Two Approaches Top Down Bottom Up Top down


Two Approaches • Top Down • Bottom Up Top down approach refers to slicing or successive cutting of a bulk material to get nano sized particle. Bottom up approach refers to the build up of a material from the bottom: atom by atom, molecule by molecule or cluster by cluster.

Top Down • Electron Beam Lithography • Plasma Etching • Ball Milling / Attrition

Bottom Up • Colloidal Synthesis • Sol-Gel Method • Co-precipitation method Mostly chemical methods Assembling atom by atom or molecule by molecule

The biggest problem with top down approach is the imperfection of surface structure and significant crystallographic damage to the processed patterns. These imperfections which in turn leads to extra challenges in the device design and fabrication. But this approach leads to the bulk production of nano material.

All the living beings in nature observe growth by Bottom Up approach only It plays an important role in the fabrication and processing of nano structures. On the contrary, top down approach most likely introduces internal stress, in addition to surface defects.

Modern Chemical Methods 1. Sol gel method 2. Chemical precipitation 3. Hydrothermal method 4. Gas phase chemical reactions 5. Inert gas condensation 6. Chemical vapor deposition 7. Mechanical alloying/High energy ball milling 8. Electro-deposition 9. Freeze drying 10. Spray drying 11. Precursor techniques 12. Non-aqueous liq. Phase reactions 13. Laser pyrolysis 14. Ion-insertion chemistry 15. Microwave Synthesis

QUANTUM DOTS ü Quantum Dots (QDs) are fluorescent semiconductor nanoparticles whose excitons are confined in all three spatial dimensions. ü Generally within the size range 2 – 10 nm. *depends on Bohr radius ü Also regarded as zero dimensional (0 D) points. ü Smaller the size of the crystal, larger is the energy gap. ü The dots can be grown to any needed size, allowing them to be tuned. ü Many interesting phenomenon like photoluminescence, multiple exciton generation.


The kind of method used for the synthesis of nanomaterials/quantum dots depends on the properties. Ideally, nanocrystals should have the following properties: 1. 2. 3. Monodispersity Possibility of further chemical derivatization High degree of crystallinity and specificity (avoiding polymorphic phases) 4. Chemical integrity 5. Lack of defects
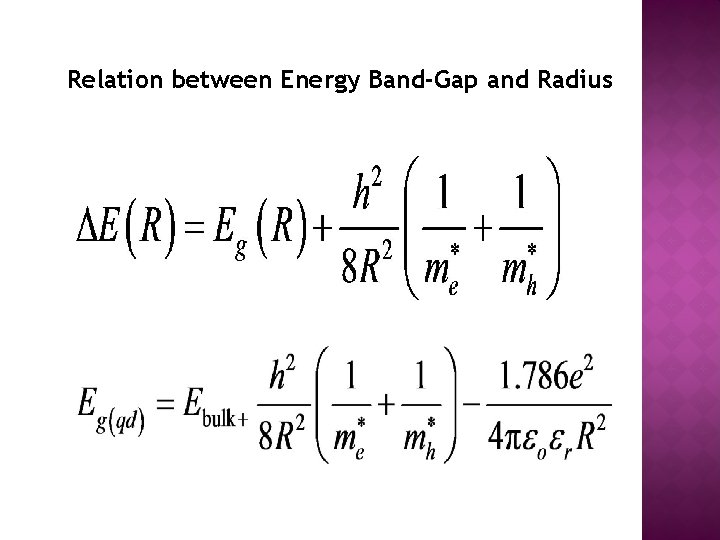
Relation between Energy Band-Gap and Radius
- Slides: 11