Types and descriptions Objectives By the end of

Types and descriptions Objectives: By the end of this Power. Point students will be able to: • Categorize matter by type • Explain the difference between pure substances and mixtures. • Differentiate between homogeneous and heterogeneous mixtures.

Matter Anything that takes up space and has mass Examples of things that are matter Water Air Energy and forces are not matter Examples of things that are not matter Heat Light Gravity

There are Two Major Categories of Matter Pure Substance Matter which has the same composition and the same properties Examples water oxygen salt Mixtures Two or more types of matter physically mixed together in the same container Examples salt water air cup of coffee

There are Two Types of Pure Substances Elements Compounds

Elements are the simplest type of matter They can not be broken down any further by chemical means Examples carbon (C) oxygen (O)

Compounds are two or more elements chemically “stuck” together These can be broken down in chemical reactions Examples carbon dioxide (CO 2) water (H 2 O)
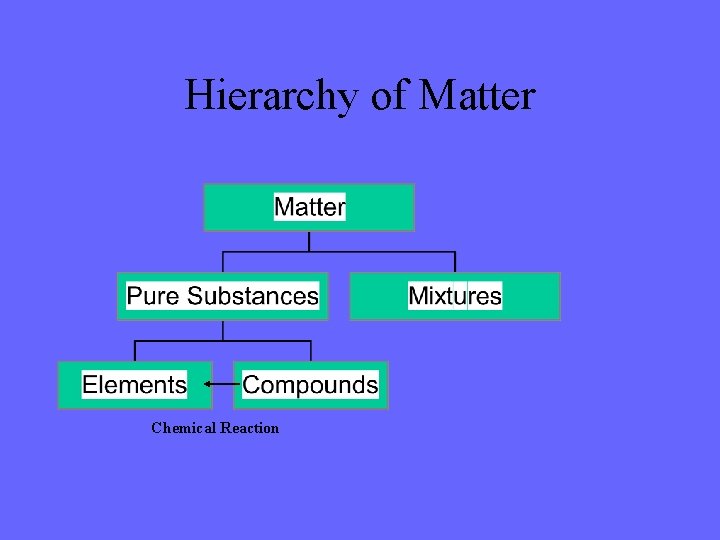
Hierarchy of Matter Chemical Reaction
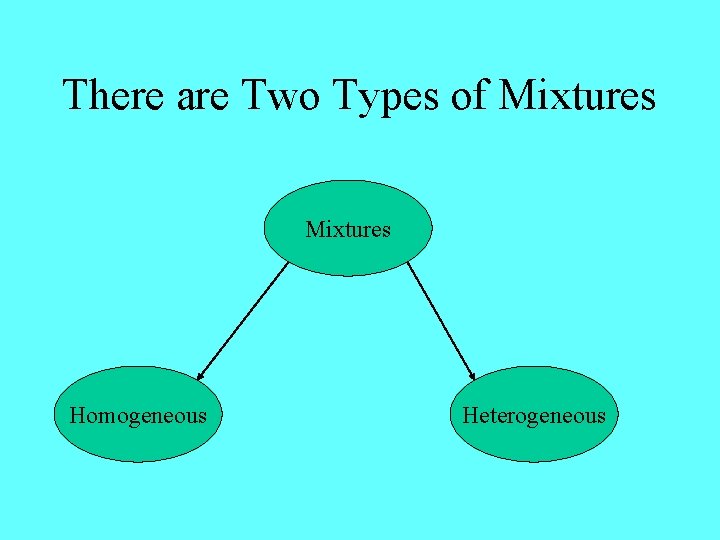
There are Two Types of Mixtures Homogeneous Heterogeneous

Homogeneous Mixtures Homogeneous mixtures have the same composition throughout They are mixed evenly Examples salt water lotions

Heterogeneous Mixtures The composition of a heterogeneous mixture will vary They are not mixed evenly Examples oil and water vegetable soup

Hierarchy of Matter Physical separation Chemical Reaction
- Slides: 11