Types of Chemical Reactions Combination Reactions Reactions where

Types of Chemical Reactions

Combination Reactions • Reactions where two or more substances combine to form a new substance • General Formula: A + B AB
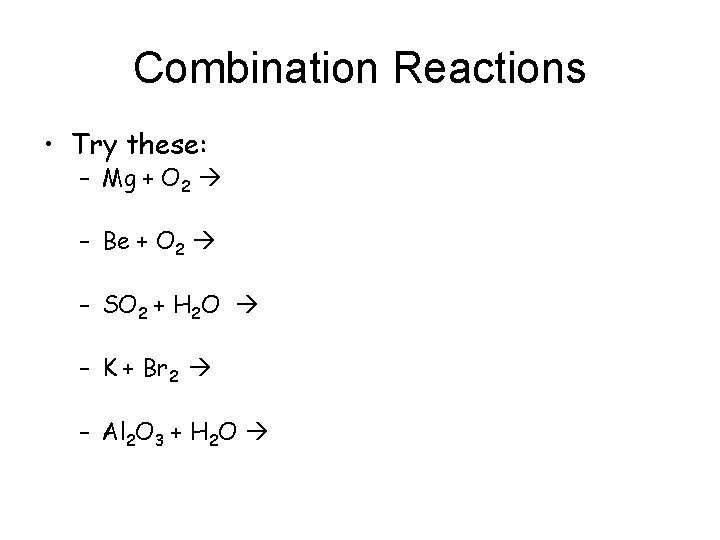
Combination Reactions • Try these: – Mg + O 2 – Be + O 2 – SO 2 + H 2 O – K + Br 2 – Al 2 O 3 + H 2 O

Combination Reactions • Answers: – 2 Mg + O 2 2 Mg. O – 2 Be + O 2 2 Be. O – SO 2 + H 2 O H 2 SO 3 – 2 K + Br 2 2 KBr – 2 Al 2 O 3 + 6 H 2 O 4 Al(OH)3

Decomposition Reactions • Reactions where a single compound breaks apart into two or more new compounds • General Formula: AB A + B

Decomposition Reactions Try these: – Ca. CO 3 – Na. Cl – Hg. O – Mg(Cl. O 3) 2 – Fe(OH)3

Decomposition Reactions Answers: – Ca. CO 3 Ca. O + CO 2 – 2 Na. Cl 2 Na + Cl 2 – 2 Hg. O 2 Hg + O 2 – Mg(Cl. O 3) 2 Mg. Cl 2 + 3 O 2 – Fe(OH)3 Fe 2 O 3 + H 2 O

Combustion Reactions • Reactions where a compound combines with oxygen to form one or more new compounds. • General Formula: A + O 2 AO + (H 2 O) + (B)

Combustion Reactions – Try These • C 6 H 6 + O 2 • C 6 H 12 O 6 + O 2 • Na + O 2 • P + O 2 • Cu + O 2

Combustion Reactions – Answers: • 2 C 6 H 6 + 15 O 2 12 CO 2 + 6 H 2 O • C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O • 4 Na + O 2 2 Na 2 O • 4 P + 5 O 2 2 P 2 O 5 • 2 Cu + O 2 2 Cu. O

Single Replacement Reactions • Reactions where one element replaces a second element in a compound • General Formula: A + BC AC + B

Single Replacement Reactions • Try these: – K + Ag. NO 3 – HCl + Zn – Cu. Br 2 + Na – Mg + Zn(NO 3)2 – Fe 2 O 3 + H 2

Single Replacement Reactions • Answers: – K + Ag. NO 3 KNO 3 + Ag – Zn + 2 HCl Zn. Cl 2 + H 2 – Cu. Br 2 + 2 Na. Br + Cu – Mg + Zn(NO 3)2 Zn + Mg(NO 3)2 – Fe 2 O 3 + 3 H 2 2 Fe + 3 H 2 O

