Types of chemical reactions Lesson 12 Types of












- Slides: 14

Types of chemical reactions Lesson 12

Types of chemical reactions 1. Combustion Reaction - The reaction of a substance with oxygen, producing oxides and energy • Fuel + oxygen oxides + energy • AB + oxygen common oxides of A and B (ex AO, BO)

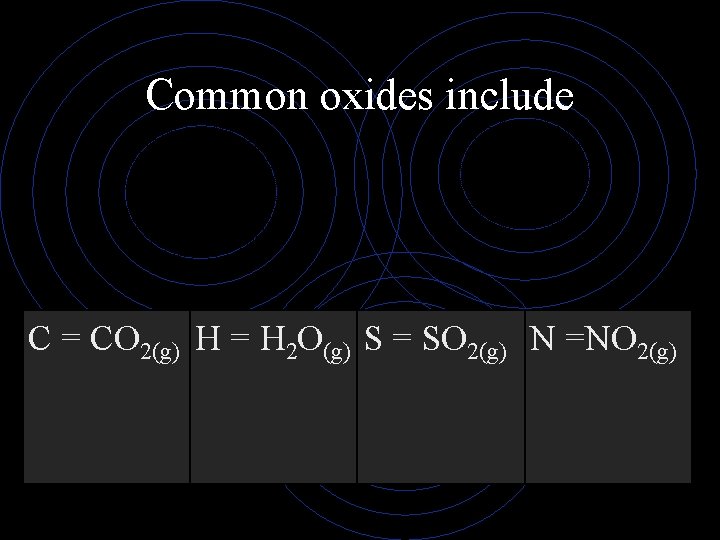
Common oxides include C = CO 2(g) H = H 2 O(g) S = SO 2(g) N =NO 2(g)

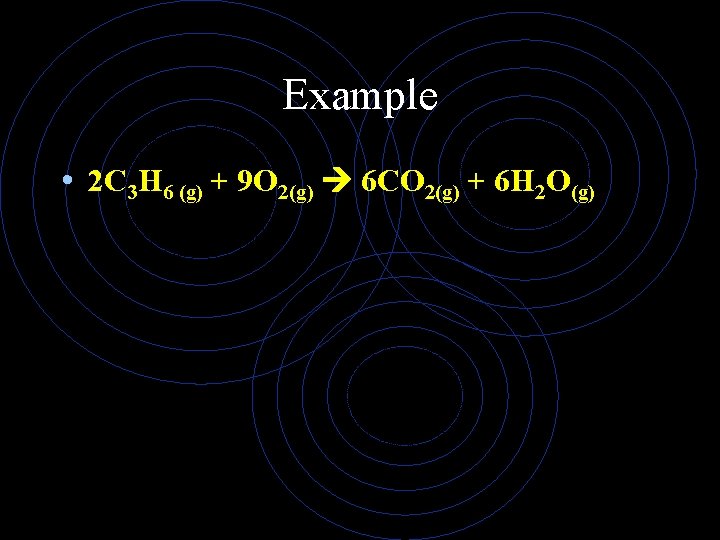
Example • 2 C 3 H 6 (g) + 9 O 2(g) 6 CO 2(g) + 6 H 2 O(g)

• Incomplete combustion occurs when there is not sufficient oxygen. When this occurs 4 products are produced instead of the usual H 2 O and CO 2. Incomplete combustion also produces CO(g) and C(s). This is commonly seen when lighting an acetylene torch. • 3 C 2 H 2 (g) + 3 O 2(g) 2 CO 2(g) + 3 H 2 O(g) + 2 C(s)

2. Synthesis Reaction • -A chemical reaction in which two or more substances combine to form a more complex substance. • A + B AB • Example: • 2 CO(g) + O 2(g) pt 2 CO 2(g)

3. Decomposition reaction • -A chemical reaction in which a compound is broken down into two or more simpler substances. • AB A + B • Example: The decomposition of water. • 2 H 2 O(l) + electricity 2 H 2(g) + O 2(g)

Chemical reactions in solution • A Solution is a homogenous mixture in which a pure substance, called the solute, is dissolved in another pure substance called the solvent. • • The solution is often an aqueous solution which is a solution where water is the solvent.

4. Single Displacement reaction • -A reaction of an element with a compound to produce a new element and a new compound. The reaction will only occur if the element is higher on the reactivity series than the metal in the compound. • A + BC AC + B • Example: • Cu(s) + 2 Ag. NO 3(aq) 2 Ag(s) + Cu(NO 3)2(aq)


5. Double displacement reaction • - A reaction in which aqueous ionic compounds rearrange cations and anions, resulting in the formation of new compounds. • AB + CD AD + CB • Example: • Pb(NO 3)2 (aq) + 2 KI (aq) Pb. I 2 (s) + 2 KNO 3 (aq)

Double displacement is likely to occur if; • - a precipitate is produced • - a gas is produced • - a acid base neutralization occurs

Examples • 2 As + 3 O 2 2 As 2 O 3 • Type of reaction? • KCl. O 4 KCl + 2 O 2 • Type of reaction? • Zn(s) + Cu. Cl 2(s) the table. Will it occur? Look at

• Cu(s) + Zn. Cl 2(s) • will it occur • Mg. Cl 2(aq) + Ca(OH)2(aq)