TYPES OF CHEMICAL BONDS IONIC BONDS COVALENT BONDS

TYPES OF CHEMICAL BONDS • IONIC BONDS • COVALENT BONDS • HYDROGEN BONDS • METALLIC BONDS


IONIC BONDING When an atom of a nonmetal takes one or more electrons from an atom of a metal so both atoms end up with eight valence electrons

IONIC BONDING IS THE COMPOUND AN IONIC COMPOUND? METAL NONMETAL SUBSCRIPTS

IONIC BOND FORMATION Neutral atoms come near each other. Electron(s) are transferred from the Metal atom to the Non-metal atom. They stick together because of electrostatic forces, like magnets.

IONIC BONDING Metals will tend to lose electrons and become POSITIVE CATIONS Normal sodium atom loses one electron to become sodium ion

IONIC BONDING Nonmetals will tend to gain electrons and become NEGATIVE ANIONS Normal chlorine atom gains an electron to become a chloride ion

Properties of Ionic Compounds • Crystalline structure. • A regular repeating arrangement of ions in the solid. • Ions are strongly bonded. • Structure is rigid. • High melting points- because of strong forces between ions.
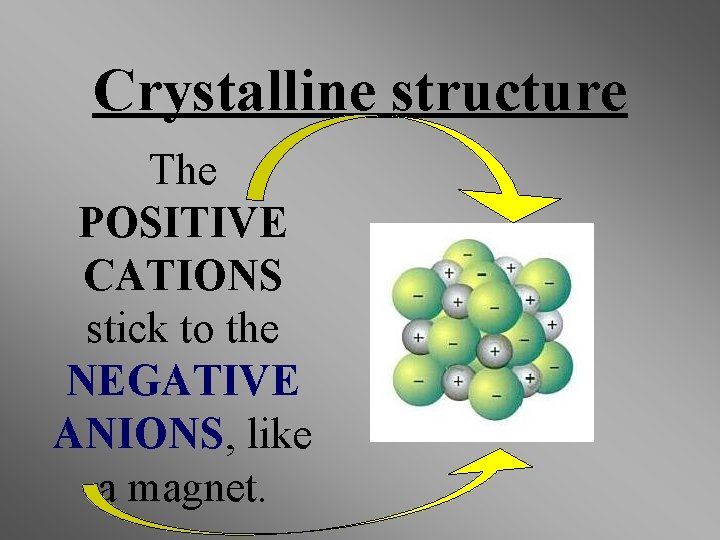
Crystalline structure The POSITIVE CATIONS stick to the NEGATIVE ANIONS, like a magnet.

Do they Conduct? • Conducting electricity is allowing charges to move. • In a solid, the ions are locked in place. • Ionic solids are insulators. • When melted, the ions can move around. • Melted ionic compounds conduct. • Melting points always above 800ºC. • Dissolved in water they conduct.
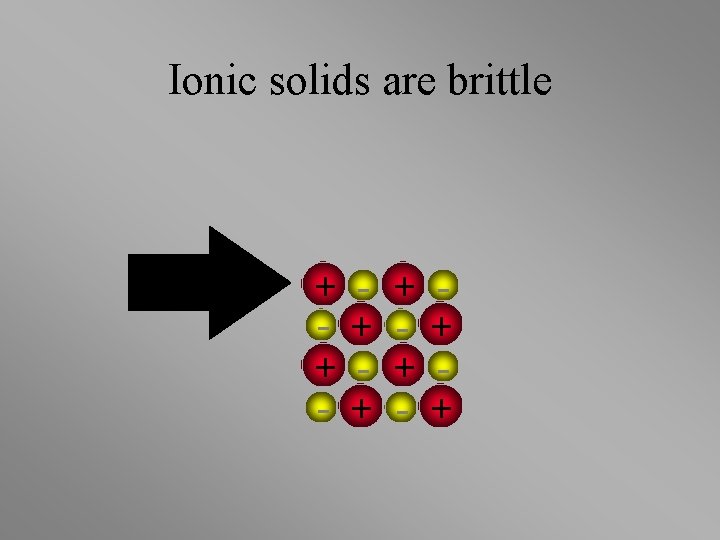
Ionic solids are brittle + + - + +

Ionic solids are brittle • Strong Repulsion breaks crystal apart. + + - + - + - +


COVALENT BOND FORMATION When one nonmetal shares one or more electrons with an atom of another nonmetal so both atoms end up with eight valence electrons

COVALENT BONDING IS THE COMPOUND A COVALENT COMPOUND? NONMETAL YES since it is made of only nonmetal elements

Covalent bonding • Fluorine has seven valence electrons F

Covalent bonding • Fluorine has seven valence electrons • A second atom also has seven F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l Both end with full orbitals l F F

