Type 2 WHAT Science Scoop TYPE OF BONDS

Type: 2 WHAT Science Scoop TYPE OF BONDS DO THE COMPOUNDS BELOW POSSESS? Compound A Compound B Crystalline solid Gas at room temperature Melting point 800ºC Melting point 23ºC When melted or placed in water Does not conduct electricity in it conducts electricity any state Dissolves in water Does not dissolve in water (a polar liquid)

NAMING COVALENT COMPOUNDS

WRITING COVALENT COMPOUNDS Made of two nonmetals Identify the less electronegative element 1 st (one farthest left & farthest down on Periodic Table) The 1 st nonmetal is just given the name as found on the Periodic Table (just like ionic) The 2 nd nonmetal ending is changed to –ide (just like ionic…don’t change polyatomics) Differences = numerical prefixes are used to express how many of each nonmetal are present And no criss-crossing!

COVALENT BONDING USING LEWIS DOTS In covalent bonding, pairs of electrons are shared F+F H + Cl C+O
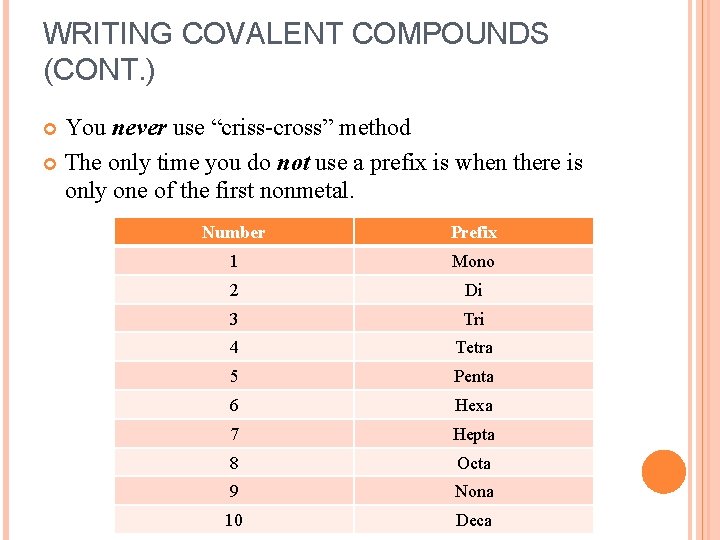
WRITING COVALENT COMPOUNDS (CONT. ) You never use “criss-cross” method The only time you do not use a prefix is when there is only one of the first nonmetal. Number Prefix 1 Mono 2 Di 3 Tri 4 Tetra 5 Penta 6 Hexa 7 Hepta 8 Octa 9 Nona 10 Deca

COVALENT COMPOUND PRACTICE 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. SO 2 N 2 O NO 2 CCl 4 Cl 2 O 7 PCl 3 SF 6 Si 3 N 4 N(Br. O 3)5 H 2 O 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Sulfur Dioxide Dinitrogen Monoxide Nitrogen Dioxide Carbon Tetrachloride Dichlorine Heptaoxide Phosphorus Trichloride Sulfur Hexaflouride Trisilicon Tetranitride Nitrogen Pentabromate Dihydrogen Monoxide

COVALENT COMPOUND PRACTICE 1. 2. 3. 4. 5. 6. 7. 8. Phosphorus Trioxide Dinitrogen Pentacarbide Tellurium Noniodide Carbon Monoxide Selenium Heptafluoride Tetraphosphorous Decoxide Arsenic Hexabromide Silicon Dichloride 1. 2. 3. 4. 5. 6. 7. 8. PO 3 N 2 C 5 Te. I 9 CO Se. F 7 P 4 O 10 As. Br 6 Si. Cl 2
- Slides: 7