TYPES OF BONDS CHEMICAL BONDING Chemical Bond An

TYPES OF BONDS

CHEMICAL BONDING Chemical Bond: An electrical attraction between the nuclei and valence electrons of different atoms Three types of Bonds Metallic Bond Ionic Bond Covalent bonding (Next unit)
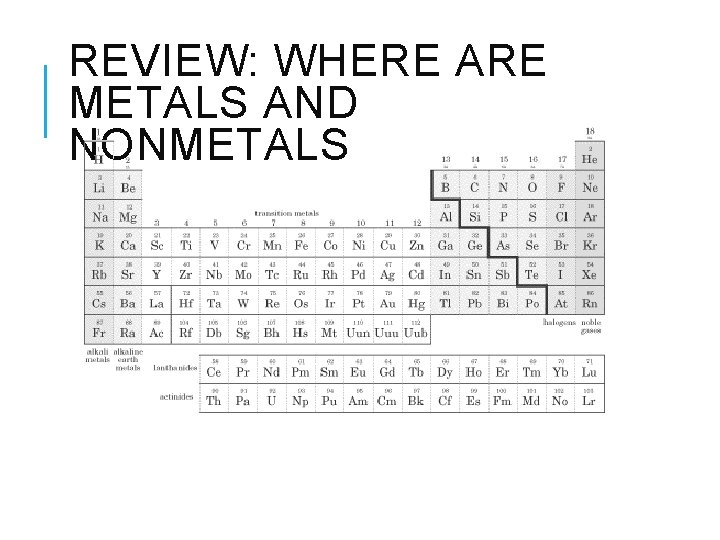
REVIEW: WHERE ARE METALS AND NONMETALS

1. METALLIC BONDS made up of tightly packed cations surrounded by mobile valence electrons (sea of electrons) this explains why metals are such great conductors of electricity!! Sea of electrons

METALLIC PROPERTIES 1. High electrical and thermal conductivity due to the mobile electrons 2. Strong absorbers and reflectors of light (high luster) Energy differences are small in orbitals, so it is easy for the electrons to get excited and then relax back down which emits a photon! Remember those? ? 3. Malleable: ability of a substance to be hammered or beaten into sheets 4. Ductility: Ability of a substance to be pulled into a wire These are both a property, because metallic bonding is the same in all directions. Atoms in the metal can slide past another without breaking

All lone metals have metallic bonding. Ex. Fe, Ag, Al… Once they combine with a nonmetal, they do a different type of bonding…

2. IONIC BONDING Ionic Bond: results the transfer of electrons between cations and anions Usually a metal and non metal Ex) Mg 2+ Cl- combine to make Mg. Cl 2 Cations: positive charge (typically metals) Anions: negative charge (typically nonmetals) **CATIONS ARE ALWAYS WRITTEN FIRST IN FORMULA!!!***

FORMULA UNIT Formula unit: Chemical formula for an ionic compound that shows the lowest whole number ratio of ions. Ex) Na. Cl

PROPERTIES OF IONIC BONDS 1. Most are crystal structures at room temperature (repeated three dimensional patterns) 2. Ionic compounds have a high melting point It requires a lot of energy to break the ions apart because each ion is surrounded by ions of opposite charge – VERY STABLE)

3. Ionic compounds are brittle, not malleable 4. When melted or in aqueous solution (water), ionic compounds conduct electricity 5. Not Volatile when dissolved in water Volatile: Liquid vaporizes at room temperature (giving off fumes) Ex. Nail polish is very volatile!!

3. COVALENT BOND Covalent bonds: The result of two nonmetals Sharing electrons ONLY between nonmetals! Remember hydrogen is a nonmetal!

MOLECULES/MOLECULA R COMPOUNDS Molecule: neutral group of atoms that act as 1 unit (electrons are shared) Molecules give the exact number of atoms. Ex: H 2 O 2

DIATOMIC & TRIATOMIC MOLECULES Diatomic molecules: Consist of two atoms of the same element Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2 Triatomic molecules: Consist of three atoms of the same element (ex O 3 ozone)

PROPERTIES OF MOLECULES 1. Lower melting & boiling points (than Ionic) 2. Soft or brittle (shatter easily) 3. Poor or no electrical conductivity when dissolved or melted 4. Volatile when dissolved in water

POLYATOMIC IONS Polyatomic Ions: A covalently bonded group of atoms that have a charge §Only exist in ionic compounds §Mostly negative §Almost ALL end in “-ate” or “-ite”

MODELING IONIC COVALENT METALLIC
- Slides: 16