Type 1 Diabetes Treatment Options Part 3 Stanley

Type 1 Diabetes Treatment Options Part 3 Stanley Schwartz Emeritus, Univ of Pa Mark Stolar
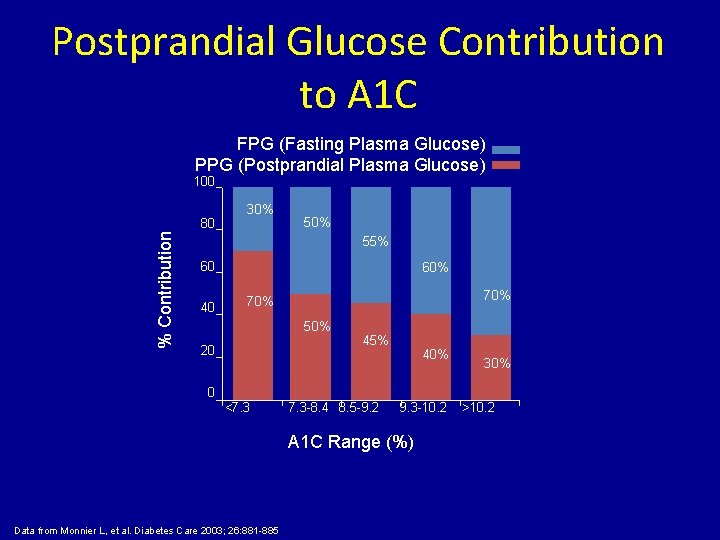
Postprandial Glucose Contribution to A 1 C FPG (Fasting Plasma Glucose) PPG (Postprandial Plasma Glucose) 100 % Contribution 80 30% 55% 60 40 60% 70% 50% 20 0 <7. 3 45% 7. 3 -8. 4 8. 5 -9. 2 40% 9. 3 -10. 2 A 1 C Range (%) Data from Monnier L, et al. Diabetes Care 2003; 26: 881 -885 30% >10. 2

The Basal/Bolus Insulin Concept • Basal insulin – Suppresses glucose production between meals and overnight – Nearly constant levels – ~50% of daily needs • Bolus insulin (mealtime or prandial) – Limits hyperglycemia after meals – Immediate rise and sharp peak at 1 hr – ~10% to 20% of daily requirement at each meal • Ideally, for insulin replacement therapy, each component should come from a different insulin with specific profile Rosenstock J. Clin Cornerstone. 2001; 4: 50
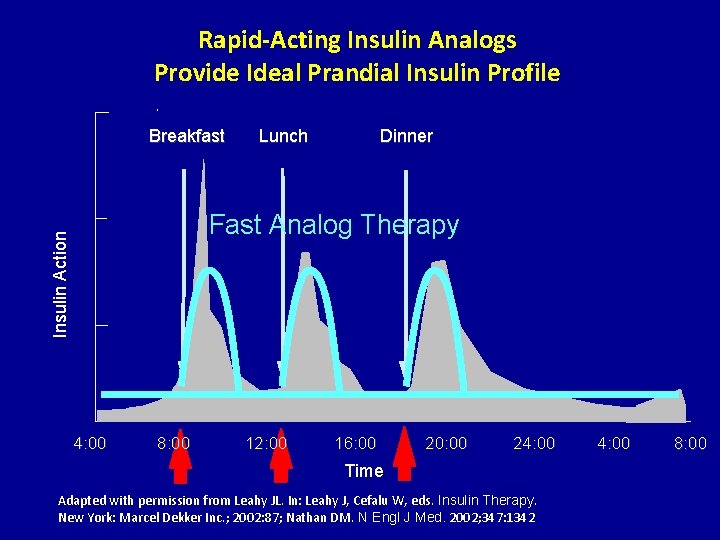
Rapid-Acting Insulin Analogs Provide Ideal Prandial Insulin Profile Breakfast Lunch Dinner Insulin Action Fast Analog Therapy 4: 00 8: 00 12: 00 16: 00 20: 00 24: 00 Time Adapted with permission from Leahy JL. In: Leahy J, Cefalu W, eds. Insulin Therapy. New York: Marcel Dekker Inc. ; 2002: 87; Nathan DM. N Engl J Med. 2002; 347: 1342 4: 00 8: 00
