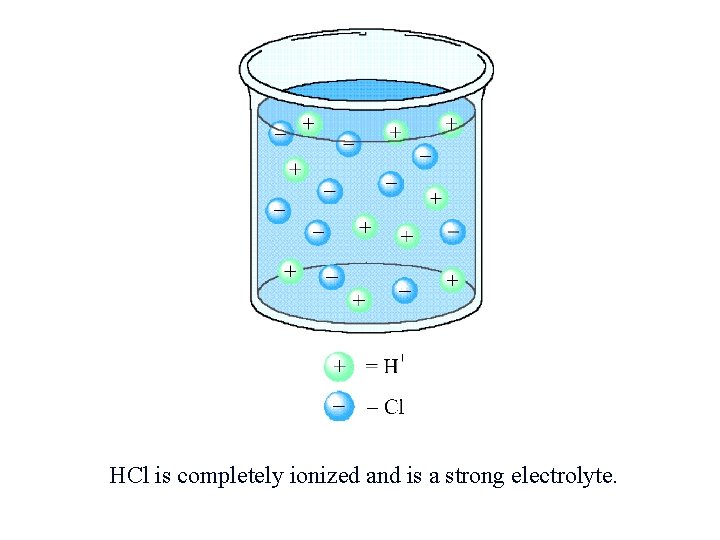
HCl is completely ionized and is a strong electrolyte.

Bases Strong bases - react completely with water to give OH ions. sodium hydroxide Na. OH(s) ---> Na+(aq) + OH-(aq) Weak bases - react only slightly with water to give OH ions. ammonia NH 3(aq) + HOH(l) <---> NH 4+(aq) + OH-(aq)

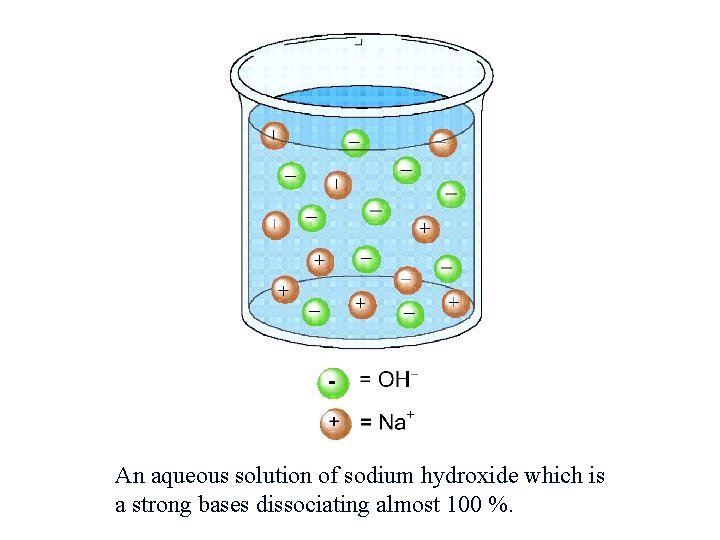
An aqueous solution of sodium hydroxide which is a strong bases dissociating almost 100 %.

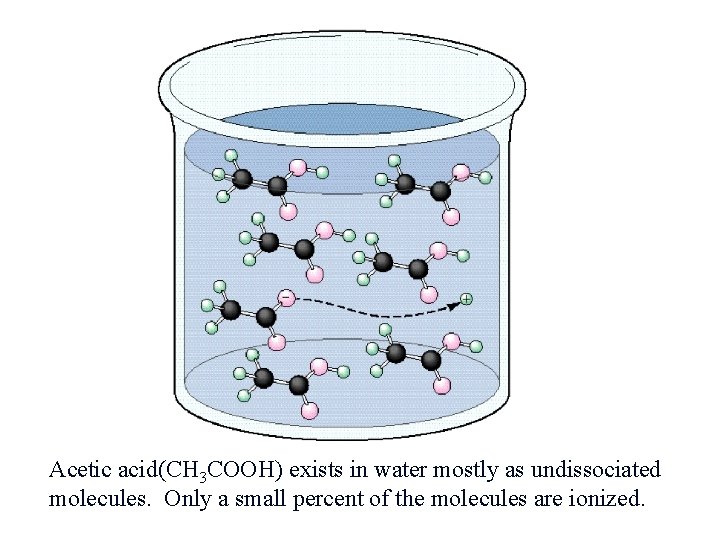
Acetic acid(CH 3 COOH) exists in water mostly as undissociated molecules. Only a small percent of the molecules are ionized.