Double Replacement Reactions • Reactions where positive ions are exchanged between compounds • General Formula: AB + CD AD + BC

Double Replacement Reactions • Try These: – Pb(NO 3)2 + Cu. SO 4 – Ag. Mn. O 4 + Zn. Cl 2 – Na. OH + HCl – H 2 SO 4 + Al(OH) 3 – Ba(NO 3) 2 + Na. SO 4
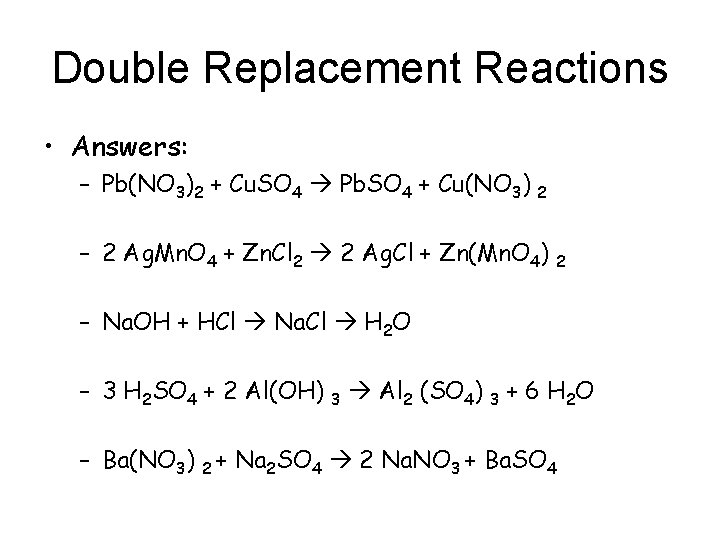
Double Replacement Reactions • Answers: – Pb(NO 3)2 + Cu. SO 4 Pb. SO 4 + Cu(NO 3) 2 – 2 Ag. Mn. O 4 + Zn. Cl 2 2 Ag. Cl + Zn(Mn. O 4) 2 – Na. OH + HCl Na. Cl H 2 O – 3 H 2 SO 4 + 2 Al(OH) 3 Al 2 (SO 4) 3 + 6 H 2 O – Ba(NO 3) 2 + Na 2 SO 4 2 Na. NO 3 + Ba. SO 4

Single Replacement Reactions H from Acids H from H 2 O • Metal reactivity series applies

Single Replacement Reactions • Try these - metal reactivity series applies – Mg + 2 Ag. NO 3 – 2 Li + 2 H 2 O – Ag + KNO 3 – Mg + Li. NO 3 – 3 Ca + 2 Al. Cl 3

Single Replacement Reactions • Answers - metal reactivity series applies – Mg + 2 Ag. NO 3 Mg(NO 3)2 + 2 Ag – 2 Li + 2 H 2 O 2 Li. OH + H 2 – Ag + KNO 3 no reaction – Mg + Li. NO 3 no reaction – 3 Ca + 2 Al. Cl 3 3 Ca. Cl 2 + 2 Al

Double Replacement Reactions • Solubility Table Applies

Double Replacement Reactions • Answers - Solubility Table Applies – K 2 CO 3 + Sr(NO 3)2 – NH 4 Cl + Na 3 PO 4 – HCl + Zn. S – NH 4 Cl + Pb(NO 3)2 – Na 3 PO 4+ Li 2 SO 4

Double Replacement Reactions • Answers - Solubility Table Applies – K 2 CO 3 + Sr(NO 3)2 Sr. CO 3↓ + 2 KNO 3 – 3 NH 4 Cl + Na 3 PO 4 Na. Cl + (NH 4)3 PO 4↓ – 2 HCl + Zn. S Zn. Cl 2+ H 2 S ↑ – 2 NH 4 Cl + Pb(NO 3)2 Pb. Cl 2 ↓ + NH 4 NO 3 – Na 3 PO 4+ Li 2 SO 4 no reaction
- Slides: 22