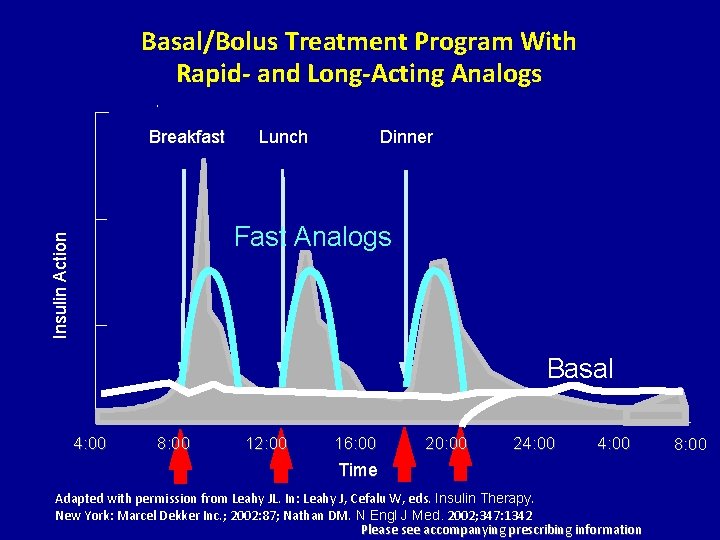
Basal/Bolus Treatment Program With Rapid- and Long-Acting Analogs Breakfast Lunch Dinner Insulin Action Fast Analogs Basal 4: 00 8: 00 12: 00 16: 00 20: 00 24: 00 Time Adapted with permission from Leahy JL. In: Leahy J, Cefalu W, eds. Insulin Therapy. New York: Marcel Dekker Inc. ; 2002: 87; Nathan DM. N Engl J Med. 2002; 347: 1342 Please see accompanying prescribing information 8: 00
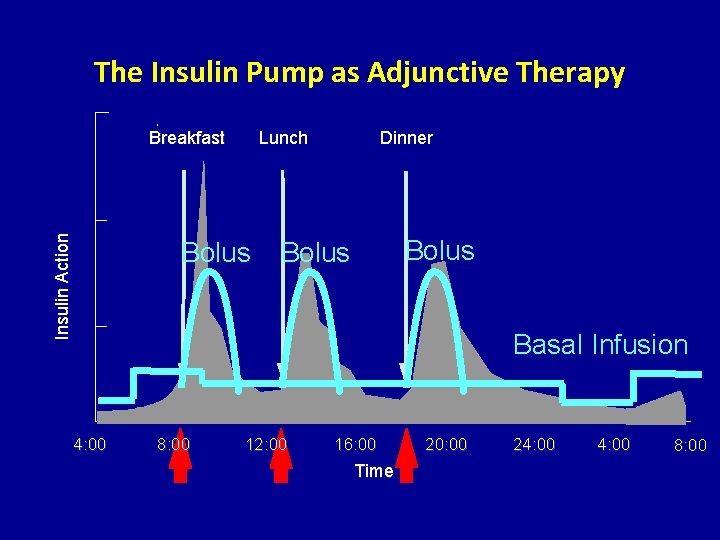
The Insulin Pump as Adjunctive Therapy Insulin Action Breakfast Lunch Bolus Dinner Bolus Basal Infusion 4: 00 8: 00 12: 00 16: 00 Time 20: 00 24: 00 8: 00

β-Cell (Islet Cell) Classification Model. Implications for Therapy: Targets for Adjunctive Therapies Direct Effect on β-Cells On #1 -3 of ‘Egregious Eleven’ Egregious Eleven Defect Intervention / Therapy 1 β-CELL Incretin 2 α-Cell Glucagon Incretin, Pramlintide 3 ↓INCRETIN EFFECT Incretin 4

Disrupted Insulin and Glucagon Regulation: Central Role in Diabetes Pathophysiology • Hyperglucagonemia is a characteristic of T 1 DM and T 2 DM • Data suggest a central role for glucagon dysregulation in diabetes pathophysiology – Without paracrine intraislet insulin signals