Type of Bonds Intramolecular Bonds Ionic Bonds Covalent

Type of Bonds Intramolecular Bonds Ionic Bonds Covalent Bond Metallic Bonds Author: J R Reid

Intramolecular Bonds Intramolecular bonds are those hold two or more atoms together: Note: Intra = Inside, Molecular = molecules. These bonds hold compounds and poly-atomic elements together: Compound = a chemical made from two or more different elements bonded together e. g. H 2 O and H 2 SO 4 Polyatomic = an element made from two or more atoms from the same element bonded together e. g. O 2 and S 8 These bonds come in three forms – Ionic, Covalent and Metallic

Ionic Bonds - Basics Form between cation and anions Ions are charged atoms. They are charged because they have lost or gained electrons. This is due to great EN difference between atoms. Bond formed is reliant on the electrostatic attraction between oppositely charges ions Most compounds made from a metal and a non -metal will have ionic bonds holding them together

Ionic Bonds - Characteristics Ions group themselves together into huge lattices (see picture) Ionic substances are solids and most dissolve in water High melting points and boiling point = lots of energy required to break bonds Properties: Hard but brittle and conduct electricity when molten or in solution but not in the solid state Bonding forces increase as the size of the ion decreases (less distance between ions) AND as charge on the ion increases.

Covalent Bonds - Basics Covalent bonds form between non-metals A covalent bond is created by two atoms sharing their valence (outer/bonding) electrons to gain full shells (see Electron Configuration) The diagram to the right shows to fluorine atoms with 7 valence electrons sharing one each to create outer shells of 8 electrons

Covalent Bonds - Basics Strong electrostatic forces between the nuclei of two atoms and the shared electrons are involved in the bond Bonds between identical atoms or equal EN – equal sharing which are non polar or pure covalent bonds e. g. H 2, Cl 2, O 2 Bonds between atoms of high but similar EN – unequal sharing which are polar covalent bonds, shared pair of electrons move closer to more EN atom causing a slightly – and slightly + ends or dipoles e. g HCl

Covalent Bonds - Characteristics Covalent bonds are strong bonds. Strength decreases as we go down the PT – bonding pair further from nucleus, shielding But covalent molecules are normally gases or liquids if they do not form long chains or networks: Gases – O 2, F 2, Cl 2, N 2 and H 2 Liquid – H 2 O and Br 2 Solids – C (as graphite and diamond)

Covalent network solids Rigid 3 D structure held by covalent bonds Very high mp and bp as large amount of energy required to break bonds Generally insoluble in all solvents due to large amount of energy required to break bonds cannot be broken by the dissolving process.

Covalent network solids

Metallic Bonds Metals have a loose hold on their electrons. Therefore a metallic substance shares its electrons very easily with its neighbours. ( delocalised electrons) A metallic substance can be considered to be nuclei (lots of nucleuses) in a sea of electrons. Metals form a 3 d lattice of metal ions Metallic bond is the electrostatic attraction between the positive nuclei of the metal and the surrounding negative electron sea
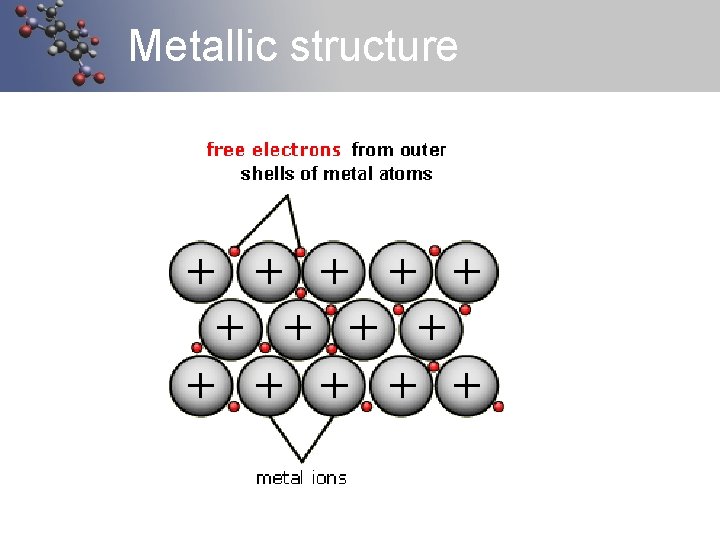
Metallic structure

Metals - Characteristics Metals have a loose hold on their electrons. They also form large lattices (like ionic substances). Therefore they have the following characteristics: Electrical conductivity Thermal conductivity Malleable and ductile (dentable and stretchy) Sonority (they ring when hit) Solid (except mercury) These characteristics are due to the fact that the bonds are not rigid (they can flow), they don’t have to bond in particular directions and the electrons are free to move from one atom to the next

Notes time Worksheet exercise “ Sorting intra molecular bonds”
- Slides: 13