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l Both end with full orbitals l F F 8 Valence electrons

Covalent bonding Fluorine has seven valence electrons l A second atom also has seven l By sharing electrons l Both end with full orbitals l 8 Valence electrons F F

Single Covalent Bond • A sharing of two valence electrons. • Only nonmetals and Hydrogen. • Different from an ionic bond because they actually form molecules. • Two specific atoms are joined. • In an ionic solid you can’t tell where the electrons came from

Water H Each hydrogen has 1 valence electron Each hydrogen wants 1 more O The oxygen has 6 valence electrons The oxygen wants 2 more They share to make each other happy

Water • Put the pieces together • The first hydrogen is happy • The oxygen still wants one more HO

Water • The second hydrogen attaches • Every atom has full energy levels HO H

Two types of covalent bonds: • Sigma bonds: represented by the symbol two orbitals with a DIRECT overlay (so it falls DIRECTLY between the nuclei of the atoms in the bond)

Two types of bonds Another view of sigma bonds (showing electron clouds):
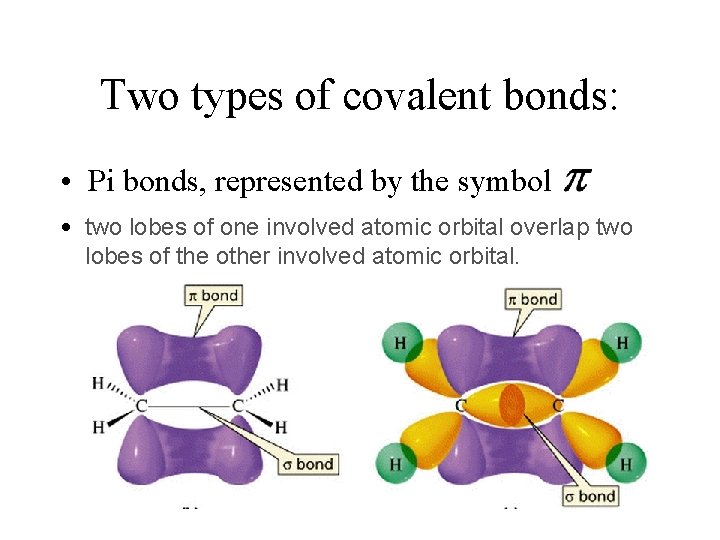
Two types of covalent bonds: • Pi bonds, represented by the symbol • two lobes of one involved atomic orbital overlap two lobes of the other involved atomic orbital.
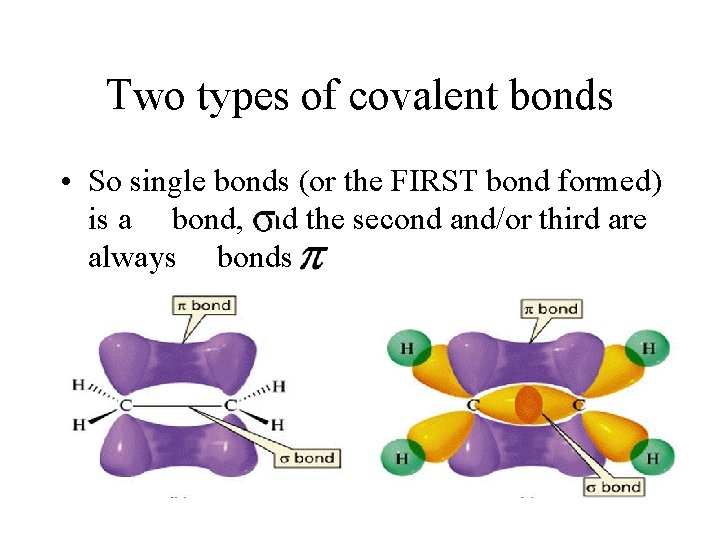
Two types of covalent bonds • So single bonds (or the FIRST bond formed) is a bond, and the second and/or third are always bonds
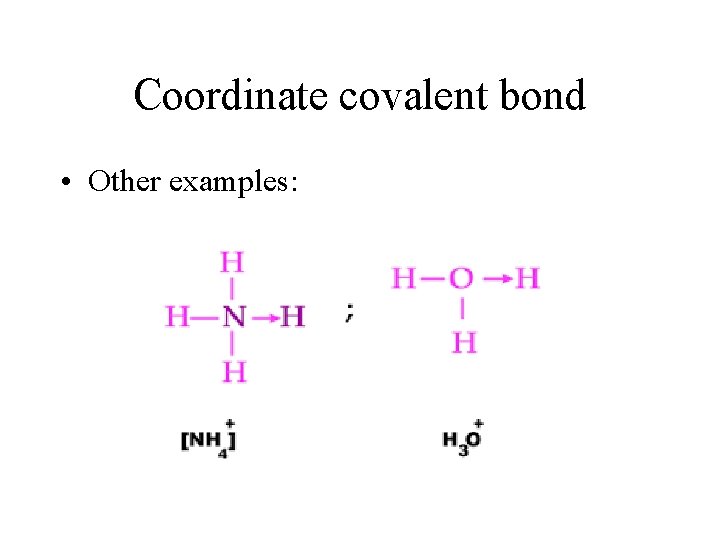
Coordinate covalent bond • Other examples:

Polar Molecules • Molecules with a positive and a negative end • Requires two things to be true ¬ The molecule must contain polar bonds This can be determined from differences in electronegativity. Symmetry can not cancel out the effects of the polar bonds. Must determine geometry first.

Is it polar? • HF • H 2 O • NH 3 • CCl 4 • CO 2

Intermolecular Forces What holds molecules to each other

Intermolecular Forces • They are what make solid and liquid molecular compounds possible. • The weakest are called van der Waal’s forces there are two kinds • Dispersion forces • Dipole Interactions – depend on the number of electrons – more electrons stronger forces – Bigger molecules

Dispersion forces • When electrons temporarily ‘clump’ in one part of a molecule • Results in a temporary attractive force from a temporary dipole

Dispersion Forces • Also called London Forces or Van der Waals forces • The larger the molecule, the greater the additive effect

Dispersion Forces • This is why gasoline is a solid at room temperature, but methane is a gas

Dipole interactions • Depend on the number of electrons • More electrons stronger forces • Bigger molecules more electrons • Fluorine is a gas • Bromine is a liquid • Iodine is a solid

Dipole interactions • Occur when polar molecules are attracted to each other. • Slightly stronger than dispersion forces. • Opposites attract but not completely hooked like in ionic solids. + d d H F
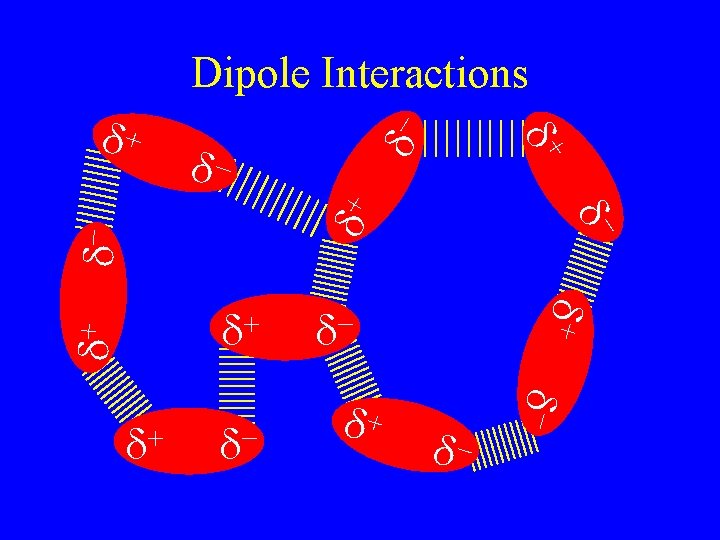
dd+ - d- d d+ d- d- d+ d+ + d d d+ + d- d d+ d- Dipole Interactions