Write the equation of the dissolving of the following compounds. Ca. Cl 2 HCl Fe(NO 3)3 KBr (NH 4)2 Cr 2 O 7

Molarity (M) = moles of solute per volume of solution in liters: 1. Calculate the molarity of a solution prepared by dissolving 11. 5 g of solid Na. OH in enough water to make 1. 50 L of solution. 2. Calculate the molarity of a solution prepared by dissolving 1. 56 g of gaseous HCl in enough water to make 26. 8 m. L of solution .


Calculating moles in a solution How many moles of Co(NO 3)2 are present in 25. 00 m. L of a 0. 75 M Co(NO 3)2 solution? How many moles of nitrate ions are present in 25. 00 m. L of a 0. 75 M Co(NO 3)2 solution?


Calculating moles in a solution Calculate the moles of each of the ions in 40. 00 ml of. 20 M Na 2 CO 3 solution. Calculate the moles of potassium ions in 50. 00 ml of the following solution. 2 M K 3 P


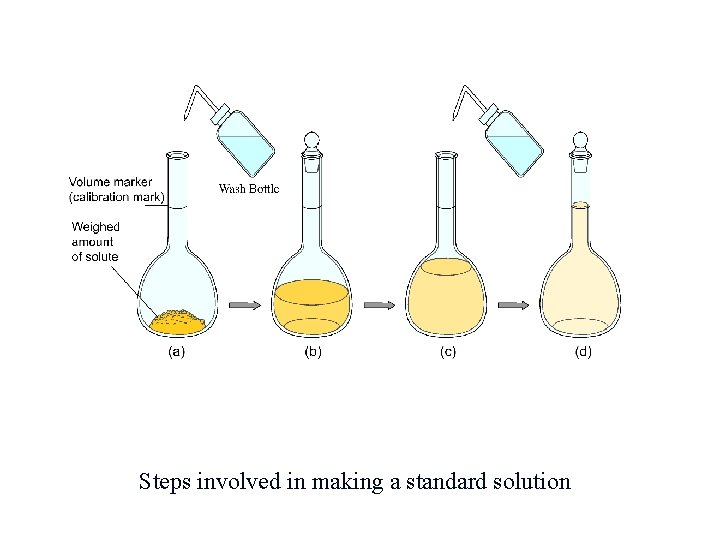
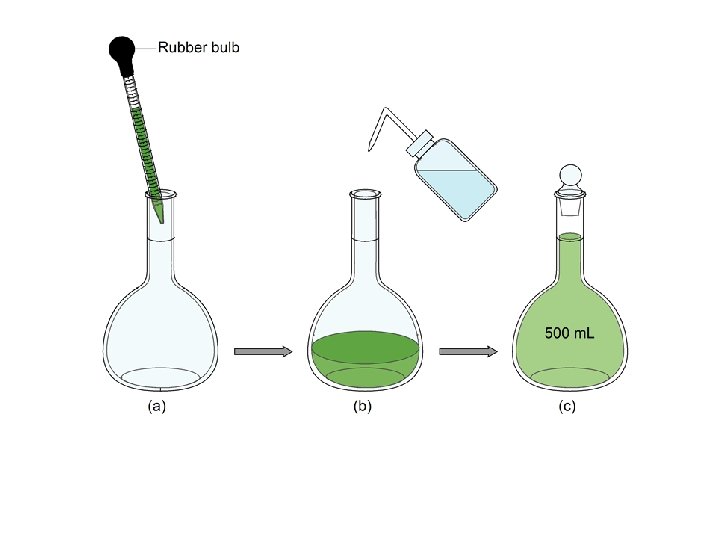
Standard Solution A standard solution is a solution whose concentration is accurately known. Standard solutions are made using a volumetric flask as follows: • mass the solute accurately and add it to the volumetric flask • add a small quantity of distilled HOH • dissolve the solute by gently swirling the flask • add more distilled HOH until the level of the solution reaches the mark on the neck • invert the capped volumetric 25 X to thoroughly mix the solution.

Steps involved in making a standard solution.

Steps to dilute a stock solution.

Common Terms of Solution Concentration Stock - routinely used solutions prepared in concentrated form. Concentrated - relatively large ratio of solute to solvent. (5. 0 M Na. Cl) Dilute - relatively small ratio of solute to solvent. (0. 01 M Na. Cl)



When diluting stock solutions, the moles of solute after dilution must equal the moles of solute before dilution. Stock solutions are diluted using either a measuring or a delivery pipet and a volumetric flask. What volume of 6 M sulfuric acid must be used to prepare 600 ml of a 1. 0 M H 2 SO 4 solution? use dilution formula What volume of 2 M sulfuric acid must be used to prepare 1 L of a. 5 M H 2 SO 4 solution?