from juxtaposed β cells (image), α cells hypersecrete glucagon (diagram) – Hyperglucagonemia can account for abnormalities associated with lack of insulin (eg, increased ketogenesis) α cells β cells Glucose, etc β cell No Insulin Cryer P. Endocrinology. 2012; 153: 1039 -1048. Unger R, et al. J Clin Invest. 2012; 122: 4 -12. Bosco D, et al. Diabetes. 2010; 59: 1202 -1210. α cell Glucagon
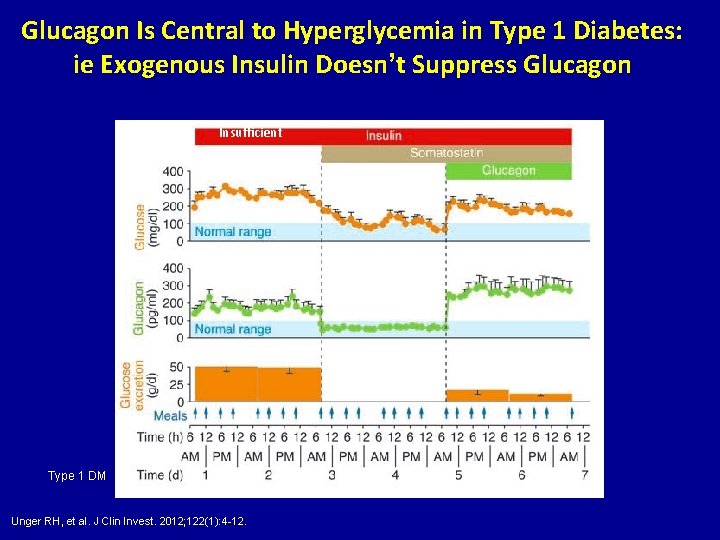
Glucagon Is Central to Hyperglycemia in Type 1 Diabetes: ie Exogenous Insulin Doesn’t Suppress Glucagon Insufficient Type 1 DM Unger RH, et al. J Clin Invest. 2012; 122(1): 4 -12.
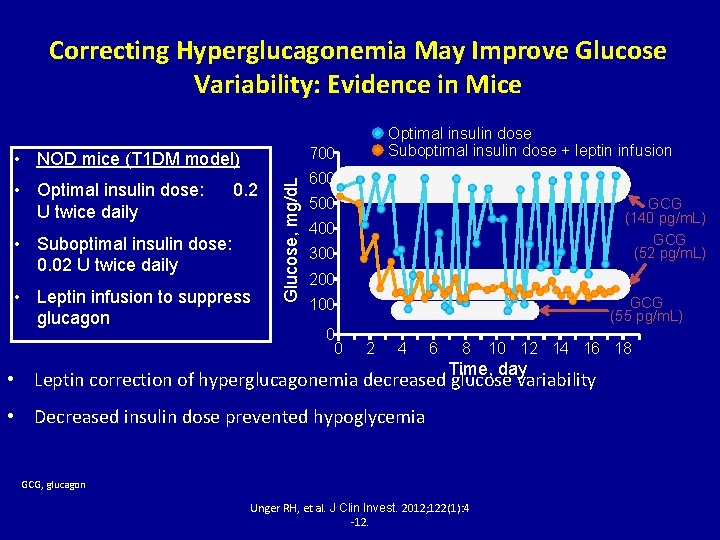
Correcting Hyperglucagonemia May Improve Glucose Variability: Evidence in Mice 700 0. 2 • Suboptimal insulin dose: 0. 02 U twice daily • Leptin infusion to suppress glucagon Glucose, mg/d. L • NOD mice (T 1 DM model) • Optimal insulin dose: U twice daily Optimal insulin dose Suboptimal insulin dose + leptin infusion 600 500 GCG (140 pg/m. L) GCG (52 pg/m. L) 400 300 200 GCG (55 pg/m. L) 100 0 0 2 4 6 8 10 12 14 16 18 Time, day • Leptin correction of hyperglucagonemia decreased glucose variability • Decreased insulin dose prevented hypoglycemia GCG, glucagon Unger RH, et al. J Clin Invest. 2012; 122(1): 4 -12.

