Types of Chemical Reactions 6 2 Assigning oxidation





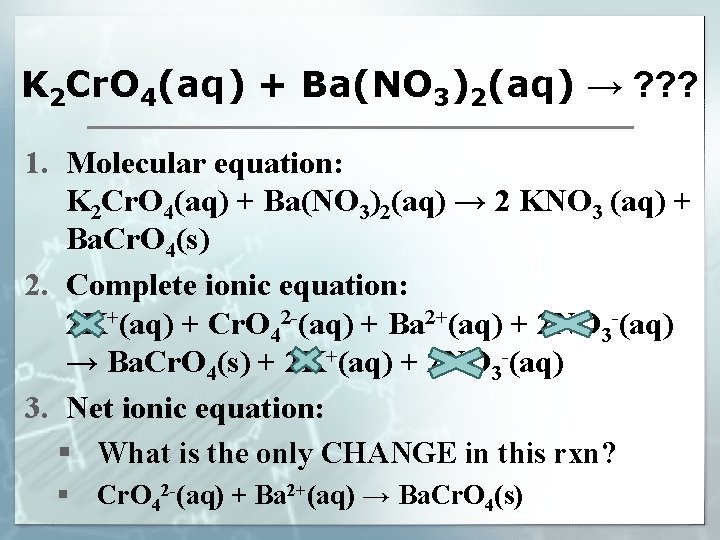


- Slides: 8

Types of Chemical Reactions 6. 2 Assigning oxidation numbers for individual atoms of monatomic and polyatomic ions 6. 3 Identifying the nomenclature of ionic compounds, binary compounds, and acids 6. 4 Classifying chemical reactions as composition, decomposition, single replacement, or double replacement AHSGE Reading 4. 2 Demonstrate the ability to preview and predict. Other AOD C. 6. 1 Define stoichiometry, reactants, and products.

What are some types of reactions? 1. 2. 3. 4. 5. chemical Synthesis Decompostion Single Replacement/Single Displacement Double Replacement/Double Displacement Combustion § Complete combustion yields ____. § Incomplete combustion yields _____.

“Classes” of Reactions 1. Precipitation reactions 2. Acid-base reactions 3. Oxidation-reduction reactions § Almost all reaction types can be put into one of these three “classes”.

Precipitation Reactions § What do you think this reaction would involve? § AKA, double replacement/double displacement § Example: K 2 Cr. O 4(aq) + Ba(NO 3)2(aq) → ? ? ? § What does (aq) mean? § What are some other designations? § What is the name of K 2 Cr. O 4? Ba(NO 3)2? § What would the products be in a double displacement reaction? § Would either product be a solid (precipitate)?

Three Ways to Write the Reaction Equation 1. Molecular equation: shows the overall balanced reaction (reactants and products) 2. Complete ionic equation: shows a balanced equation of the actual FORMS of the reactants and products in the reaction 3. Net ionic equation: balanced equation showing ONLY the ions involved in the reaction. § Ions that remain in solution (as ions) are not shown. (“spectator ions”)
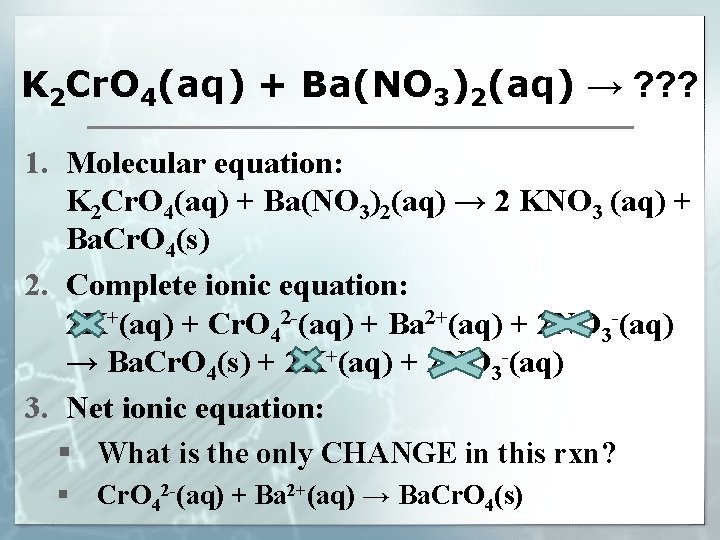
K 2 Cr. O 4(aq) + Ba(NO 3)2(aq) → ? ? ? 1. Molecular equation: K 2 Cr. O 4(aq) + Ba(NO 3)2(aq) → 2 KNO 3 (aq) + Ba. Cr. O 4(s) 2. Complete ionic equation: 2 K+(aq) + Cr. O 42 -(aq) + Ba 2+(aq) + 2 NO 3 -(aq) → Ba. Cr. O 4(s) + 2 K+(aq) + 2 NO 3 -(aq) 3. Net ionic equation: § What is the only CHANGE in this rxn? § Cr. O 42 -(aq) + Ba 2+(aq) → Ba. Cr. O 4(s)

Examples § See Sample Exercise 4. 9, p. 155. § P. 182 (29 a): § Ba. Cl 2(aq) + Na 2 SO 4(aq) → Ba. SO 4(s) + 2 Na. Cl(aq) § Net ionic equation: Ba 2+(aq) + SO 42 -(aq) → Ba. SO 4(s) § What happened to the “Cl 2” and the “Na 2” part of the molecular equation? § What would the complete ionic equation look like?

Assignment § P. 182: 30, 34, 36 § Special note on #34: You are MAKING UP a chemical reaction that will produce the specified solid. § It should be a precipitation (double displacement) reaction.