Types of Solution Reactions - Precipitation reactions Ag. NO 3(aq) + Na. Cl(aq) Ag. Cl(s) + Na. NO 3(aq) - Acid-base reactions Na. OH(aq) + HCl(aq) Na. Cl(aq) + H 2 O(l) - Oxidation-reduction reactions Fe 2 O 3(s) + 2 Al(s) 2 Fe(l) + Al 2 O 3(s)

1. Which of the following substances would you expect to be insoluble in water? Barium hydroxide Magnesium sulfate Silver chloride Calcium carbonate Ammonium acetate Sodium hydroxide Silver nitrate Hydrochloric acid Ammonium nitrate Lithium carbonate Barium sulfate Lead I Chloride Ammonium nitrate

Solubility Using the solubility rules, predict what will happen when the following pairs of solutions are mixed. a) KOH(aq) & Mg(NO 3)2(aq) Mg(OH)2(s) forms b) Na 2 SO 4(aq) & Pb(NO 3)2(aq) c) KNO 3(aq) & Ba. Cl 2(aq) Pb. SO 4(s) forms No precipitate forms

Describing Reactions in Solution 1. Molecular equation (reactants and products as compounds) Ag. NO 3(aq) + Na. Cl(aq) Ag. Cl(s) + Na. NO 3(aq) 2. Complete ionic equation (all strong electrolytes shown as ions) Ag+(aq) + NO 3 (aq) + Na+(aq) + Cl (aq) Ag. Cl(s) + Na+(aq) + NO 3 (aq)

Describing Reactions in Solution (continued) 3. Net ionic equation (show only components that actually react) Ag+(aq) + Cl (aq) Ag. Cl(s) Na+ and NO 3 are spectator ions.

Write the balanced complete ionic and net ionic equations: Cu. SO 4(aq) + Li. Cl(aq) → P 108 q 28

Sodium Sulfate and Lead II Nitrate


Old limiting problem to compare If 68. 5 g of CO(g) is reacted with 8. 60 g of H 2(g), what is theoretical yield of methanol that can be produced? __H 2(g) + __CO(g) ---> __CH 3 OH(l)

Precipitation Calculations When aqueous solutions of Na 2 SO 4 & Pb(NO 3)2 are mixed. Calculate the mass of the percipitate formed when 1. 25 L of 0. 0500 M Pb(NO 3)2 & 2. 00 L of 0. 0250 M Na 2 SO 4 are mixed. 1.

Now it’s the same as limiting you already know.

Precipitation Calculations Continued 1 (1. 25 L)(0. 0500 mol Pb(NO 3)2/1 L) = 0. 0625 mol. Pb(NO 3)2 (2. 00 L)(0. 0250 mol Na 2 SO 4/1 L) = 0. 0500 mol. Na 2 SO 4 (Now that you have moles it’s the same as limiting you know. ) 2. Divide by the coefficients Na 2 SO 4 is the limiting reactant Start with the limititng 3. (0. 0500 mol Na 2 SO 4)(1 mol Pb. SO 4/1 mol Na 2 SO 4) (303. 3 g/1 mol Pb. SO 4) = 15. 2 g Pb. SO 4

PRACTICE What mass of precipitate should result when 0. 550 L of 0. 500 M aluminum nitrate solution is mixed with 0. 240 L of 1. 50 M sodium hydroxide solution?


What mass of precipitate should result when 0. 350 L of 0. 200 M aluminum nitrate solution is mixed with 0. 540 L of. 50 M sodium hydroxide solution? What is the net ionic equation? P 108 29 -32


Mixed Limiting A. 750 g sample of Iron II Chloride is dissolved in water and a mixed with 22. 40 ml of. 515 M Ag. NO 3. Calculate the grams of precipitate.


Practice precipitation What mass of Fe(OH)3 is produced when 35. ml of a. 250 M Fe(NO 3)3 solution is mixed with 55 ml of a 0. 180 M KOH? Green book p 95


In the experiment, a student is given 2. 94 g of a mixture containing anhydrous Mg. Cl 2 and KNO 3. To determine the percentage by mass of Mg. Cl 2 in the mixture, the student uses excess Ag. NO 3(aq) to precipitate the chloride ion as Ag. Cl(s). The student determines the mass of the Ag. Cl precipitate to be 5. 48 g. On the basis of this information, calculate The number of moles of Mg. Cl 2 in the original mixture.




Green book review p 106 # 44 practice gravimetric analysis A 2. 10 g sample of silver alloy was dissolved and the precipitated as Ag. Br. After washing and drying the silver bromide weighed 2. 00 g. Calculate the percentage silver in the alloy.





What volume of 0. 415 M silver nitrate will be required to precipitate as silver bromide all the bromide ion in 35. 0 m. L of 0. 128 M calcium bromide?