Addressing the alpha cell in type 1 Diabetes • Glucagon dysregulation is a key contributor to increased risk of hypoglycemia and hyperglycemia in T 1 DM and T 2 DM • Increased risk of hypoglycemia can be mitigated by educating patients regarding signs, symptoms, and treatment of hypoglycemia and can be treated with exogenous glucagon • Approaches to correct hyperglucagonemia may improve glycemic control in patients with T 1 DM • At present only pramlintide is approved to treat the glucagon defect in Type 1 Diabetes
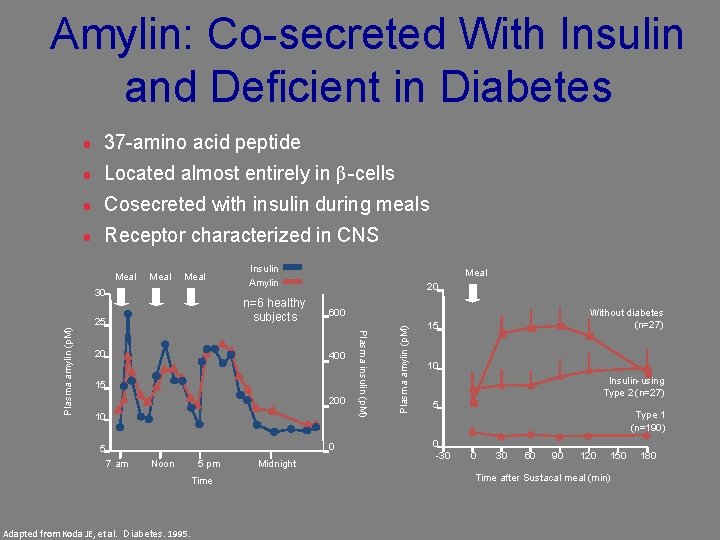
Amylin: Co-secreted With Insulin and Deficient in Diabetes ● 37 -amino acid peptide ● Located almost entirely in β-cells ● Cosecreted with insulin during meals ● Receptor characterized in CNS Meal 30 n=6 healthy subjects 25 20 Meal 20 600 400 15 200 10 0 5 7 am Noon 5 pm Time Adapted from Koda JE, et al. Diabetes. 1995. Midnight Plasma insulin (p. M) Plasma amylin (p. M) Insulin Amylin Plasma amylin (p. M) Meal Without diabetes (n=27) 15 10 Insulin-using Type 2 (n=27) 5 0 -30 Type 1 (n=190) 0 30 60 90 120 150 Time after Sustacal meal (min) 180
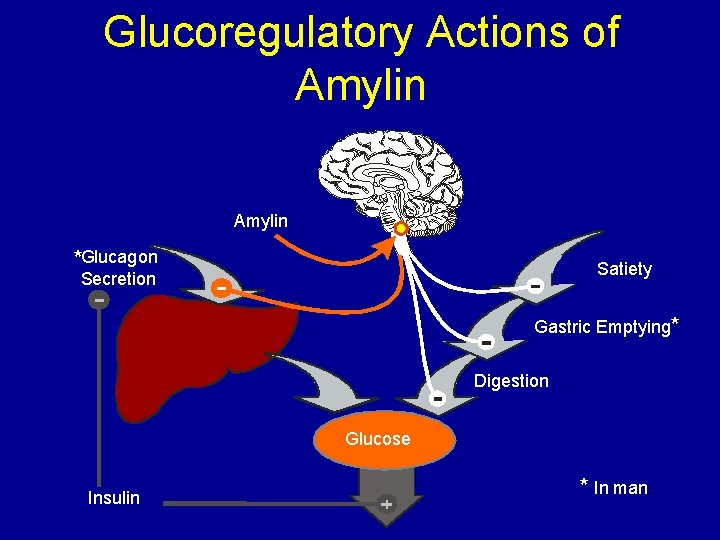
Glucoregulatory Actions of Amylin *Glucagon Satiety Secretion Gastric Emptying* Digestion Glucose Insulin * In man
- Slides: 13