Hydrogen bonding • Are the attractive force caused by hydrogen bonded to F, O, or N. • F, O, and N are very electronegative so it is a very strong dipole. • The hydrogen partially share with the lone pair in the molecule next to it. • The strongest of the intermolecular forces.
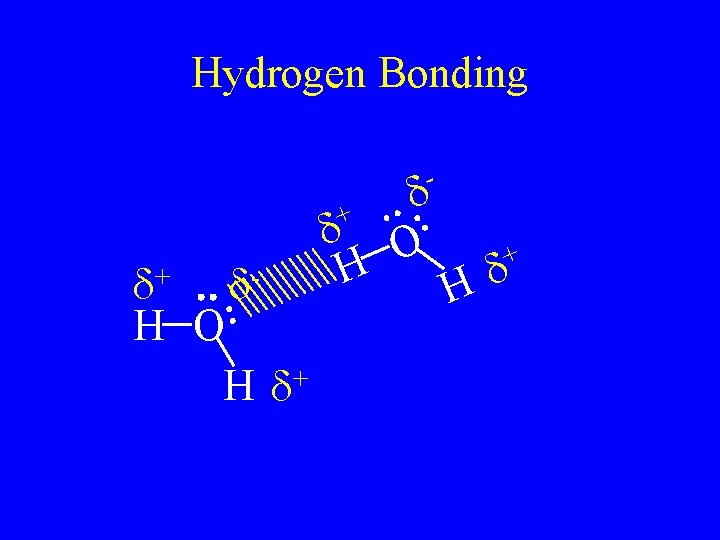
Hydrogen Bonding - + d+ d. H O + Hd d H d O + d H
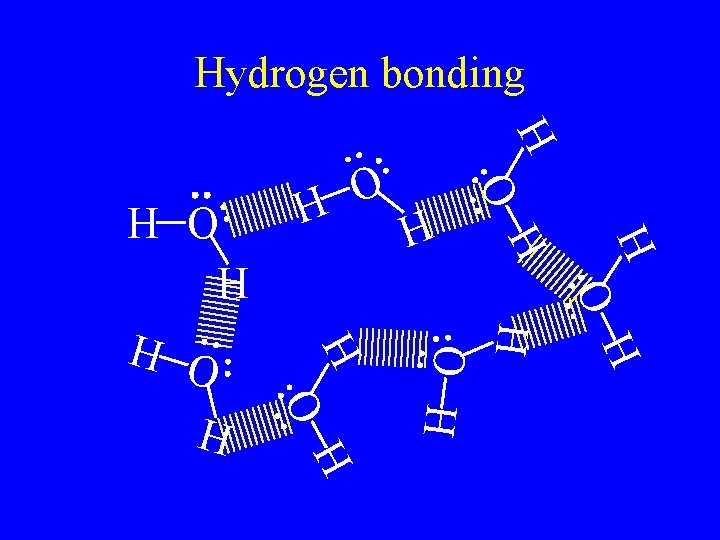
H H O H H O O H Hydrogen bonding

Metallic Bonds • Metals hold onto their valence electrons very weakly. • Think of them as positive ions floating in a sea of electrons.

Sea of Electrons • Valence electrons are free to move through the solid. • Metals conduct electricity. + + +

Metals are Malleable • Hammered into shape (bend). • Ductile - drawn into wires.
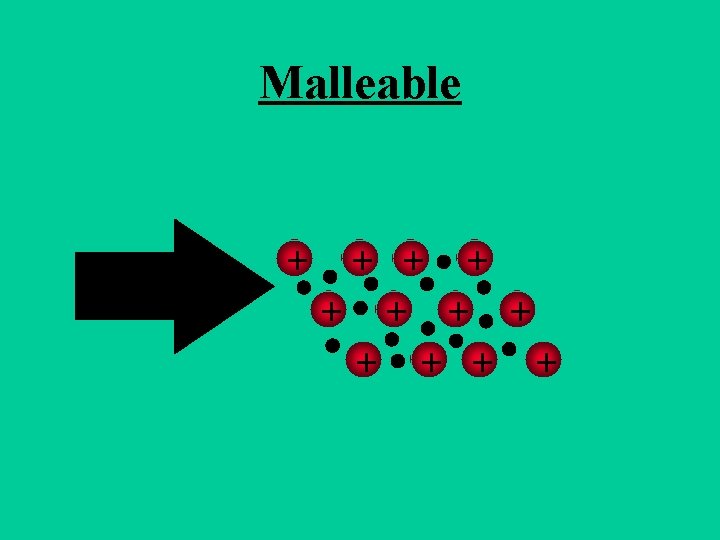
Malleable + + +
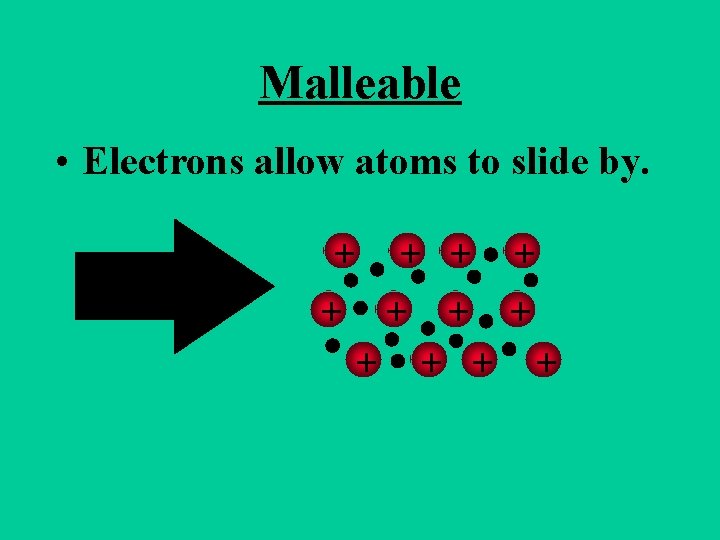
Malleable • Electrons allow atoms to slide by. + + +
- Slides: 52